Research Article
Biodegradation of Keratinous Waste Substrates by Arthroderma multifidum
Department of Biotechnology,
Anima Sharma
Department of Biotechnology,
Seema Bhadauria
Department of Microbiology, JECRC University, Jaipur, Rajasthan, India
The keratinous wastes accumulate in nature mostly in the form of feathers, hairs, hooves, horns and nail clippings. These keratinous substrates considered as environmental pollutants and generated mostly from the poultry farms, slaughterhouses and leather industries1-3. Each year, globally 24 billion chickens are slaughtered and approximately 8.5 billion tons of feather are produced. According to a present report, India's part alone is 350 million tons4,5. The leather industries throw out wide amount of waste products and considered as extremely polluting industries with negative environmental impact6. The poultry feathers and other keratin-containing wastes are dumped, land filled and incinerated. These activities cause the soil, water and air pollution. Discarded feather, furthermore causes various human ailments including, chlorosis and fowl cholera7.
Keratin protein is major structural fibrous protein, providing an outer covering such as hair, wool, feathers and nails etc.8,9. Due to the strength and steadiness of keratin, only some microorganisms are able to break it down and degrade. Nature has provided the planet earth with an assortment of beneficial organisms. Keratinophilic fungi are one of the nature’s gifts and the largest group of organisms, which have the capability to degrade the keratin and their substrates10-12. Soils are rich in keratinous substrates and organic materials and the best candidate for the growth and occurrence of keratinophilic fungi13,14. The keratin-degradation ability of keratinophilic fungi has been credited with the production of the keratinase enzyme15-17. Fungal keratinase is the industrially significant enzyme offers bioconversion of keratinous wastes, utilization as animal feed supplements and dehairing agents in tannery in eco-friendly way18,19.
This study aimed to detect and evaluated the keratinous waste degradation by Arthroderma multifidum. The genus Arthroderma has rarely been described for the biodegradation of keratinous waste substrates, therefore, making this study important in the search and evaluation of the biodegrading potential of geophilic keratinophilic fungi.
Collection of samples: Soil samples were collected from poultry farm soil in Jaipur district, Rajasthan. Keratinous substrates, e.g., human hair, animal hair and chicken feathers were collected from the barber’s dump, animal habitat and poultry waste, respectively. These keratinous wastes were washed extensively with water and overnight defatted with chloroform: methanol (1:1, v/v) and dried again at 60°C. Keratin substrates were fragmented into small pieces.
Isolation of microorganism: The isolation of keratin degrading fungi was carried out using hair bait technique20. Antibiotic streptomycin (40 mg L–1) was added to prevent bacterial growth. After 21 days inoculum from fungus growth was taken and placed and maintained on plate of Sabouraud’s Dextrose Agar (SDA). The strain was identified by morphological traits as well as 18S rRNA sequence analysis.
DNA sequence analysis: A molecular characteristic of the strain was achieved by DNA sequence analysis of the ITS1-5.8S-ITS2 region. First, the fungus was grown in flask containing Sabouraud’s dextrose broth and incubated at 27°C for several days using an orbital shaking incubator (Remi CIS-24 Plus). After the colony growth, the genomic DNA was extracted by the Miniprep protocol with mild modification21. The frozen mycelium mass was smashed by mechanical pressure using sterile mortar piston and liquid nitrogen. The powder was mixed with lyses buffer and the DNA was extracted. The ITS1-5.8S-ITS2 rDNA was amplified using ITS4 and ITS5 universal primers22. Amplification was performed in 50 μL master mixture reaction volumes containing 5 μL of 10x buffer, 1 μL of dNTP (10 mM), 0.5 μL of ITS4 primer (10 pm), 0.5 μL of ITS5 primer (10 pm), 0.5 μL of Taq polymerase (3.0 U) with the addition of MgCl2 (15 mM) and 42.5 μL Milli Q water with 1 μL of DNA Sample (100 ng μL–1). The PCR reaction was carried out using a eppendorf Mastercycler Gradient Thermal Cycler with the following conditions: Initial de-naturation at 94°C for 10 min, 35 cycles of (1 min at 94°C for DNA de-naturation, 30 sec at 56°C for annealing and 1 min at 72°C for extension) final extension at 72°C for 10 min and storage at 4°C. Negative controls were also used in each set of reactions. The final products were analyzed by electrophoresis on 1.5% agarose (Himedia) and stained with 0.5 μg mL–1 ethidium bromide. The PCR products of the expected size were sequenced using ITS4 and ITS5 primers in an Applied Biosystem (ABI) Prism 3700 DNA analyzer at PGIMER, Chandigarh, India. Similarity analysis of the nucleotides were performed by using the web-based blasting program, Basic Local Alignment Search Tool (BLAST) searches against sequences available in GenBank23. After performing the morphological traits and DNA sequence analysis the organism was confirmed as Arthroderma multifidum.
