Research Article
Investigation on the in vitro Rearing of Trissolcus grandis an Egg Parasitoid of Eurygaster integriceps by Use of Artificial Diet
Biological Control Research Department, Plant Pests and Diseases Research Institute, Tehran, Iran
Progress in entomological research and the success of pest management programs depends on our ability to rear insects and establish colonies in the laboratory (Singh and Moore, 1985). In fact, in an in vivo production system for a parasitoid such as Trichogramma, more than 70% of the total resources are devoted to host production (Newton, 1993). Variety of beneficial arthropods have been reared and used successfully in traditional way but it has been very tedious and difficult (Wagge et al., 1985). After decades of intensive research and dozens of published reports on such subject, yet there are no commercial mass rearing programs for entomophages based on artificial rearing (Cohen et al., 1999). However, in vitro rearing of beneficial insects has been intensively investigated in recent years but success has been limited (Grenier, 1994).
In vitro rearing of insects started with efforts of Bogdanow (1908) in mass culture of blowfly, Calliphora vomitoria L. (Diptera: Calliphoridae) on a diet consisted of peptone, meat extract, starch and minerals (Singh and Moore, 1985). The great success of factory scale production of screwworm in 1936 was a turning point in mass culture of insects (Bush, 1978). The development of artificial diets for entomophages would greatly improve the potential for biological control of arthropods (Cohen et al., 1999; Singh and Moore, 1985). Efforts of House (1972) to define a diet for Agria housei (Shewell) (Diptera: Sarcophagidae) provided a precious knowledge based on which the research on in vitro rearing of other parasitoids was established (Thompson, 1986, 1999). An exact chronological review on the published materials and species reared in vitro has been given by Thompson and Hagen (1999). Cohen et al. (1999) in a review article have discussed the potential and problems associated with in vitro rearing of beneficial insects.
Regarding egg parasitoids, Hoffmann et al. (1975) reared Trichogramma pretiosum Riley (Hymenoptera: Trichogrammatidae) form egg to adult on insect hemolymph for the first time. The results of Chinese scientists on in vitro rearing of a number of parasitoids have been complied by Li (1993). A considerable effort was also made to rear Trichogramma sp. in vitro by Li (1990, 1992), Li et al. (1988), Liu et al. 1979; 1983), Xie et al. (1986a, b), Grenier et al. (1995), Consoli and Parra (1996, 1999), Nordlund et al. (1997), Dahlan (1999), Heslin et al. (2005a, b). There have also been attempts to rear other egg parasitoids such as Anastatus japonicus Ashmead (Hymenoptera: Eupelmidae) (Li et al., 1988, Li, 1990, 1992; Xie et al., 1991), Telenomus heliothidis Ashmead (Hymenoptera: Scelionidae) (Strand et al., 1988) and Trissolcus basalis (Wollaston) (Hymenoptera: Scelionidae) (Volkoff et al., 1992).
Most of artificial diets used have been consisted of insect hemolymph, chicken egg yolk, minerals, yeast, amino acids and other nutritional materials. Recent studies have been focused on using diets free of insect derivatives and quality control aspects. Grenier et al. (1995) compared diets contained insect derivatives with those without them. Parasitoids (Trichogramma) reared on diets lacking insect materials were inferior regarding their biological traits compared with those reared on diets containing insect hemolymph or host eggs. Consoli and Parra (1996) reported that diets contained larval hemolymph of Helicoverpa zea Boddie (Lepidoptera: Noctuidae) supported the best growth and development for T. galloi Zucchi and T. pretiosum Riley. Nordlund et al. (1997) reared T. minutum Riley for 10 generations on artificial diet containing yeast extract, amino acids, nonfat dry milk, chicken egg yolk, chicken embryo extract and Manduca sexta (L.) (Lepidoptera: Sphingidae) egg extract. Developmental rate and percentage of deformed female were significantly more in progenies of wasps reared in vitro compared with those reared on H. zea Boddie eggs. Dahlan (1999) reared T. australicum Girault on an artificial diet for the first time and compared its development and reproductive compatibility with those reared on Helicoverpa armigera (Hübner) (Lepidoptera: Nuctoidae) eggs. Moreover, studies on commercializing of egg parasitoids by in vitro rearing have been documented. Consoli and Parra (1999) used plastic membranes of different thickness to manufacture artificial eggs for in vitro production of T. galloi Zucchi and T. pretiosum Riley. High density polyethylene 7-8 and 9-10 μm thickness resulted in the best acceptance and parasitization by parasitoids.
