Review Article
Nematodes, an Emerging Threat to Global Forests: Assessment and Management
Department of Plant Protection, Faculty of Agricultural Sciences, Aligarh Muslim University, Aligarh 202 002, India
Forests, the world’s one of the precious natural resources, are essentially important for the survival of every kind of life on this planet. Significance of forest cover can be understood with the fact that existing one-third of the global geographical area (approximately 3952 million hectares) under forest cover is considered as shrunken natural flora and is a matter of serious concern to scientists and planners (Khan and Khan, 2010). Of the existing forest cover, 95% is natural forest and 5% is planted forest. About 47% of the forests world over are tropical, 9% subtropical, 11% temperate and 33% boreal (Maini, 2003). The forest resource of the world has shrunk by 40% since agriculture began 11,000 years ago. Three quarters of this loss has occurred during the last two centuries as land was cleared to make way for farms to meet the increasing demand for food and wood. The global wood harvest accounted for 3.4 billion cubic meters in 2004, 52% of this is used as fuel (Lee, 2008). The decreasing forest area has led to a change in the biodiversity creating ecological imbalance world over (Khan and Khan, 2010).
Diversified flora in forests provides ideal conditions for survival and multiplication of an array of micro and macro-organisms. Among plant pathogenic microorganisms, the researches have been mostly dealt with fungi and bacteria and least consideration has been given to nematodes, as the damage caused by them remains indistinctive and is not easily recognized. There is limited research that has focused on nematode infestation in forests. Nematodes are world wide in distribution; their abundance depends on many factors such as soil type (Chirchir et al., 2008) and vegetation type (Sahu et al., 2011). However, development of plant diseases including those caused by nematodes depends largely on environmental factors besides the host susceptibility (Khan and Anwer, 2011).
Since nematodes are generally soil inhabitants and attack underground parts of plant, the damage caused by them cannot be diagnosed by symptoms alone, general symptoms mostly resemble with those of several other biotic agents or some nutrient deficiency (Khan, 2008). However, in nurseries, the infested seedlings could be identified on the basis of vigor, small, distorted root system, root galls and general decay of the feeder roots. In addition to causing direct damage, nematodes may also act as vector for plant viruses (Taylor and Brown, 1997) and facilitate the infection by several fungi and bacteria (Khan and Dasgupta, 1993). Nematode infestation also adversely affects activity and association of beneficial soil microbes such as mycorrhizal fungi, rhizobia, Frankia etc. (Khan, 2010).
Various aspects of nematode infestations in plants such as nematode species composition, nature of damage, host range, histopathological, biochemical, ecological and management aspects on agricultural crops including food crops (Khan and Jairajpuri, 2010a), industrial crops (Khan and Jairajpuri, 2010b) and horticultural crops (Khan and Jairajpuri, 2012) have been adequately studied. But in forestry, there is limited information on species composition under different forest trees. No information is available on the host range of some of the potential nematode pests of forests and the annual losses caused by them to the timber industry. The genera and species composition of plant nematodes in forest ecosystem depend on the species of plants growing in the area, prevailing environmental conditions and presence or absence of nematode vectors. The known destructive nature of these worms on other woody crops suggests that they may also be involved in causing some of the unexplainable losses in vigor and mortality of forest trees. Due to the minute size, nematodes often get neglected and the losses caused by them go on increasing year after year leading to forest decline (Khan and Khan, 2010).
Nematode infestation in major forest trees: Self sustaining habitats like forest ecosystems are generally rich in energy cycling and soil, the major sink of activities generally contains adequate amount of organic matter. Moderate temperature coupled with available moisture and organic nutrients make a suitable habitat for root growth and nematode activity (Khan and Khan, 1995). Yeates (2007) recorded more than 400 nematode species at a population of 10x106 nematodes/m2 in the terrestrial ecosystem rich in litter. Root-feeding nematodes may be pathogenic to young plants; microbial-feeding nematodes may increase turnover of the microbial pool; while predacious and omnivorous nematodes represent higher trophic levels. Yeates (1979) reported the average nematode population in the range of 39x105 m-2 for Tundra Biome, 28.11x104 m-2 for 9 deciduous forests and 56x103 m-2 for tropical forests. The spatial distribution and abundance of nematode species in forests reflect soil type, soil fertility, climate, canopy, plant species, litter depth, forest age etc. Although, there are certain instances where nematodes have caused enormous damage to huge trees but in most cases they are not known to cause disease to forest trees with some exceptions. The damage to the forests may be direct due to decay and malfunctioning of feeder roots. The wounding and other modifications induced by the nematodes in the host may greatly facilitate the passage and pace of infection of the secondary pathogens (Khan and Dasgupta, 1993). Nematodes often interact synergistically with fungi and bacteria to form disease complexes (Khan, 1993). In addition, nematodes of the family Longidoridae and Trichodoridae vector NEPO and tobra viruses respectively and introduce them in to susceptible plants and trees (Taylor and Brown, 1997). The presence of fungal feeding nematodes in forest soils may lead to the reduction in root colonization by the mycorrhiza and subsequently the reduced plant growth (Francl, 1993).