Degradation of keratinous waste substrates: The degradation of keratinous substrates were assessed at different incubation period, i.e., 12 and 24 days in order to optimize incubation time. The keratin degradation activity of Arthroderma multifidum was tested in the modified Basal Salt Medium (BSM)24 and the pH was adjusted to 7.0. Arthroderma multifidum was inoculated into the flask containing BSM with 2.5% (w/v) keratinous substrates and incubated at 30°C on an orbital shaking incubator (Remi CIS-24 Plus) at 70 rpm for 12 and 24 days. A substrate control was also run along with the test flasks. The degradation by A. multifidum was expressed as percentage weight loss of keratinous waste substrates.
Concentration of hydrogen ions: A clear-cut confirmation to assess the progress of keratin waste degradation was obtained by observing the changes in hydrogen ion concentration of the basal salt media. The pH of culture filtrate was measured after every incubation period by using a digital pH meter (Model 181, Electronics India).
Determination of degree of degradation (DD): The percent of keratinous waste degradation was determined and calculated as degree of degradation (DD) by using following equation25:
where, IW is the initial weight of keratinous substrates and FW is the final weight of degraded keratinous substrates.
Microscopic view of degraded keratinous wastes: The control and degraded keratinous waste substrates were stained with cotton blue and observed under a light microscope (Olympus Research Microscope-Model CX21i) at different incubation days.
In this study, A. multifidum strain yielded a unique PCR amplification. The sequence of ITS1-5.8S-ITS2 rDNA region were 545 bp. The ITS data of the isolated strain of A. multifidum were identical to the ITS data of A. multifidum (GenBank Accession Number AB861837.1). Pair wise alignment data showed more than 99% identities, which confirmed the identification of the A. multifidum strain. GenBank Accession Number for isolated A. multifidum was KU560574 (Fig. 1).
In the present study, biodegradation of four different keratinous waste substrates (chicken feathers, human hair, animal hair and human nail clippings) were studied. Arthroderma multifidum in basal salt media hydrolyzed keratin substrates at pH 7.0 and 30°C under shaking condition. The degradation of chicken feathers was more rapid than other substrates. The degradation of keratin substrates was in the following order chicken feathers>animal hair>human nail clippings>human hair after 12 days incubation. After 24 days of incubation the degradation of keratin substrates degradation order was found chicken feathers>human nail clippings>animal hair> human hair. Arthroderma multifidum caused 21.2 and 27.6% degradation for human hair, 27.6 and 30.8% degradation for animal hair, 24.8 and 35.2% degradation for human nail clippings and 31.6 and 39.2% degradation of chicken feathers in shaking conditions after 12 and 24 days of incubation (Table 1 and Fig. 2).
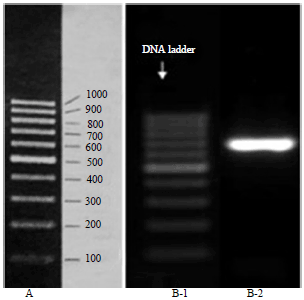 | |
| Fig. 1: | A: Gene Ruler of DNA ladder, B-1: DNA ladder and B-2: Electrophoretic pattern of PCR product of A. multifidum |
| Table 1: | Degradation of keratinous substrates by Arthroderma multifidum (optimum temperature = 30°C and optimum pH = 7.0) |
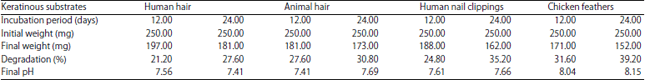 | |
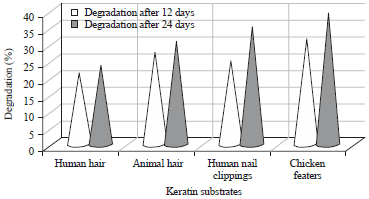 | |
| Fig. 2: | Degradation percentage by Arthroderma multifidum on keratinous substrates |
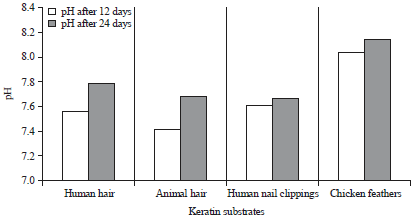 | |
| Fig. 3: | Changes in pH of basal salt medium after keratin degradation by Arthroderma multifidum |
 | |
| Fig. 4(a-b): | Microscopic view of keratin substrates, (a) Control chicken feather and (b) Degraded chicken feather |
The changes in pH were assessed after hydrolysis of keratinous wastes at 12th and 24th day of incubation. There was an increase in pH of the medium containing keratinous wastes after 12 and 24 days of incubation (Fig. 3). The Basal Salt Medium (BSM) shown the pH to be 7.56 and 7.79 for human hair, 7.41 and 7.69 for animal hair, 7.61 and 7.66 for human nail clippings and 8.04 and 8.15 for chicken feathers, respectively after 12th and 24th day of incubation.
For microscopic observations, keratinous substrates from control flasks showed no signs of degradation. In contrast, keratin substrates removed from experimental flasks, which showed varying degrees of digestion at different incubation days. Initial growth of the fungus in keratin substrates was intercellular; later on the hyphae were found mostly inside the cells. The digestion showed all features of an enzymatic breakdown (Fig. 4 and 5).