As T. grandis is one of the most important parasitoids of Sunn pest eggs in Iran (Amirmoafi, 2000), augmentative release of this parasitoid relies on simple and low cost techniques for its mass production. So far, mass production of this parasitoid has been limited to the production of a few Heteropteran hosts such as Eurygaster integriceps Puton (Heteroptera: Scutelleridae) or Graphosoma lineatum (L.) (Heteroptera: Pentatomidae) (Asgari, 1995; Shirazi, 1996; Shahrokhi, 1997). The very close work to the present research is a study conducted by Volkoff et al. (1992) who reared T. basalis on artificial diet up to 3rd larval instar, which is considered a giant step in rearing oligophages parasitoids in vitro. This challenge was taken up to evaluate the possibility of using some artificial diets based on previous studies to rear T. grandis.
Eurygaster integriceps rearing: The culture of Sunn pest was established in lab by collecting population from overwintering sites in winter or from wheat fields in spring during 1999-2000. The collected adults were kept in metal screened cages (0.5x0.5x0.5 m) at 25±1°C, 60±5% RH and 16:8 h L:D. Wheat grains were used loosely or pasted by wetted wheat flour on a piece of paper (10x20 cm) to feed the adults. A glass tube filled with distilled water and sealed with cotton was used as water supplement in each cage. Strips of white A4 papers (1x30 cm) were hanged from the ceiling of cages as oviposition sites. Eggs were collected daily by cutting paper strips and used in experiments or kept at 5±1°C, 65±10% RH and total darkness for later use. The method of Sunn pest rearing was taken after Remaudiere (1961).
T. grandis culture: The stock of T. grandis was taken from a mass production colony reared in Biological Control Research Dept. (BCRD), Plant Pests and Disease Research Institute (PPDRI). The parasitoid was cultured on Sunn pest eggs in glass tubes (2.5x20 cm) at 25±1°C, 60±10% RH and 16:8 h L:D.
Hemolymph sources: Hemolymph was obtained from 3 insects: a) Last instar larvae of Chilo suppressalis (Walker) (Lepidoptera: Pyralidae), B) Pupal stages of Pseudaletia unipuncta (Haworth) (Lepidoptera: Noctuidae) and C) Pupal stages of Antheraea pernyi (Guénerin-Méneville) (Lepidoptera: Saturnidae). To extract hemolymph from larval stage, 100 live larvae were selected for each experiment and kept for 6-8 min in 60°C water. After surface sterilizing with ethanol 96%, legs and head capsules were removed and they were crashed in a china bowl. The mixture was then centrifuged at 20000 RPM for 30 min. The supernatant was filtered by filter paper size 0.22 m (Milipore®) and used. Pupae were treated at 60°C water for 10 min and after surface sterilizing with ethanol 96%, the hemolymph was extracted with hand by incising pupal head. The rest of procedure is the same as mentioned for larval hemolymph.
Salt solution: Salt solution was based on Nettles et al. (1983) and consisted of 1.55 g KCl and 1.50 g MgSO4, 7H2O, solved in 250 ml of distilled water and autoclaved before usage.
Chicken egg yolk: Eggs were surfaced sterilized by ethanol 96% for 5 min. Then, yolk was taken out under a flow laminar hood and directly used under sterilized conditions.