Nematodes have very wide host range and attack all kinds of plants. Besides crop plants nematodes to attack numerous angiospermic and gymnospermic trees which includes commercially important wood and timber trees. The major trees and the associated nematode problems are discussed in the following paragraphs.
Angiospermic trees
Eucalyptus species: Eucalyptus grow under sub-tropical, tropical and temperate climate and serve as an important tree species of social forestry. Numerous nematodes have been reported from the root zone of the Eucalyptus (Table 1), but there is lack of evidence for their pathogenicity. Khair (1987) recorded the association of 22 genera and 41 species of nematodes with 45 species of Eucalyptus. Commonly occurring nematodes in the root zone of Eucalyptus are Criconema, Criconemella, Cryphodera, Helicotylenchus, Hemicycliophora, Meloidogyne, Morulaimus, Paralongidorus, Paratylenchus, Pratylenchus, Radopholus, Scutellonema, Tylenchorhynchus, Tylodorus and Xiphinema (Andrassy, 1986). Two species of Xiphinema, X. parasetariae and X. souchaudi were found to be the potential threat to Eucalyptus plantations in the Congo (Baujard et al., 1998). X parasetariae attacks the root apex and prevents normal root growth (Wallace, 1973). The resulting wounds enable certain bacteria and fungi to infect the plant and thus results in reduced vigor in young plantations. Hoplolaimus indicus has also been reported to infest Eucalyptus sp. (Anonymous, 2001).
In North Queensland Pateracephalanema alticolum was found attacking E. polycarpa (Ruehle, 1968). Acontylus vipriensis. Helicotylenchus dihystera and Pratylenchus brachyurus are the other important nematodes associated with Eucalyptus (Lordello, 1967). Infestation with P. brachyurus leads to reduced root system, necrosis and cracking in seedling and young trees of E. alba and E. saligna and subsequently stunting, chlorosis and plant death (Lordello, 1967).
| Table 1: | Commonly occurring plant parasitic nematodes encountered with some forest trees |
 | |
Shah and Chaudhary (1975) recorded 24 genera of plant parasitic nematodes from Eucalyptus plantations in Pakistan. There are also some reports of Meloidogyne infestation in Eucalyptus from East Africa (Whitehead and Kariuki, 1960) and Latin America (Garces, 1964). However, Ibrahim and Kandeel (1986) reported that Eucalyptus are resistant to M. incognita, M. javanica and M. arenaria. Corymbia citriodora was found resistant to P. brachyurus but was susceptible to M. javanica as giant cells and egg masses were formed (Ferraz, 1982). Eucalyptus deglupta and koa are poor hosts to root-knot and reniform nematodes as these plants do not support reproduction of M. javanica, M. konaensis or Rotylenchulus reniformis. Koa, however, was susceptible to moderate population levels of root-knot nematodes.
Another important parasite of Eucalyptus is Fergusobia tumifaciens (Fergusobidae). The nematode is associated and transmitted by the gall flies, Fergusonina species. (Fisher and Nickle, 1968; Siddiqi, 1986). Fergusobia-Fergusonina galls are strictly restricted to the family Myrtaceae and have been reported from 41 species of Eucalyptus, 4 species of Corymbia, 2 species of Angophora, 8 species of Melaleuca, one species Callistemon and Syzygium cumini (McLeod et al., 1994; Siddiqi, 1986, 1994). Nematode has unique life cycle in which it is completed partly in the insect host and rest inside the galls on leaves, buds and flowers of Eucalyptus. The nematode and fly interaction is an example of mutualism as both the partners are partly responsible for the gall formation on leaves and flowers. Four types of galls viz., shoot and inflorescence bud galls (terminal and axial), leaf galls (flat callus like, terminal or axial), stem galls and flower bud galls are formed by the nematode-fly complex (Currie, 1937; Giblin-Davis, 1993; Giblin-Davis et al., 2001). Gall formation on Eucalyptus leaves leads to poor development of overall vigor of the tree with reduced foliage. Seed setting and oil production are greatly affected (Currie, 1937). The fly feeds and oviposits on the leaves of E. stuartiana or flower buds of E. macrorhyncha during summer. The nematode larvae (1-50) are deposited along with the insect eggs in buds which reproduce parthenogenetically. The nematode larvae feed on the primordia of the anther forming a circle around the inner wall of the flower bud cavity. It is thought that the larvae cause proliferation of primordia of buds producing irregular masses of thin walled parenchyma cells filled with sticky sap. However, the insect eggs hatch after six months and the emerging larvae cuts a small crypt between two opposed masses of the proliferating cells and start feeding. This is followed by the aggregation of nematodes around the fly larvae. The tissue in the crypt fuses to form a stalked gall-let. It has been reported that a single flower bud of E. macrorhyncha can have 20 gall-lets (Currie, 1937). Competition for resources occurs between the nematode and vector fly, as a result fecundity of the later is known to be affected in the galls. By the time of appearance of the 3rd instar larvae of the fly, the fertilized female nematodes are available to enter into the insect larvae prior to pupation. Inside the pupa the nematode enlarges to a parasitic female with no stylet or gut and filled with ovary. The parasitic nematode is always associated with the fat bodies of the pupa (Currie, 1937) and absorbs nutrient through highly specialized cuticle or epidermis.
