ABSTRACT
The marine invertebrates become one of hot spot for the lead of antimicrobial compounds. Two species of commercially available and edible bivalves (M. meretrix and M. casta) were assayed for antimicrobial activity against 10 bacterial pathogens and 6 fungal pathogens and its biochemical composition. The bivalves were extracted with three different solvent systems respectively methanol, ethanol and acetic acid. All the three extracts of both the species M. meretrix and M. casta showed highest antibacterial activities against S. aureus, E. coli, B. substillus, K. pneumonia, P. fleuroscence and V. cholera. In present investigation the methanolic extract of the two bivalve species of M. meretrix and M. casta was showed inhibition activities against all pathogenic fungal forms. The two bivalve extracts showed high amounts of protein content, which made the variation up to 160-180 μg mg-1 (wet weight). Both samples had low amount of carbohydrates 4.77-5.77 μg mg-1 and lipids 0.11-0.17 μg mg-1, respectively. The results of thin layer chromatography were revealed that presence of pink color spots it clearly indicates the presence of amino acid or peptides in bivalve’s samples. Presuming that the antimicrobial compounds were proteins or peptides. In SDS-PAGE on 12% gel, the crude proteins M. meretrix and M. casta showed 5-6 bands ranging from 45-223 kDa. They represent potential pharmacological leads perhaps possessing novel and uncharacterized mechanisms of action that might ultimately benefit the ongoing global search for clinically useful antimicrobial agents.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2013.38.43
URL: https://scialert.net/abstract/?doi=pjbs.2013.38.43
INTRODUCTION
The ocean have a great biodiversity of flora and fauna estimated to over 5,00,000 species, more than double of the terrestrial species (Kamboj, 1999). The richness of the diversity offers a vast potential of novel chemicals that may be useful for finding new bioactive compounds with greater efficiency and specificity for treatment of many dread full human diseases (Bergmann and Feeney, 1951; Faulkner, 2000a, b; Da Rocha et al., 2001). Among the marine phylum, the invertebrates are the most potential sources for the pharmaceutical industry. Because of the sessile (sponges, tunicates, soft corals, etc.) animals which have lack of physical defenses, produce toxic chemicals to protect themselves in a very hostile environment and the chemical defenses have been developing in the oceans for thousands of years and resulted in nature’s most toxic chemicals (Kathiresan et al., 2008).
The emergence of multiple-drug-resistant strains of microbial forms and the indiscriminate use of antibiotics highlight the urgent need for development of novel strategies to treat microbial infections. Marine organisms are the potential source for producing new innovative drugs. Recently, the marine invertebrates become one of hot spot for the lead of antimicrobial compounds. Bryostatin-1, a kind of macrocyclic lactone isolated from marine bryozoa Bugula neritina has already been in the secondary clinical trials and it found to be an excellent control over several types of cancers especially non-Hodgkin’s lymphoma, chronic lymphocytic leukemia and acute lymphocytic leukemia (Kraft et al., 1996; Mohammad et al., 1998).
The discovery of new classes of antibiotic is necessary due to the increased incidence of multiple resistances among the pathogenic microorganisms to drugs that are currently in clinical use (Burgess et al., 1999). Peptides from marine invertebrates are promising candidates as new antibacterial agents due to their broad antimicrobial spectrum, highly selective toxicities and their difficulty for bacteria to develop resistant (Jimeno and Hidalgo, 2006; Tincu and Taylor, 2004). In the present study, two bivalve species (M. meretrix and M. casta) was screened to evaluate antimicrobial activities, already high percentage of bioactivities has been reported in marine bivalves (Chatterji et al., 2002, Lixin et al., 2005; Sumita et al., 2009). Two species of commercially available, edible bivalves (M. meretrix and M. casta) were assayed for antimicrobial activity.
MATERIALS AND METHODS
Collection of specimens: The bivalves (M. meretrix and M. casta) were collected from Jan. 2009 to June 2010 in the mouth region of Vellar estuary and they were identified by using standard references (Satyamurthi, 1952) and also confirmed with zoological survey of India, Calcutta. The animals were not collected during the summer months to avoid stress related to diseases.
Preparation of bivalve extracts: The collected live animals were brought to the laboratory and the shells were broken and soft body, mantle fluid was removed. The extraction method was followed by Sumita et al. (2009). The collected materials were extracted in three different solvents such as ethanol, methanol and acetic acid (1:1). The extracts were homogenized at 15000 rpm for 15 min and the supernatant was filtered through No.2 filter paper. The filtrate was lyophilized to a powdered and used for antimicrobial studies.
