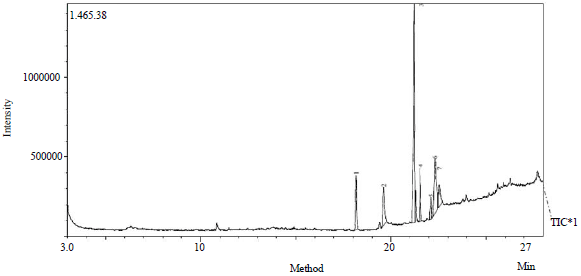
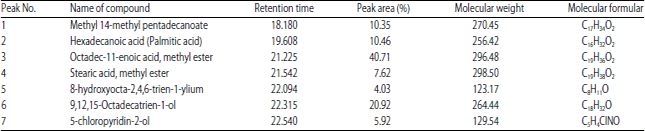
Research Article
Phytochemical and GC-MS Evaluation of Bioactive Principle of Vitis vinifera Peels
Department of Biochemistry, Ebonyi State University, P.M.B. 053 Abakaliki, Ebonyi State, Nigeria
LiveDNA: 234.25095
Uraku Oluchi Helen
Department of Biochemistry, Ebonyi State University, P.M.B. 053 Abakaliki, Ebonyi State, Nigeria
Nweke Friday Nwalo
Department of Biology/Microbiology/Biotechnology, Federal University Ndufu Alike Ikwo P.M.B. 1010 Abakaliki, Ebonyi State, Nigeria
Orji Obasi Uche
Department of Biochemistry, Ebonyi State University, P.M.B. 053 Abakaliki, Ebonyi State, Nigeria
Igwenyi Ikechukwu Okorie
Department of Biochemistry, Ebonyi State University, P.M.B. 053 Abakaliki, Ebonyi State, Nigeria
Edwin Nzubechukwu
Department of Biochemistry, Ebonyi State University, P.M.B. 053 Abakaliki, Ebonyi State, Nigeria
Ezeani Nkiru Nwamaka
Department of Biochemistry, Ebonyi State University, P.M.B. 053 Abakaliki, Ebonyi State, Nigeria