Host egg contents: In each experiment, 1000 eggs of Eurygaster integriceps were surface sterilized by ethanol 96% for 5 min and then rinsed by distilled water. They were air dried under a laminar flow chamber and crashed in a china bowl. After filtering out chorion bits the remaining liquid was used.
Milk: Ten grams of commercial powder milk (Maria Jimg®, China) in 100 ml of distilled water was used after 10 min boiling under sterilized conditions.
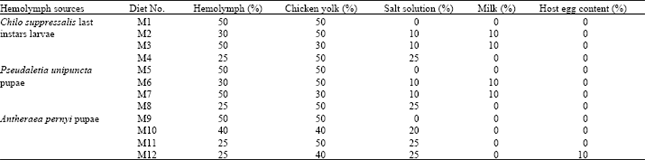
| Table 1: | Different artificial media tested for in vitro rearing of Trissolcus grandis (Thompson) (Hymenoptera: Scelionidae) |
 | |
Rearing containers: Rearing containers were serological plates with 9-12 concave cavities (ø=18 mm and 1.5-1.75 mm deep).
Diet preparation: After mixing the proper amount of elements for each diet, 15 μL of that was transferred into each hole of the rearing container by micropipette (Eppendrof®, 10-100 μL adjustable). Then, an egg or neonate larvae of T. grandis was placed into the liquid diet by dissecting E. integriceps 4 h (for eggs) or 18-20 h (for larvae) parasitized eggs through a dissecting microscope under a laminar flow hood. Parasitoid immature stages were taken out from host eggs by a fine paint brush and host egg contents around them was removed quickly before transferring into artificial diet. After completing a rearing container (9-12 holes), it was covered with a same size glass plate and were kept tight together by 4 metal clips. Each 5 rearing containers were kept into a desiccator (~100% RH) inside an incubator at 26±1°C, 60±10% RH and 16:8 h L:D. For each diet 150 cells (each 50 cells considered as 1 replication) were tested. After 3, 24, 48, 72 and 168 h rearing containers were checked under dissecting microscope and number of dead or alive larvae, the developmental status and larval stage were recorded. Table 1 shows the kind of material and their ratio in each diet used during this study.
Statistical methods: The data was subjected to square root of X+0.5 before analysis. First, the transferred data was analyzed by PROC GLM (SAS Institute, 2002) considering a split plot design in which diets were as main plots and time intervals as subplots. The same procedure was employed for hemolymph sources as main plots. As the interaction of TreatmentsxTime was strongly significant, for each treatment (diets or hemolymph sources) a separate analysis of variance was considered over each time interval. However, the data was pooled together to find out the best treatments for all times. To make sure of correct level of significance among treatments, PROC GLM and PROC MIXED (SAS Institute, 2002) was applied in multivariate approach and the G-G and F-H adjusted p>F was calculated (Littell et al., 1998) for the diets and hemolymph as source of variation over times, separately.
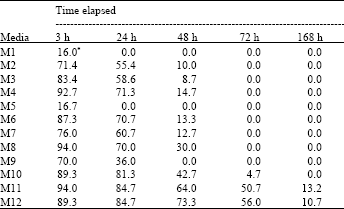
| Table 2: | Mean percent of Trissolcus grandis (Thompson) (Hymenoptera: Scelionidae) larval alive on 12 artificial diets after 3, 24, 48, 72 and 168 h |
 | |
| *Means of 3 replicates | |
Means were compared and separated by Duncan’s Multiple Range Test
RESULTS
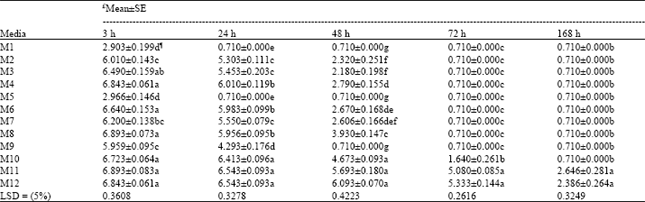
As parasitoid eggs did not hatch on any of tested diets, the experiments continued only by rearing neonate larvae. There were strong significant differences among diets based on analysis of data (F11, 24= 136.81, 342.7, 163.27, 375.1 and 40.15 at 3, 24, 48, 72 and 168 h, respectively, all with p≤0.0001 for both adjusted G-G and H-F p>F). Table 2 shows the percentage of parasitoid larvae alive in each diet with respect to the time. Comparison among M1, M2, M3 and M4 in which C. suppressalis larval hemolymph was used, revealed that in M1 after 3 h all larvae died (Table 2). By reducing hemolymph concentration and adding salt solution and milk in M2 the larvae grew up significantly better compared with M1 (Table 2 and 3). Again in M3 hemolymph was kept by 50% and yolk was reduced up to 30%, with the same rate of salt solution and milk. Here, at 3 h, M3 had significantly higher survived larvae compared with M2 but at 24 and 48 h both media had the same results (Table 2 and 3).