Antimicrobial assay: The following bacterial and fungal pathogens were used for the antimicrobial assay: E. coli, Salmonella typhi, S. paratyphi, Enterococcus faecalis, P. fluresence, Staphylococcus aureus, S. pyogenes, Proteus vulgaris, Klebsiella pnuemoniae and Vibrio cholera) fungal pathogens are Aspergillus flavus, A. fumigatus, Candida albicans, Mucur sp., Penicillium sp. and Cryptococcus pneuoformis and these pathogenic forms were isolated from HIV+ve patients from Raja Muthiah Medical College, Annamalai University, Annamalai Nagar, Chidambaram. In vitro antimicrobial activity was determined by using the technique of Bauer et al. (1966).
Biochemical composition
Estimation of proteins: Proteins were estimated following the modified method of Bergmann and Feeney (1951) described by Peterson (1977). This method is applicable to soluble, membrane and conjugated proteins in dilute solutions, eliminating most interfering substances.
Estimation of carbohydrates: Carbohydrates were estimated following method of DuBois et al. (1956) was adapted.
Estimation of lipid: The chloroform-methanol extraction procedure of Folch et al. (1957) was used for extracting lipid from the soft body.
Thin layer chromatography and SDS-page: TLC profiling was done with both bivalve extracts with using two different solvent systems I and II. The solvent system I consist of methanol and chloroform (1:9) and solvent system II consist of combination of butanol, acetic acid and water (B:A:W) in the proportion of 5:1:4. The plates were showed pink color spots in both solvent systems when sprayed with ninhydrin solution. SDS-page was performed in 12% separating gels, according to the method of Laemmli (1970).
RESULTS
Antibacterial activities: The methanolic extract of bivalve M. meretrix showed maximum inhibition zone against S. aureus (10 mm), E. coli (10 mm) (Fig. 1) and minimum inhibition zone against K. pneumonia (2 mm). In ethanolic extract highest inhibition zone was observed in E. coli (12 mm), Likewise acetic acid extract of M. meretrix was showed maximum activities against E. coli (12 mm) and it showed minimum activities against P. vulgaris, K. pneumonia (3 mm).
In the crude methnolic extract of M. casta was showed highest inhibition of zone against S. typhi (12 mm). The P. fluorescence, B. substillus were showed highly resistant to crude methanolic extract. Ethanol extract of showed maximum zone of inhibition against B. substillus, K. pneumonia (12 mm). Acetic acid extract of M. casta was showed maximum activities against E. coli (13 mm), Salmonella typhi and S. paratyphi were showed resistant towards the acetic acid extract of M. casta (Fig. 2).
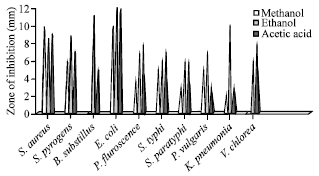 | |
| Fig. 1: | Antibacterial activities of M. meretrix |
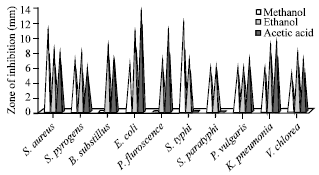 | |
| Fig. 2: | Antibacterial activities of M. casta |
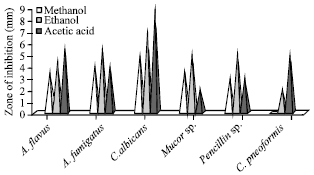 | |
| Fig. 3: | Antifungal activities of M. meretrix |
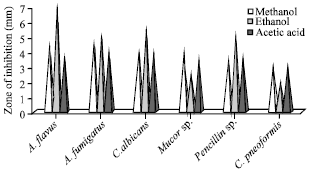 | |
| Fig. 4: | Antifungal activities of M. casta |
| Table 1: | Biochemical composition of marine bivalves |
 | |
Antifungal activities: The methanolic extract of M. meretrix was showed maximum zone of inhibition against C. albicans (5 mm). C. pneoformis was highly resistant to the methanolic extract of M. meretrix. In ethanolic extract highest zone of inhibition was observed against C. albicans (7 mm) and the lowest activity against Penicillium sp. (2 mm). The acetic acid extract of M. meretrix was revealed that maximum zone of inhibition against C. albicans (9 mm) and it minimum activity against Mucor sp. (Fig. 3).
