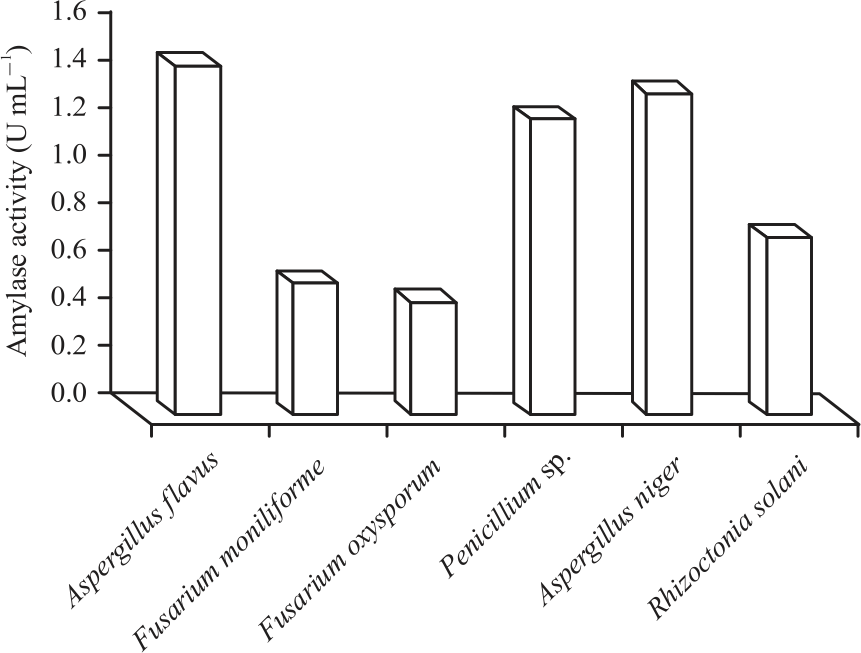
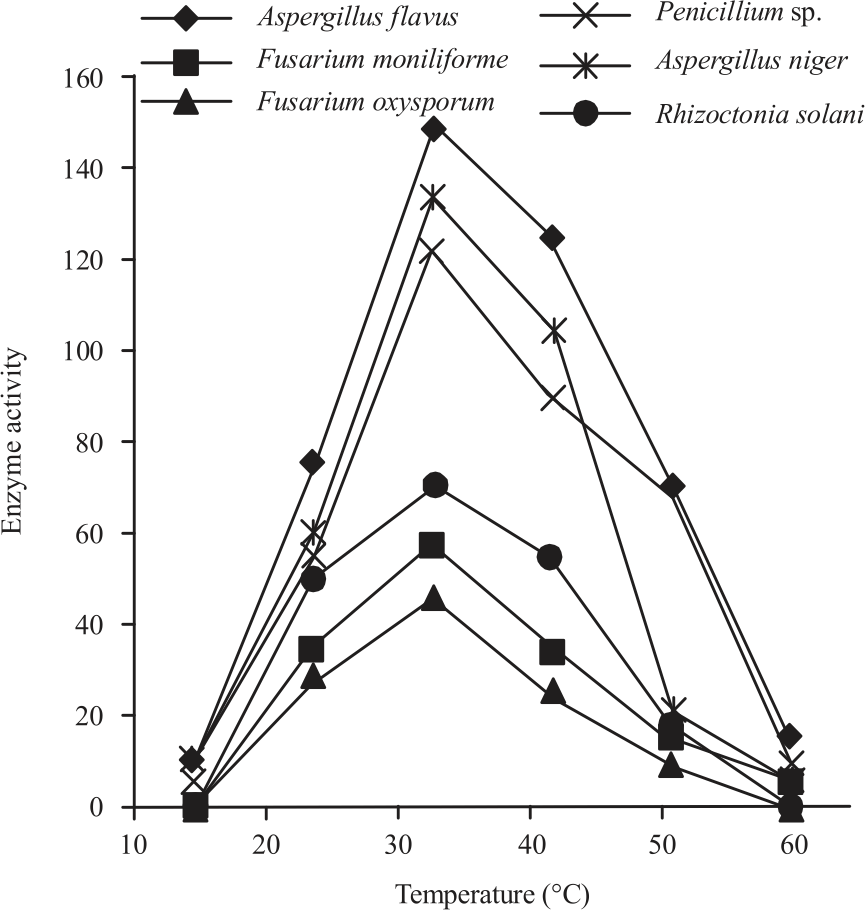
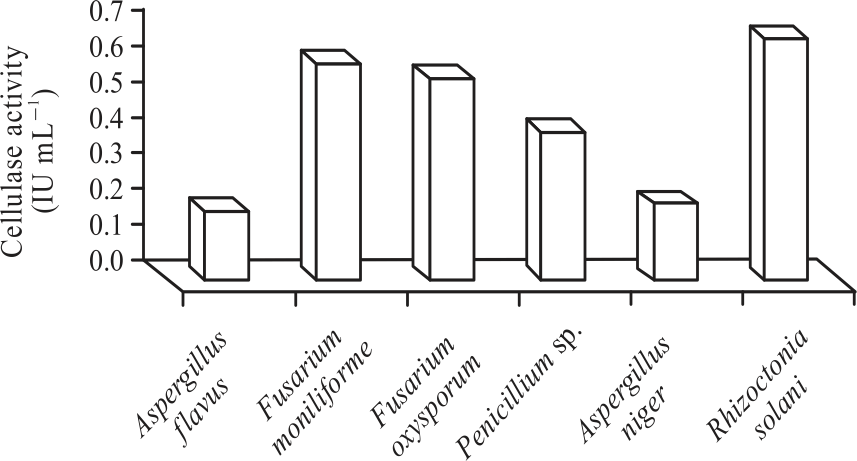
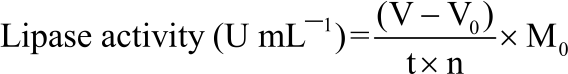Research Article
Fungi Associated with Peanut Seeds and Their Extracellular Enzyme Activities under the Influence of Physical Factors
Department of Seed Pathology Research, Institute of Plant Pathology Research, Agriculture Research Center, Giza, Egypt
LiveDNA: 20.29424