Acacia species: Acacia spp. are versatile tree and grow in all kinds of soils. They are also planted in agro-forestry systems and are capable of reducing inert gaseous nitrogen to organic forms through rhizobial symbiosis. This allows woody legumes to grow in nitrogen deficient soils. Moreover, much of the nitrogen fixed by rhizobia in the nodules of Acacia roots is returned to the soil as humus that improves the soil fertility. Root-knot nematodes, Meloidogyne spp. are the most abundant phytonematodes encountered under Acacia plantation (Loubana, 1996). Ruponnois et al. (1999) tested the susceptibility of four Acacia species to the root-knot nematode, M. javanica and noted that Faidherbia albida and Acacia senegal were resistant to the nematode. On the contrary, A. raddiana, A. nilotica and A. mangium were susceptible. Meloidogyne javanica suppressed the growth of A. nilotica and A. mangium but not of A. raddiana (tolerant). The rhizobial symbiosis with F. albida and A. senegal was stimulated by the nematode. The most common symptom of the nematode infestation is the formation of galls followed by the root decay (Fig. 1).
The host status of 13 indigenous and exotic species of Acacia grown in the pots with M. javanica infested soil was tested under artificial inoculated condition (Ibrahim and Aref, 2000). Seedlings of Acacia were inoculated with 5,000 eggs of M. javanica/plant 30 days after transplanting. Acacia salicina expressed resistance to M. javanica while, A. farnesiana, A. gerrardii subsp. negevensis var. najdensis, A. saligna, A. nilotica and A. stenophylla were found highly susceptible. However, A. ampliceps, A. ehrenbergiana, A. gerrardii subsp. negevensis var negevensis, A. sclerosperma, A. seyal, A. tortilis and A. tortilis subsp. spirocarpa showed moderate susceptibility. Inoculation of Acacia nilotica seedlings with more than 50 J2 of M. incognita led to a significant reduction in the shoot length 60 days after inoculation (Veeraragavathatham et al., 2002). Swain and Dash (2010) inoculated A. mangium seedlings with 10, 100, 1000 and 10000 J2 of M. incognita. The plant growth was reduced with all treatments being greatest at the highest inoculum level. The population of 100 J2 or more caused economic injury to Acacia plants.
Loubana and Reversat (1996) has reported occurrence of Xiphinema spp. abundantly in Acacia rhizosphere. Three genera of plant parasitic nematodes viz., Scutellonema cavenessi, Tylenchorhynchus germanii and Hoplolaimus pararobustus were recorded in the root zone of Acacia (Dupponis et al., 2000). Important plant parasitic nematodes which infest Acacia roots, have been enlisted in Table 1. Six plant parasitic nematodes in A. nilotica roots were encountered infesting acacia growing in six villages of Coimbatore district in India. Among them, Helicotylenchus dihystera, M. incognita and T. brassicae were more frequently encountered. The frequency of occurrence of these nematodes was 22, 48 and 8%, respectively.
 | |
| Fig. 1(a-d): | Root galls caused by Meloidogyne sp. on (a) Acacia, www.ctahr.hawaii.edu and (b) Ficus, www.zera3y.com/.../02/root-knot-nematode.jpg, (c) Leaf chlorosis and necrosis in Nothofagus obliqua caused by Subanguina chilensis, www.ipmimages.org and (d) Wilting of needles of a branch of pine tree caused by Bursaphelenchus xylophilus, www.eppo.org |
Ficus species: Ficus is a terrestrial afro-tropical tree used as an antiepileptic remedy in African traditional medicine (Woode et al., 2011). It has also shown antifungal activity and phytochemical analysis of column chromatographic fractions of stem bark extracts of Ficus has been found effective for the treatment of fungal diseases (Hassan et al., 2007). Fig is an important Ficus and is grown as ornamental, edible and Capri fig. The figs are quite susceptible to nematode attack (Table 1). Studies conducted in the Netherlands have shown that Ficus spp. are good hosts of Meloidogyne enterolobii (EPPO, 2008). Brickell and Zuk (1997) reported that Ficus plants are attacked by root-knot nematodes. Meloidogyne incognita is another species of root-knot nematode that infects Ficus and causes extensive galling on the roots (Fig. 1). Aphelenchoides spp. are parasitic to Ficus elastica (Holtzmann, 1968). De Maeseneer (1964) reported invasion of leaves of F. elastica decora by A. fragariae resulting to leaf necrosis. The Ficus species susceptible to the nematode are F. elastica, F. elastica decora variegata, F. religiosa, F. benghalensis and F. rubiginosa.
Heterodera fici, the fig cyst nematode, is known to attack both edible and ornamental fig species (Sher and Raski, 1956). Other Ficus species e.g. rubber plant (F. elastica) are also attacked by the cyst nematode (Vovlas et al., 1989). Infestation of H. fici has been reported in edible fig orchards, F. carica in the Mediterranean region and in Pakistan (Golden et al., 1988). Among the ornamental figs, F. benghalensis, F. elastica, F. lyrata and F. rubiginosa are susceptible to nematode attack (Mulvey and Golden, 1983; Sher and Raski, 1956).