In crude methanolic extract of M. casta maximum zone of inhibition was observed against A. flavus, A. fumigatus (4.5 mm). The crude ethanolic extract of M. casta was showed highest zone of inhibition against A. flavus (5 mm) and it minimum activity against Mucor sp. (2.5 mm). The crude acetic acid extract of M. casta was showed maximum zone of clearance in A. fumigates, C. albicans (Fig. 4).
Biochemical composition: The results of bivalve species (protein, carbohydrates and lipid) were given in the Table 1. The bivalve M. meretrix were exhibited 165.4 μg mg-1 protein, 4.77 μg mg-1 carbohydrates and 0.11 μg mg-1 lipid. The M. casta was showed 180 μg mg-1.
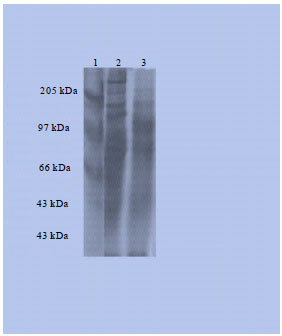 | |
| Fig. 5: | SDS-page showed molecular weight of crude proteins, Lane 1: Standard markers, Lane 2: Crude extract of M. meretrix, Lane 3: Crude extract of M. casta |
Thin layer chromatography (TLC): TLC profiling was done with both bivalve extracts with using two different solvent systems I and II. The solvent system I consist of methanol and chloroform (1:9) and solvent system II consist of combination of butanol, acetic acid and water (B:A:W) in the proportion of 5:1:4. The plates were showed pink color spots in both solvent systems when sprayed with ninhydrin solution. The pink color spot in TLC plate was clearly indicating the presence of amino acids and peptides.
SDS-PAGE: The crude extracts of both bivalve species were showed antimicrobial activities was subjected to SDS-PAGE for the estimation of the molecular weight of proteins present in it. For the estimation of molecular weights on crude extract of bivalve species protein using different standards such as, α-Macroglobulin (170 KDa), β-Galactosidase (116 KDa), Fructose-6-phosphate kinase (85.2 KDa), Glutamate dehydrogenase (55.6 KDa), Aldotase (39.2 KDa), Trios phosphate isomerase (26.6 KDa) and Trypsin inhibitor (20.1 KDa). In this present context, it revealed that both bivalve species have similar molecular weight proteins. Crude protein of M. meretrix yielded 6 bands ranging from 45 to 261 KDa with well defined bands at 45, 104, 149, 185, 223 and 261 KDa and in M. casta was showed 5 bands ranging from 50-218 KDa with well defined bands at 50, 86, 98, 125 and 218 (Fig. 5).
DISCUSSION
Bivalves possess several types of defense molecules including agglutinins and glycoproteins that have bactericidal activities (Johnshon, 1964; Arimoto and Tripp, 1977; Mori et al., 1980; Renwrantz and Stahmerge, 1983; Renwrantz et al., 1998). Study of modern medicine and pharmacology also showed that the flesh of M. meretrix has showed effects of antimutation (Yu et al., 1994), antitumor (Don et al., 1999) and immune promotion (Luo et al., 1996) activities. Several cystines rich AMPs were isolated from M. galloprovincialis, M. edulis (Mitta et al., 1999a; Charlet et al., 1996). The flesh of M. meretrix was used widely in India and China as a fisher folk medicine to treat the several liver diseases like Jaundice, Hepatitis-A and B (Wang et al., 2006). Likewise, the steroid extract of M. meretrix was inhibiting the cell growth and induction of G1-phase cell cycle arrest in hepatoma cells (Wu et al., 2006). Sumita et al. (2009) reported the antibacterial activities from edible bivalves Perna viridis and M. casta. The molluscan outer shells were found to have the ability to inhibit growth of pathogenic bacteria E. coli, Staphylococcus aureus and Salmonella enteridis, which cause food borne illness (Takama et al., 1999).