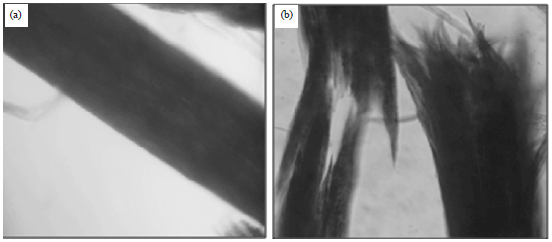 | |
| Fig. 5(a-b): | Microscopic view of keratin substrates, (a) Control animal hair and (b) Degraded animal hair |
Extensive quantities of keratinous wastes (chicken feathers and hair) are disposed annually by animal processing industry, leather industry and poultry farms causing a mounting ecological problem due to the extreme resilience of these materials26. Keratinophilic fungi with their keratinolytic potential play a significant role in keratinase mediated degradation of keratin wastes. These soil fungi could be effective in hastening keratin biodegradation27. Keratin hydrolysis in basal salt medium with microbial cultures is one of the most promising techniques not only to utilize this protein, but also to obtain valuable by products28,29.
This study describes the degradation of α and β keratins and also illustrates the changes of pH on the enzyme production during degradation by Arthroderma multifidum. The present study has exposed that A. multifidum has been the most active keratinophile on chicken feathers 31.6 and 39.2% after 12 and 24 days incubation, respectively. This is an agreement with the study of Kannahi et al.30 who also reported that the maximum degradation of hen’s feathers were noted in Fusarium solani (33.6%) and lowest keratin degradation noted in Aspergillus flavus (19.2%). Muhsin and Hadi31 who also reported that chicken feather was highly degraded by Aspergillus flavus (32%). El-Naghy et al.32 studied that the Chrysosporium georgiae degrade chicken feathers. Moreira et al.33 studied that Myrothecium verrucaria produced protease and hydrolyzed keratin substrates at pH 9.0 and 40°C in the following order: Poultry feather keratin>sheep wool keratin>human nail keratin> human hair keratin. Cavello et al.34 demonstrated that Paecilomyces lilacinus (LPS 876) efficiently degraded chicken feather during submerged cultivation producing extracellular proteases. Darah et al.3 stated that Microsporum fulvum IBRL SD3 was isolated from a soil sample collected from a chicken feather dumping site using a baiting technique and capable to produce keratinase significantly. The crude keratinase was able to degrade whole chicken feathers effectively.
In the present study, A. multifidum cause 21.2 and 23.6% degradation of the human hair after 12 and 24 day intervals in shaking condition. Deshmukh and Agrawal35 studied the ability of five keratinophilic fungi i.e., Auxarthron conjugatum, Chrysosporium indicum, Chrysosporium pannicola, Keratinomyces ajelloi and Microsporum gypseum to digest human hair in shake cultures. Muhsin and Hadi31 observed the rate of degradation, in which human hair had the highest degradation rate Chrysosporium pannicola 62% and Microsporum gypseum 48%. Kunert36 has also reported rapid digestion of human hair by Microsporum gypseum. The degradation percentage for the human nail clippings was 24.8 and 35.2% after 12 and 24 days incubation, respectively. Oyeka and Gugnani37 reported that Scytalidium hyalinum degraded 40%, whereas, Scytalidium japonicum degraded 38% of human nail clippings. In the similar study, in another case Fusarium solani degraded the human nail clippings (43%).
In this study, in case of animal hair weight loss was 27.6 and 30.8% after 12 and 24 days incubation period. Sharma et al.38 studied that Microsporum gypseum (49.34%) and Trichophyton verrucosum (49.34%) showed the greatest degradation of animal hair. These keratin substrates exhibited low degradation by Fusarium oxysporum (16.66%). In this study the changes in pH were assessed after hydrolysis of keratinous wastes. There was an increase in pH of the medium containing human hairs, chicken feather, animal hairs and human nails clipping on 12 and 24 days incubation in experimental flask. The tendency towards alkalization of the medium may be due to the hydrolysis and degradation of keratin substrates by keratinophilic fungi. Alkalinisation of the medium may be due to excretion of excess nitrogen via deamination and ammonium excretion39,40. Godheja and Shekhar41 also stated that the keratin degradation involves rupturing the disulphide linkage between the peptide chain of keratin molecules by some extra and intra cellular enzymes collectively called keratinase. Ingle et al.42 reported that maximum degradation of feathers was within the pH range of 6-7. From the above-mentioned results, it can be concluded that the keratinous wastes were degraded by Arthroderma multifidum effectively on 30°C and 7.0 pH. At this temperature and pH keratinase secretion is responsible for keratin degradation.
Arthroderma multifidum, which is keratinophilic in nature play an important role in the degradation of keratinous waste substrates. Future study on the use of this keratinophilic fungus for the production of biodegradable films, coatings, glue and natural gas for fuel from keratinous waste is to be conducted.
We thank to Director, School of Sciences for encouragement and Head, Department of Biotechnology, JECRC University, Jaipur and Department of Medical Microbiology, PGIMER, Chandigarh (India) for providing the opportunity and facilities to complete the study.