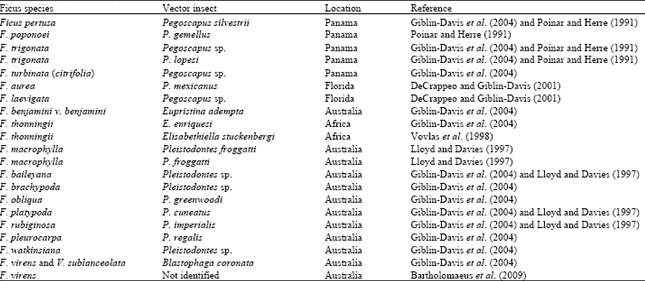
Another important nematode that attacks fig is Schistonchus caprifici (fig nematode) which causes galls on leaves and flowers of edible and Capri figs (Greco, 1992). The nematode is transmitted by fig wasp, Blastophaga psenes. The nematode juveniles are deposited into pistillate florets during oviposition. The nematode development and reproduction result in necrosis and cavity formation in the cortex (Vovlas et al., 1992). Both male and female florets are attacked but in Capri figs female florets are preferred most by the nematode. There are other genera of fig wasp viz., Pleistodontes, Pegoscapus and Eupristina that can transmit fig nematode (Table 2). Two more species of Schistonchus viz., S. guangzhouensis and S. centerae have been described from Ficus hispida in China (Zeng et al., 2007).
Syzygium cumini: Besides ectoparasites, jamun tree is attacked by an important foliar nematode, Fergusobia sp. which is transmitted by fergusoninid fly, Fergusonina syzygii. In India (Hyderabad) the adults, larvae and galls of the fergusoninid fly were described from material collected from jamun plum (Harris, 1981). This was the first record of occurrence of a fergusoninid fly outside Australia and on a host plant other than Eucalyptus. The nematode, Fergusobia sp. is introduced by the fergusonid fly in to jamun leaves where the nematode incites galls. As a result of gall formation, the leaves become chlorotic and the galled portion becomes non-photosynthetic. The infected trees become poor in growth with reduced vigor and gradually tree decline occurs. The nematode reproduces inside the galls where larvae of the fergusonid fly also develop and mature.
| Table 2: | Ficus species susceptible to Schistonchus species and their vector insects |
 | |
Bamboo: Bamboos are intermediate forms between grasses and coniferous wood trees and are grown in every climate. In India over 125 bamboos species are found but Bambusa and Dendrocalamus are the dominating genera. Nematodes parasitizing bamboo are Pratylenchus, Xiphinema, Meloidogyne, Longidorus, Paralongidorus, Trichodorus and Criconemoides. Among all nematodes, Pratylenchus brachyurus and Xiphinema americanum are the most common and damaging ones. Nematode attack facilitates the invasion by soil borne fungi and bacteria resulting in severe damage to bamboo plantations.
Nematodes feed on below-ground parts of bamboos and injure the root system. Pratylenchus brachyurus and other members of this genus cause root lesion that mainly appear on the young feeder roots of bamboo plants. The root necrosis followed by secondary rotting is a characteristic symptom of Pratylenchus infestation. Xiphinema spp. cause maximum damage to bamboo production and inflict about 15% yield loss while, P. brachyurus and other species of the genus are responsible for 10-12% bamboo loss. Root-knot nematode may cause 4-5% decline in the bamboo production, whereas, 2-3% loss has been reported due to infestation with Trichodorus, Longidorus and Paralongidorus spp. In addition to direct damage, nematodes synergies fungal pathogens leading to greater damage to bamboo (Gantait, 2010).
Nothofagus obliqua: Roble beech, Nothofagus obliqua, also known as, hualle or coyán is a deciduous tree grown in temperate forests (Salas et al., 2006). The tree is known for its hardiness, as it can tolerate a temperature as low as -8°C and snow cover for a couple of weeks. The wood is valued for its durability and is used in furniture and construction. The beech tree is susceptible to a foliar nematode, Subanguina chilensis that is transmitted by some insect vector. The nematode infected leaves of N. obliqua and N. alpine (= N. nervosa) show yellow and brown necrotic areas (Fig. 1) and finally there is premature defoliation. The defoliation weakens the plant and increases its susceptibility to secondary pests such as wood-boring insects and other pathogens (FAO, 2004). In certain areas the nematode is responsible for the decline of roble beech.
Santalum album, Shorea robusta and Tectona grandis: Sandalwood tree is a precious wood yielding tree. These trees in India, are mainly distributed on the Deccan Plateau. The total extent of its distribution is around 9000 km2 of which 8200 km2 is in the states of Karnataka and Tamil Nadu (Khan and Khan, 2010). Nayak et al., (2004) recorded 13 nematode species to be parasitic on different plant species including sandal wood. Wilt disease caused by Fusarium and Phytophthora is common in nursery beds of sandal plants. The disease becomes exacerbated in presence of plant parasitic nematodes resulting in seedling mortality (Remadevi et al., 2005).