| Table 3: | Mean±SE comparisons of data (alive larvae) on in vitro rearing of Trissolcus grandis (Thompson) (Hymenoptera: Scelionidae) larvae on 12 artificial diets after 3, 24, 48, 72 and 168 h |
 | |
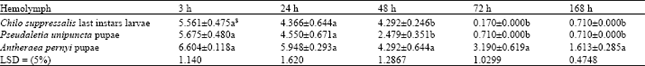
| Table 4: | Mean±SE comparisons of data (alive larvae) on in vitro rearing of Trissolcus grandis (Thompson) (Hymenoptera: Scelionidae) larvae on artificial diets containing 3 different kinds of hemolymph after 3, 24, 48, 72 and 168 h |
 | |
| Table 5: | Mean±SE comparisons of pooled data for 12 diets and 3 kinds of hemolymph treatments obtained from data on in vitro rearing of Trissolcus grandis (Thompson) (Hymenoptera: Scelionidae) alive larvae for total of 168 h |
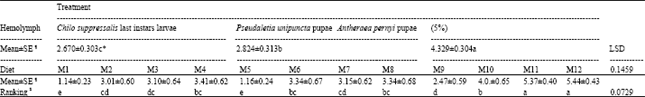 | |
| ¶ Mean of data after conversion by square root of x+0.5, * Means followed by same letter in a column are not different significantly, $Based on Duncan’s Multiple Range Test | |
In M4 that the hemolymph was reduced to 25% (yolk and salt solution were kept at 50 and 25%, respectively), the result was not significantly different at 3 h compared with M3 but significantly better than M1 and M2 at this time and all the media in this group at 24 and 48 h (Table 3). In M5, M6, M7 and M8 in which the pupal hemolymph of P. unipuncta was used, with the same concentration of ingredients of M1-M4, the trend of larval survivorship was somehow the same at 3 h but at 24 and 48 h diets in this group significantly preformed better (Table 3). Among these diets (M5-M8), M6 and M8 supported larval growth uniformly up to 24 h and were significantly superior to M5 and M7, but at 48 h, M8 was significantly better than M5, M6 and M7. In the diets containing A. pernyi pupal hemolymph (M9-M12) the results were outstanding. In M9 with the basic materials (50% hemolymph and 50% yolk) the number of larvae survived at 3 h was significantly higher compared with M1 and M5 and it supported the growth up to 24 h. However, M9 had the lowest larvae alive at 3 and 24 h compared with other diets in its group (M10-M12, Table 3). In M10 with 40, 40 and 20% of hemolymph, yolk and salt solution, respectively, the larval growth went on beyond 48 h and reached at 72 h. Media 11 contained 25% hemolymph 50% chicken egg yolk, 25% salt solution supported larval growth up to 50.7 and 13.2% at 72 and 168 h, respectively. Adding 10% of host egg contents in diet M12 by reducing equal amount of yolk did not change the results significantly compared with M11. Comparing the means of data based on types of hemolymph (Table 4) showed that up to 24 h all types of hemolymph did not differ significantly with respect to larval development (F2, 33= 2.08 and 2.35, with p≤0.1407 and p≤0.1107 at 3 and 24 h, respectively). But 48 h after larval growth (F2, 33= 7.31, p≤0.0024 for both adjusted G-G and H-F p>F) diets containing A. pernyi pupal hemolymph (4.329±0.304) were significantly superior to diets containing either larval hemolymph of C. suppressalis or P. unipuncta pupae. Moreover, diets in the 2 former groups only gave rise to 48 h of larval growth; however, P. unipuncta hemolymph was also significantly higher in terms of T. grandis alive larvae at given time intervals compared with diets containing C. suppressalis hemolymph (Table 2-4). The overall results shown in Table 5 revealed that the hemolymph of A. pernyi was significantly better compared with C. suppressalis or P. unipuncta hemolymph and diets M11 and M12 with 