All the three extracts of both the species M. meretrix and M. casta showed highest antibacterial activities against S. aureus, E. coli, B. substillus, K. pneumonia, P. fleuroscence and V. cholera. The similar work made on other species of mollusc support of other workers, antibacterial activities of green mussel (P. viridis) and edible oyster (Crassostrea madrasensis) (Annamalai et al., 2007). Rajaganapathi (2001) reported the antimicrobial activities of marine mollusc and purification of anti-HIV protein. Henry et al. (1991) reported hemocytes of Pinna nobilis exhibited the antibacterial activity. Lectin isolated from heamolyph of the horse muscle Modiolus modiolus exhibited strong antibacterial activities against V. anguillaram, V. salmonicida, V. viscosus, V. wondanis and V. ordalii (Tunkijjanukij and Olafsen, 1998). Anand et al. (1997) reported that hypobranchial glands of Chicoreous virgineous and egg capsules of Rapana rapiformis were showed wide and spectral antibacterial activities against dreadful human pathogens. In present investigation the methanolic extract of the two bivalve species of M. meretrix and M. casta was showed inhibition activities against all pathogenic fungal forms. This finds supported by Rajaganapathi (2001) reported that methanolic extract of A. rhombea inhibit the growth of Mucor sp., Penicillium sp. and A. flavus. Charlet et al. (1996) reported that a peptide (Containing 12 cystines, about 6.2 kDa molecular weight) isolated from the blood of mussel (M. edulis) was showed antifungal activity. In the crude methanolic extract of Cypraea errones exhibited the antibacterial and antifungal activity against S. aureus, S. pyogenes, A. niger and C. albicans (Anand and Edward, 2002). Sumita et al. (2009) reported the antifungal activities two edible bivalve species P. viridis and M. casta. Gill tissue of the mussel of M. edulis: A new source of antimicrobial peptides. The saline extract of M. meretrix and M. casta was showed maximum zone of clearance against A. fumigatus, A. flavus and C. albicans. It was evidence by Jang et al. (2006) reported the antifungal activity of synthetic peptide derived from halocidin, from tunicate Halocynthia aurantium which showed the inhibition of C. albicans.
The two bivalve extracts showed high amount of protein content, which made the variation up 165 to 180 μg mg-1. Both samples had low amount of carbohydrates 4.77-5.77 μg mg-1 and lipids 0.11 to 0.17 μg mg-1, respectively. These values are similar to those reports for mantle and tissues of M. casta, which contained 190 μg mg mL-1 protein, 5.76 μg mg mL-1 carbohydrates and 0.15 μg mg mL-1 lipid respectively, (Sumita et al., 2009). Gopalakrishnan and Vijayavel (2009) reported nutritional composition of three estuarine bivalve’s P. viridis, Donax caneatus and M. meretrix. Jayabal and Kalyani (1986) reported biochemical composition of M .meretrix from Bay of Bengal. Generally, bivalves are rich in nutrients, particularly proteins, fats and minerals. The Indian edible bivalves have protein (5-14%), fats (0.5-3%), calcium (0.04-1.84%) and phosphorus (0.1-0.2%) and iron (1-29 mg/100 g of the fresh weight).
The results of thin layer chromatography were revealed that presence of pink color spots it clearly indicating presence of amino acid or peptides in bivalve’s samples. Presuming that the antimicrobial compounds was proteins or peptides. There are several reports are from the antimicrobial peptides has been reported from the heamocytes (Charlet et al., 1996; Mitta et al., 1999a, b), epithelial tissues (Marshall and Arenas, 2003; Iijima et al., 2003) and tissues gut and respiratory organs (Tincu and Taylor, 2004) of bivalve molluscs.
In SDS-PAGE on 12% gel, the crude proteins M. meretrix and M. casta showed 5-6 bands ranging from 45-223 KDa. Sumita et al. (2009) observed that unclear bands ranging from 14 and 29 KDa in marine bivalves M. casta and P. viridis. This Similar result was from Perna canaliculus containing the protein with molecular weight of 35 KDa (Scotti et al., 2001). Likewise. in M. edulis are containing histidine-rich glycoprotein (Nair and Robinson, 1999; Renwrantz sand Stahmerge, 1983). Chandren et al. (2009) observed 9.7 kDa proteins in gill of estuarine bivalve P. viridis.
CONCLUSION
The present investigation concluded that M. meretrix and M. casta contain several factors with antimicrobial activities, in various organs and tissues. Thin layer chromatography results were revealed that proteinaceous compounds are responsible for antimicrobial activities. They represent potential pharmacological leads perhaps possessing novel and uncharacterized mechanisms of action that might ultimately benefit the ongoing global search for clinically useful antimicrobial agents.