Sal forests are important source of valuable wood. Rotylenchulus spp. are predominant nematode species in sal forests. Other nematodes which attack sal are Hoplolaimus seinhorsti, Hemicycliophora oostenbrinki. Hemicriconemoides and Orientylus species. These nematodes were recorded in the deeper soil layers (Pradhan and Dash, 1987). Important plant nematodes recorded in the root zone of sal are enlisted in Table 1.
Tectona grandis is an other good quality wood yielding tree. Xiphinema insigne has been found infesting the root of this tree (Thapa and Ganguly, 1990). However, the population of Paralongidorus sali was greater in sal than teak in the forests of Dehradun. The root-knot nematodes have been found most prevalent on sal and teak plants (Nayak et al., 2004). However, Caloosia exilis was parasitic on sandalwood trees. In addition, X. americanum, Macroposthonia xenoplax, Hemicriconemoides cocophilus and Tylenchorhynchus indicus were also present in good numbers in the forest. Nematodes of the genus Pratylenchus are parasitic on Shorea robusta, Tectona grandis and Santalum spp. In addition to sal and teak, Pratylenchus species have been reported to infest other tree species including Tamarindus indica in Bhavanisagar, India (Veeraragavathatham et al., 2002).
Gymnospermic trees: Gymnospermic trees constitute significant part of forest flora and are susceptible to a large number of pathogens (Khan and Khan, 2010). The nematode infestation and damage is more severe in coniferous trees especially pines. Numerous nematodes viz., Bursaphelenchus, Aphelenchoides, Tylencholaimus, Trichodorus, Helicotylenchus, Rotylenchus, Criconemoides, Hemicriconemoides, Tylenchorhynchus, Pratylenchus and Paratylenchus have been found commonly infesting coniferous and deciduous trees (Boag, 1974). Among these, pine wilt nematode, Bursaphelenchus spp. is a very important nematode pest of gymnospermic wood yielding trees in hilli areas (Netscher, 1970).
Pine wilt nematode: The Pine Wilt Nematode (PWN) belonging to the genus Bursaphelenchus has attracted the attention of nematologists around the world and has assumed the status of a quarantine pest in different countries (Zhao et al., 2009). This nematode is also called as Pine Wood Nematode (PWN) or timber nematode. There are 55 described species of Bursaphelenchus, of which approximately 75% are associated with the coniferous trees (Braasch, 2001). All Bursaphelenchus species are mycophagous and majority of them have a phoretic relationship with bark beetles and wood borers (Massey, 1974). Apart from two species i.e., B. xylophilus and B. mucronatus there is negligible knowledge of pathogenicity of other Bursaphelenchus species.
The pine wilt nematode is a native of North America, but does not cause damage to native pines, rather it attacks non-native pines that include Austrian pine (Pinus nigra), Scotch pine (Pinus sylvestris), Japanese red pine (Pinus densiflora) and black pine (Pinus thunbergii) in different countries (Table 3). However, there are other conifers as well which serve as potential hosts to the nematode e.g., Larix, Abies and Picea spp. Some of the important conifer hosts of B. xylophilus have been enlisted in Table 3. Isolated cases of death of Picea and Pseudotsuga due to pine wilt nematode infestation have been reported from USA (Malek and Appleby, 1984). In Asia (Japan), PWN was introduced in 1905 with pine logs imported from North America. Thereafter, the nematode has spread to various parts and is now a serious threat to pine forest in the country (Shi et al., 2008). The nematode is also a serious pest of pine in China, Taiwan and Korea (Agrios, 2005). It has also been reported from Vietnam and Australia. In Taiwan, the PWN was reported in 1985 on Japanese black pine and luchu pine (Pinus luchuensis). Pine wilt is the most serious disease in Japanese pine forests with a yearly damage of 800,000 m3 wood while 3 million mature pines are reported to die every year in China. Taiwan red pine (Pinus taiwanensis) and Japanese black pine (Pinus thunbergii) are highly susceptible and over 50% plants get infected either at the nursery or in the forests (Chang and Lu, 1996). In 2003, PWN killed about 1 million pine trees in Japan (Forestry Agency, 2004).
| Table 3: | Pine and other coniferous trees susceptible to Bursaphelenchus xylophilus |
 | |
Symptoms: The PWN, B. xylophilus is the greatest threat to pine forests in Europe and the southern hemisphere as the nematode causes sudden and extensive death to the host trees (Dwinell, 1997; Mota et al., 1999). Wilting, graying and browning or reddening of needles is the most prominent symptom of pine wilt followed by rapid decline and death of the entire tree within a few weeks or months. Symptom development is rapid and uniform during the warmer months of the year and all the needles die at once in young to middle aged trees. The needles first turn light grayish green in color. By this time, resin flow ceases and the tree starts wilting, drying and dying. The color changes to yellowish green then to yellowish brown and finally turns brown (Fig. 1). For a long time, it was thought that the PWN was solely responsible for the wilting in pines through production of phytotoxins and cellulases produced by the PWN and ethylene and terpenoids produced by the host pine stimulated by the nematode invasion (Nickle et al., 1981). Recently, it has been found that some bacteria are symbiotically associated with the PWNs and may play some roles in the pathogenicity of the disease (Zhao et al., 2005).
