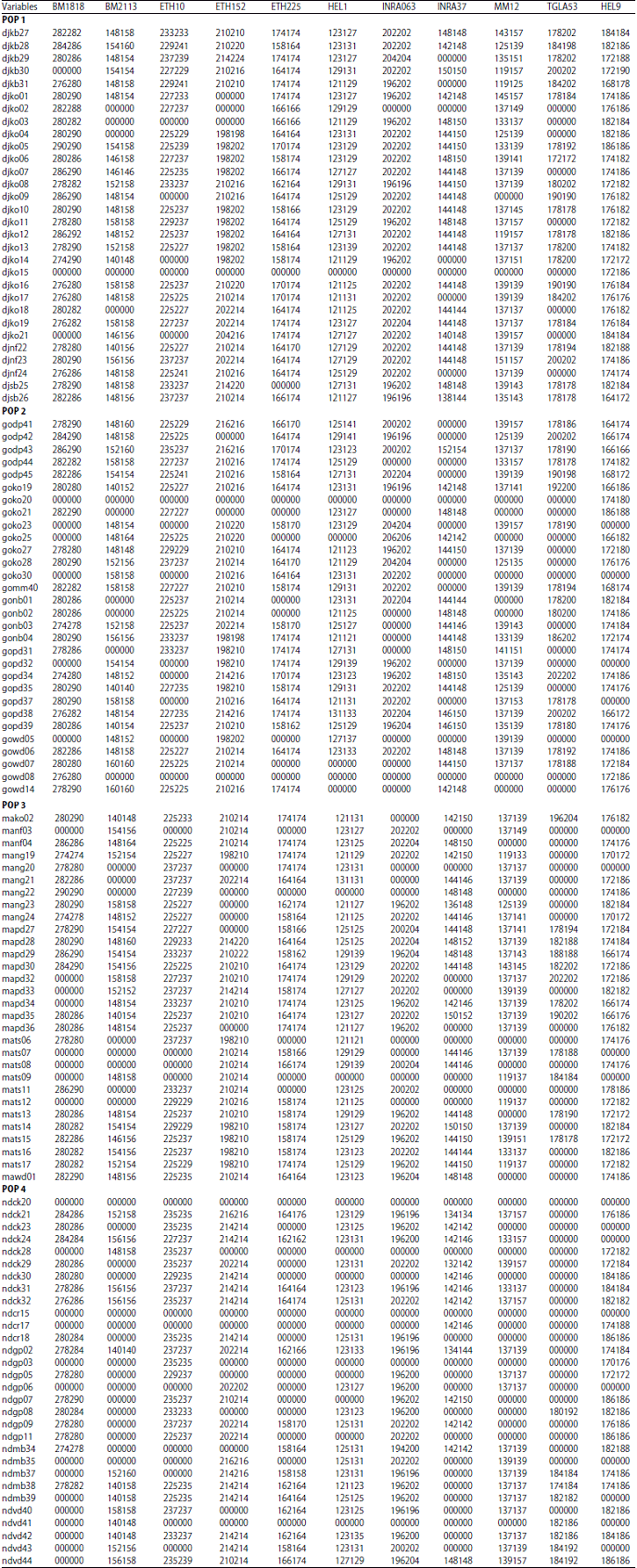
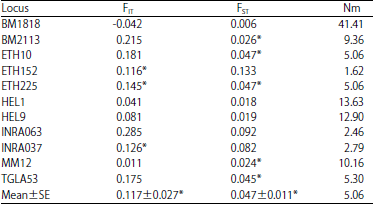
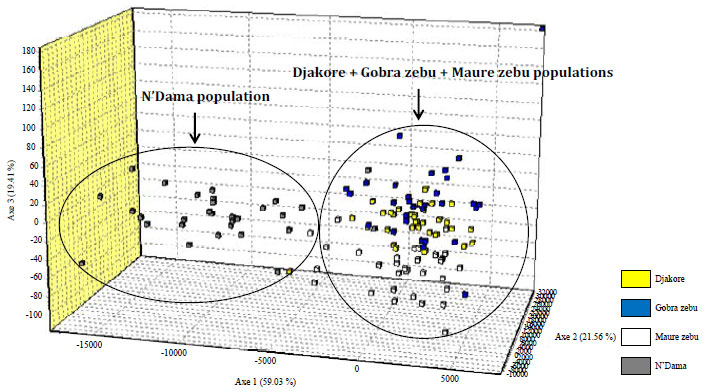
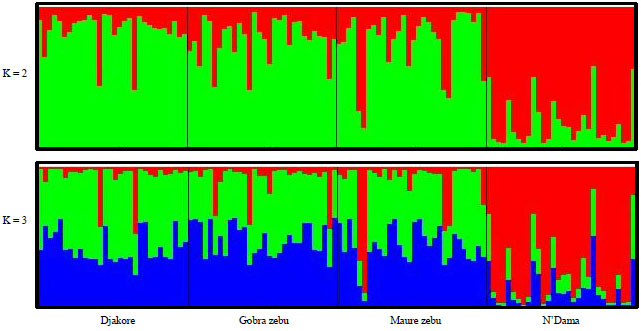
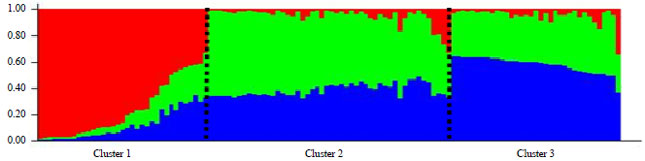
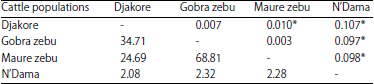
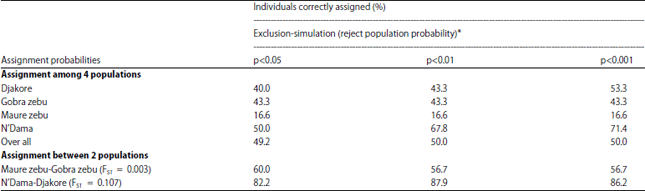
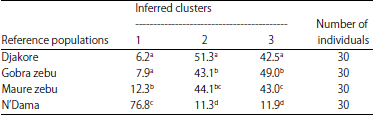
Research Article
Diversity and Genetic Structure of Senegalese Local Cattle Breeds
Department of Animal Biology, Faculty of Sciences and Techniques, Cheikh Anta Diop University of Dakar, P.O. Box 5005, Dakar, Fann, Senegal
LiveDNA: 221.25770
Adama Sow
Laboratory of Endocrinology and Radio-Immunology, Inter State School of Veterinary Sciences and Medicine of Dakar, P.O. Box 5077, Dakar, Fann, Senegal
Germain Jerome Sawadogo
Laboratory of Endocrinology and Radio-Immunology, Inter State School of Veterinary Sciences and Medicine of Dakar, P.O. Box 5077, Dakar, Fann, Senegal
Mbacke Sembene
Department of Animal Biology, Faculty of Sciences and Techniques, Cheikh Anta Diop University of Dakar, P.O. Box 5005, Dakar, Fann, Senegal