REFERENCES
- Anand, T.P., J. Rajaganapathi and J.K. Patterson Edward, 1997. Antibacterial activity of marine mollusc from Portonovo region. Indian J. Mar. Sci., 26: 206-208.
Direct Link - Anand, P.T. and J.K.P. Edward, 2002. Antimicrobial activity in the tissue extracts of five species of cowries Cyprea sp. (Mollusca: Gastropoda) and an ascidian Didemnum psammathodes (Tunicata: Didemnidae). Indian J. Mar. Sci., 31: 239-242.
Direct Link - Annamalai, N., R. Anburaj, S. Jayalakshmi and R. Thavasi, 2007. Antibacterial activities of green mussel (Perna viridis) and edible oyster (Crassostrea madrasensis). Res. J. Microbiol., 2: 978-982.
CrossRefDirect Link - Arimoto, R. and M.R. Tripp, 1977. Characterization of a bacterial agglutinin in the hemolymph of the hard clam, Mercenaria mercenaria. J. Invertebrate Pathol., 30: 406-413.
CrossRef - Bauer, A.W., W.M.M. Kirby, J.C. Sherris and M. Turck, 1966. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol., 45: 493-496.
CrossRefPubMedDirect Link - Bergmann, W. and R. Feeney, 1951. Contributions to the study of marine products. XXXII. The nucleosides of sponges. I. J. Org. Chem., 16: 981-987.
CrossRefDirect Link - Burgess, J.G., E.M. Jordan, M. Bregu, A. Mearns-Spragg and K.G. Boyd, 1999. Microbial antagonism: A neglected avenue of natural products research. J. Biotechnol., 70: 27-32.
CrossRef - Chandran, B., G. Rameshkumar and S. Ravichandran, 2009. Antimicrobial activity from the gill extraction of Perna viridis (Linnaeus, 1758). Global J. Biotechnol. Biochem., 4: 88-92.
Direct Link - Charlet, M., S. Chernysh, H. Philippe, C. Hetru, J.A. Hoffmann and P. Bulet, 1996. Innate immunity: Isolation of several cysteine-rich antimicrobial peptides from the blood of a mollusc, Mytilus edulis. J. Biol. Chem., 271: 21808-21813.
PubMed - Chatterji, A., Z.A. Ansari, B.S. Ingole, M.A. Bichurina, M. Sovetova and Y.A. Boikov, 2002. Indian marine bivalves: Potential source of antiviral drugs. Curr. Sci., 82: 1279-1282.
Direct Link - Da Rocha, A.B., R.M. Lopes and G. Schwartsmann, 2001. Natural products in anticancer therapy. Curr. Opin. Pharmacol., 1: 364-369.
CrossRef - Don, C., F. Huang and L. Huang, 1999. An experimental study on the effects of meretrix polysaccharide on antineoplastic and immunorgulation. Chin. Mar. Drug, 2: 15-15.
Direct Link - DuBois, M., K.A. Gilles, J.K. Hamilton, P.A. Rebers and F. Smith, 1956. Colorimetric method for determination of sugars and related substances. Anal. Chem., 28: 350-356.
CrossRefDirect Link - Faulkner, D.J., 2000. Highlights of marine natural products chemistry (1972-1999). Nat. Prod. Rep., 17: 1-6.
PubMedDirect Link - Folch, J., M. Lees and G.H.S. Stanley, 1957. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem., 226: 497-509.
CrossRefPubMedDirect Link - Gopalakrishnan, S. and K. Vijayavel, 2009. Nutritional composition of three estuarine bivalve mussels, Perna viridis, Donax cuneatus and Meretrix meretrix. Int. J. Food Sci. Nutr., 60: 458-463.
CrossRef - Jang, W.S., H.K. Kim, K.Y. Lee, S.A. Kim, Y.S. Han and I.H. Lee, 2006. Antifungal activity of synthetic peptides derived from Halocidin, antimicrobial peptide from the tunicate, Halocynthia aurantium. FEBS Lett., 580: 1490-1496.
CrossRefDirect Link - Jimeno, A. and M. Hidalgo, 2006. Pharmacogenomics of Epidermal Growth Factor Receptor (EGFR) tyrosine kinase inhibitors. Biochim. Biophys. Acta (BBA)-Rev. Cancer, 1766: 217-229.