Wilting of needles varies with the pine species. Longer and soft needle species of pines show a wilting of needles over the entire tree, as if its source of moisture has been suddenly cut off. Most tree mortalities occur from late summer to late fall. Towards the end of this period, symptom development slows down and becomes less uniform over the tree. Some infected trees survive the winter, usually with one or more dead branches (flags) that stand out against healthy portions of the tree. The flags are usually located in the upper crown but can occur anywhere in the tree. Up to 50% of the crown of a live tree may be flagged. Pruning out these branches seldom eliminates the disease because the nematode usually has already reached the trunk. Spring mortalities frequently show interrupted candle growth, wilting of young candles and the color changes first in second-and third year needles. On Austrian pine, the nematode-induced branch flagging can be confused with branch dieback caused by the fungi Sphaeropsis (=Diplodia) pinea, particularly when the dieback is rapid after prolonged wet weather. The drying in old Scotch pines progresses from upward to downward direction. It may take a year for complete death of the tree.
The movement and feeding of the nematode in the trunk cause destruction of parenchyma cells of resin canals. Cell death occurs due to granulation of the cytoplasm and brown cell contents are frequently observed among axial and radial xylem parenchyma cells of stem 3 days after inoculation (Mamiya, 1976, 2008). On the sixth day of nematode inoculation, death of axial and ray parenchyma cells of seedlings with reduced oleoresin exudation was observed. After complete ceasing of oleoresin exudation there was destruction of parenchyma cells of axial and radial resin canals, ray, cambium and phloem.
Pine wilt nematode and its spread: The dauer stage of the nematode (fourth-stage juveniles) are transported to new host trees by cerambycid beetles of the genus Monochamus alternatus in Japan, China, Taiwan, Lao, Korea Republic (Mamiya and Enda, 1972), whereas, M. carolinensis (Olivier), M. mutator LeConte, M. scutellatus (Say) and M. titillator (Fabricius) in North America (Edwards and Linit, 1992). The PWN is transmitted to conifers by the beetles either during feeding on the bark and phloem of twigs of live trees (primary transmission) or during oviposition by female beetles in freshly cut timber or dying trees (secondary transmission). The geographical distribution of various Monochamus spp., host species and their role in vectoring the pine wilt nematode has been studied in detail (Skarmoutsos and Michalopoulos, 2000). Nematodes introduced during primary transmission reproduce rapidly and spread through resin canals causing the tree to wilt. The wilted branch or entire tree dies within weeks under favorable conditions. The PWN has two types of life cycles one is phytophagous on pine trees and the other is mycetophagous in which the nematode feeds on blue stain fungi (Ceratocystis or Ophiostoma piceae) and probably Botrytis cinerea also.
There is no report on the occurrence of pine wilt nematode in Bhutan, Nepal, India, Pakistan, Afghanistan, Iran etc., but the increased legal or illegal movement of raw wood, wood products and packing material across the border has enhanced the risk of the introduction of the PWN in the Himalayan region especially from China and in the similar way it was introduced into Japan, China, Taiwan etc. The prevailing climatic conditions and susceptible pine species offer congenial environment for survival, establishment and spread of the PWN and its insect vector in these countries territories. If PWN has been introduced in India, it may become a serious threat to pine and other susceptible conifer trees in future.
Other nematodes: Commonly occurring nematodes such as ectoparasitic nematodes are widely distributed in the pine forests as well as nurseries. Sohlenius (2002) studied the abundance and composition of the soil nematode fauna at three stages of a pine forest i.e., clear-cut, young stand and mature forest in central Sweden. At the sites without trees and with poor vegetation, nematode abundance was much lower than in the forested plots. At the clear-cut sites, the proportion of fungal feeders was quite low. The proportions of fungal feeding members of the Dorylaimida were highest in the 10-20 year-old pine stands.
Belonolaimus longicaudatus is a major nematode pest of pine trees. Ruehle (1969) found that lance (Hoplolaimus galeatus), pine cystoid (Meloidodera floridensis) and root-lesion (Pratylenchus brachyurus) reproduced on most of the pines but Meloidogyne arenaria, M. hapla, M. incognita, M. javanica and Pratylenchus zeae were not pathogenic to pines. A positive host-parasite relationship has been observed between pine and ring nematode (Criconemoides xenoplax), sheath nematode (Hemicycliophora vidua), spiral nematode (Helicotylenchus dihystera), stunt nematode (Tylenchorhynchus claytoni) and stubby-root nematode (Trichodorus christiei) evidenced by considerable increase in the population of these nematodes after 9 months (Ruehle, 1966, 1968). These nematodes also parasitize and reproduce on loblolly pine (Pinus taeda), sand pine (Pinus clausa), slash pine (Pinus elliottii var. elliottii) and shortleaf (Pinus echinata). Magnusson (2006) noted the presence of Tylenchus spp., Paratylenchus spp. and Malenchus tantalus in the root zone of Pinus sylvestris. Rotylenchus sp. fed on pine roots and to some extent reduced seedling growth but its pathogenic status on P. sylvestris was doubtful. Tylenchorhynchus claytoni is parasitic to a variety of pine species and is associated with unthriftiness, chlorosis and death of pine nursery seedlings (Ruehle, 1966). Pinus roxburghii is found to be susceptible to lance nematode, Hoplolaimus indicus (Anonymous, 2001).
In India, PWN is not reported to occur, but nearly 21 species of other nematodes belonging to 15 genera have been found to be associated with Pinus roxburghii and P. wallichiana. Sapna et al. (2009) has found Xiphinema americanum to be the predominant species followed by X. insigne, Paratylenchus similis, Hemicriconemoides mangiferae and Varotylus symmetricus. Tylenchus arcuatus and Malenchus nanellus were predominant species infesting pine nurseries.
Mangrove forests: A substantial proportion of the world's coastlines in the tropics are occupied by tidal mangrove forests which are believed to support estuarine and coastal Avicennia and Rhizophora trees; in the high intertidal zone, coarser sands usually dominate (Boto and Wellington, 1984). In subtropical Australian mangroves, Hodda and Nicholas (1986) found that nematodes were abundant (105 to 106 m-2) and that densities were highest in the low intertidal zone-a pattern similar to nematode communities in subtropical mangroves in North America (Hooper et al., 1973). However, in tropical mangrove estuaries along the Cape York peninsula of northeastern Australia, Alongi (1987) found that meiofaunal densities were lower (102 to 105 m-2) than in other mangrove habitats and decreased with tidal elevation. Mangrove ecosystem has been found to support numerous nematodes. A diversified nematode community was found in the study made at two small mangrove swamps and adjacent sandy beaches on Lizard Island, Great Barrier (Decraemer and Coomans, 1967). Numerous nematodes, importantly Araeolaimus, Adorus, Actinonema, Acantharchus, Acanthopharynx, Bathyeurystomina, Chromadora, Dorylaimus, Halalaimus, Microlaimus, Oncholaimellus, Quadricoma, Sphaerolaimus, Viscosia etc., have been reported to occur in mangrove and saltmarsh ecosystems in Australia (Alongi, 1987).
In India, Ali et al. (1983) made studies on the composition of nematodes in mangrove ecosystem of Pichavaram. Seventeen species of stylet-bearing nematodes have been described from mangrove in Sunderban, India (Sinha and Chaudhary, 1988). Among them important plant parasitic nematodes were Hirschmanniella gracilis, Helicotylenchus sp., Hemicriconemoides sunderbanensis, Tylenchus sp., Paralongidorus sp. etc. Sinha et al. (1987) described a new species of nematode from mangrove swamp of Sunderban. Nematodes are the dominant taxon in South Andaman mangrove sediment, India (Rao, 1986). They are the important consumers in the decomposer subsystem.
Nematode management in forest trees: A lot of work has been done on the management of nematodes infesting economically important crops with botanicals (Kimenju et al., 2008), microbial formulations (Mohiddin et al., 2010; Khan et al., 2012a), chemical (Qureshi et al., 2002) and cultural methods (Chirchir et al., 2011; Khan et al., 2012c). Besides, integration of different management approaches has proved most effective tool for sustainable nematode control in diversified climatic conditions (Haggag, 2002). Nematode management is difficult in forest trees. However, in forest nurseries it can be implemented easily and would be effective in a long run. Limited management options have been explored for nematodes other than pine wilt nematode infesting forest trees. Cram and Fraedrich (2005) suggested that crop rotation and fallow could be used to manage the plant parasitic nematodes in forest tree nurseries. To put these options into practice, it is important to know the host range and survivability of parasitic nematodes to be controlled. The needle nematode (Longidorus spp.) could be controlled with 1 year of fallow or crop rotations with a non-host. In fig plantation, adoption of good sanitation practices in nursery and elimination of source of nematode contamination may greatly reduce infestation level of H. fici. As far as chemical control is concerned, nematicides have not been used on figs due to low economic value of the fruit.
Use of biocontrol agents viz., Trichoderma spp. Paecilomyces lilacinus and Pochonia chlamydosporia may prove quite effective against nematode infestation (Khan, 2007). Application of phosphate solubilizing microbes may improve the productivity in addition to suppressing nematode infestation (Khan et al., 2009). Commercial formulations of these microorganisms are available which may be applied as seed/tuber dressing (Khan and Khan, 2011). Some studies have shown that Pasteuria penetrans and some nematophagous fungi could be used for biological control of fig cyst nematode. In addition to microbes, entomopathogenic nematodes may also suppress the population of plant parasitic nematodes. Soil application of Steinernema spp. has been found to decrease the population of root-knot nematodes (Khan et al., 2010, 2012b).