CrossRef - Johnshon, H.M., 1964. Human blood group A1 specific agglutinin of the butter clam Saxidomus giganteus. Science, 146: 548-549.
CrossRef - Kraft, A.S., S. Woodley, G.R. Pettit, F. Gao, J.C. Coll and F. Wagner, 1996. Comparison of the antitumor activity of bryostatins 1, 5 and 8. Cancer Chemother. Pharmacol., 37: 271-278.
CrossRef - Laemmli, U.K., 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227: 680-685.
CrossRefDirect Link - Lixin, Z., F. Xiao and H. Lijun, 2005. Antitumor and immune regulation activities of the extracts of some Chinese marine invertebrates. Chin. J. Oceanol. Limnol., 23: 110-117.
CrossRef - Luo, Y., D. Wu and D. Ni, 1996. Pharmacologic effects of flesh of the clam Meretrix meretrix L. Chin. Mar. Drug, 15: 14-17.
Direct Link - Marshall, S.H. and G. Arenas, 2003. Antimicrobial peptides: A natural alternative to chemical antibiotics and a potential for applied biotechnology. Electron. J. Biotechnol., 6: 262-275.
Direct Link - Mitta, G., F. Vandenbulcke, F. Hubert and P. Roch, 1999. Mussel defensins are synthesised and processed in granulocytes then released into the plasma after bacterial challenge. J. Cell Sci., 112: 4233-4242.
Direct Link - Mohammad, R.M., M.L. Varterasean, V.P. Almatchy, G.N. Hannoudi, G.R. Pettit and A. Al-Katib, 1998. Successful treatment of human chronic lymphocytic leukemia xenografts with combination biological agents auristatin PE and bryostatin 1. Clin. Cancer Res., 4: 1337-1343.
Direct Link - Nair, P.S and W.E. Robinson, 1999. Purification and characterization of the histidine-rich glycoprotein that binds cadmium from the blood plasma of the bivalve Mytilus edulis. Arch. Biochem. Biophys., 366: 8-14.
CrossRef - Iijima, N., N. Tanimoto, Y. Emoto, Y. Morita, K. Uematsu, T. Murakami and T. Nakai, 2003. Purification and characterization of three isoforms of chrysophsin, a novel antimicrobial peptide in the gills of the red sea bream, Chrysophrys major. Eur. J. Biochem., 270: 675-686.
Direct Link - Peterson, G.L., 1977. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal. Biochem., 83: 346-356.
CrossRefPubMedDirect Link - Renwrantz, L., W. Schmalmack and M. Steenbuck, 1998. Molecular size of native proteins of Mytilus serum which contains a dominant fraction with heavy metal-binding properties. Comp. Biochem. Physiol. Part A: Mol. Integr. Physiol., 121: 175-180.
CrossRef - Scotti, P.D., S.C. Dearing, D.R. Greenwood and R.D. Newcomb, 2001. Pernin: A novel, self-aggregating haemolymph protein from the New Zealand green-lipped mussel, Perna canaliculus (Bivalvia: Mytilidae). Comp. Biochem. Physiol. Part B: Biochem. Mol. Biol., 128: 767-779.
CrossRef - Sumita, S., A. Chatterji and P. Das, 2009. Effect of different extraction procedures on antimicrobial activity of marine bivalves: A comparison. Pertanika J. Trop. Agric. Sci., 32: 77-83.
Direct Link - Tincu, J.A. and S.W. Taylor, 2004. Antimicrobial peptides from marine invertebrate. Antimicrob. Agents Chemother., 48: 3645-3654.
CrossRef - Wu, T.H., R.L. Yang, L.P. Xie, H.Z. Wang and L. Chen et al., 2006. Inhibition of cell growth and induction of G1-phase cell cycle arrest in hepatoma cells by steroid extract from Meretrix meretrix. Cancer Lett., 23: 199-205.
CrossRef - Tunkijjanukij, S. and J.A. Olafsen, 1998. Sialic acid-binding lectin with antibacterial activity from the horse mussel: Further characterization and immunolocalization. Dev. Comp. Immunol., 22: 139-150.
CrossRefPubMedDirect Link - Wang, G., B. Liu, B. Tang, T. Zhang and J. Xiang, 2006. Pharmacological and immunocytochemical investigation of the role of catecholamines on larval metamorphosis by β-adrenergic-like receptor in the bivalve Meretrix meretrix. Aquaculture, 258: 611-618.
CrossRef