For the management of the timber nematode in pines and other conifer trees, the best strategy is to grow locally adapted pine species or the resistant pines (Dwinell, 1997). Removal of attacked trees may indirectly influence the resistance by affecting the degree of damage caused by Monochamus alternatus and the distribution pattern of pine species (Shi et al., 2009). The inoculation of pine trees with avirulent strains of the nematode may lead to the development of induced systemic resistance against the virulent strains of B. xylophilus. Injection of nematicides in tree trunk is a good method of minimizing the damage especially at small scale for example in parks, garden etc. (Kosaka et al., 2001).
Intensive quarantine, clear cutting and methyl bromide fumigation of pines killed by the pine wilt nematode are effective tools for PWN control. Application of fenitrothion (aerial and ground sprays) can reduce tree death rates by killing the beetle vectors. Other effective treatments to control the pine sawyer beetle, Monochamus alternatus are hot water treatment of the infested wood and high temperature treatment of affected logs and branches, submerging in water and burying in soil and direct trapping of adult beetles using terpene-baited traps (Dwinell, 1997). Heat processing by kiln drying the wood material at 56°C for 30 min can completely kill the vector insect and B. xylophilus (Duncan and Moens, 2006).
Chemical pesticides can provide effective control of nematodes in forest trees. Application of nematicides in combination with insecticides is another option for the management of pine wilt disease. The nematicides effective against Bursaphelenchus spp. include Disulfton, Fensulfothione and Thorazine. These nematicides are injected into the tree trunk to achieve effective management of the nematode for 2-3 years. Recently the efficacy of abamectin and emamectin benzoate against pine wilt disease has been established but their use is restricted due to shorter residual effects needing repeated application after every 2 years. Foliar spray of carbaryl on healthy trees could kill the weevil when it comes to feed in the leaf or bud axil.
Spray with morantal tartrate on tree trunks can manage the nematode but its application is not economic in terms of cost, thus restricted to nursery and ornamental trees. In Korea the infected trees are felled and cut into 1-2 m section and applied with metham sodium (11 m-3) followed by covering with thick vinyl sheet (0.1 mm). This method resulted to 100% mortality of the nematode and its vector. within 7 days. Zhao (1996) reported that a quinolizidine alkaloid (aloperine) derived from a shrub, Sophora alopecuroides possesses high toxicity against PWN (LC50 2.63x105 g). In addition, the entomophilic fungus, Beauveria bassiana and a parasitic wasp, Scleroderma guani could provide long lasting control of Monochamus spp. (Shimazu et al., 1995; Zhao, 2009). Recently, antibiotics such as Oxolinic Acid (OA) have been found to provide 71% control of the PWD in three-year-old seedlings of Pinus densiflora when injected @ 3 mg per seedling (Kwon et al., 2010). Application of oxolinic acid alone and a mixture of oxolinic acid and Abamectin controlled PWD on 20 year old pine trees under field conditions.
In view of high commercial value, some efforts have been made to manage nematodes in sandalwood cultivation in India. Application of Thimet @ 500 g per bed of 10x1 m in the seed bed and the potting mix may prove effective to reduce nematode damage. To avoid the combined infestation of fungus and nematode, seedbeds are sprayed with Dithane Z-78 (0.25%) once in 15 days and monthly application of 0.02% Ekalux solution (Rai, 1990). Soil drenching with a mixture of copper fungicides (blitox or Bordeaux mixture) and nematicides (quinalphos or phorate) along with reduced watering checked the damage by both the pathogens. Orwa et al. (2009) suggested spray of Dithane Z-78 (0.2%) after 15 days and application of 0.02% thimet solution after a month as effective strategy for the management of wilt fungus and nematode on Santalum album. Selection of healthy propagating material, use of clean seeds, removal and destruction of infected plants and soil amendment with manures, composts etc. may greatly help in the suppression of nematode populations in bamboo fields. Solarization of the bamboo field before bamboo plantation is also an effective nematode management tactics (Salch et al., 1988). Gantait (2010) reported that application of aldicarb, oxamyl, carbofuran and phorate proved highly effective against a wide range of nematodes and is safe for use in bamboo plantation.
The study has shown that plant nematodes pose a serious threat to the global forest wealth and the situation warrants adequate attention of plant pathologists, nematologists and entomologists. The available information on distribution of nematodes in forests is insufficient to assess the actual damage, but sufficient enough to require implementation of adequate management practices. The ectoparasitic nematodes which are universal component of soil microbial community pose greater threat to forest plantations because their debilitating effects are slow and unrecognizable leading to a slow, undetectable and gradual decline of trees. The ectoparasites are prevalent in all kinds of terrestrial as well as aquatic ecosystems (mangrove forests). These nematodes besides causing direct damage also facilitate the infection by several fungi, bacteria and viruses. In addition, some endoparasitic nematodes such as root-knot nematode, pine wilt nematode, flower bud and leaf gall nematodes cause specific and recognizable symptoms on a number of trees. Besides, there is immediate need for strict enforcement of quarantine measures on the import of wood and wood products from the PWN prone neighboring countries to prevent introduction of pine wilt nematode in the countries having climate suitable for the nematodes and its vector especially in Asia.
The financial assistance from the Aligarh Muslim University to meet processing and publication charges of this paper is thankfully acknowledged.