5.37±0.40 and 5.44±0.43 average number of larval survived for 168 h developmental time, respectively, were the same but significantly superior to all other diets and M1 with a mean of 1.14±0.23 was significantly inferior compared to all other diets (Table 5). No pupa was observed in vitro. Not statistically analyzed data showed that larval development on artificial diet was longer than that on natural host eggs (72 h vs. 48 h in the first instar larvae and 96 h vs. 72 h in the second instar larvae, respectively on artificial diets and host eggs).
Among the diets tested, the best result was obtained when the hemolymph of A. pernyi was combined with chicken egg yolk and salt solution. Hemolymph is assumed as oviposition stimulant as well as supporting growth and development and it is quite essential for pupal formation and adult eclosion (Liu et al., 1979, 1983). The kind of hemolymph has proved to be significantly effective on results of in vitro rearing and pupal stage hemolymph of large butterflies has been superior to other stages and insects (Consoli and Parra, 1996). In this study from uniformity of parasitoid larval growth point of view, hemolymph of A. pernyi pupae> P. unipuncta pupae> and C. suppressalis larvae were better, respectively. However, on the best diets (M11 and M12) larval mortality reached 100% after 168 h (Table 2 and 3). Consoli and Parra (1996) observed 100% larval mortality for T. pretiosum Riley before reaching 3rd instar larvae on diet with Diatraea saccharalis (Fabricius) (Lepidoptera: Pyralidae) and Bombyx mori (L.) (Lepidoptera: Bombycidae) pupal hemolymph. They could only rear T. galloi well-formed on diets made of H. zea larval hemolymph. Grenier et al. (1995) proved that generation time (egg to adult) was longer on media devoid of hemolymph. They also found lower rate of pupation and adult emergence on such diets which is at par with part of the findings of this study. Carpenter et al. (2000) reported that pupal hemolymph of Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) gave rise to significantly better results in artificial rearing of Diapetimorpha introita (Cresson) (Hymenoptera: Ichneumonidae) compared with diet devoid hemolymph. However, there have been efforts to rear parasitoids on diets without insect materials (Wu et al., 1982; Guerra et al., 1993; Grenier et al., 1995; Heslin et al., 2005a, b).
The ratio of 50% chicken egg yolk along with alteration of other main components such as hemolymph and salt solution has given the best results comparing M4, M8 and M11 by other diets in their related group and the least mortality of larvae happened with this ratio of chicken egg yolk (Table 3). Chicken egg yolk which contains lipids, proteins and minerals is considered as embryo growth promoter and quickens early larval growth (Volkoff et al., 1992; Thompson, 1999). Egg yolk used as the only source of lipids in defined artificial media for T. brassicae was necessary to obtain high percentage of larvae and adults (Wu et al., 1982; Li et al., 1988). For some pupal parasitoids such as D. introita the amount of lipids in egg yolk did not seem adequate and additional sources of lipids was required for normal growth of immature parasitoids (Carpenter et al., 2000). For in vitro rearing of T. basalis 50% chicken egg yolk led to high percentage of egg hatching and larval growth (Volkoff et al., 1992).
Salt solutions used in artificial diets of parasitoids have been mainly Nettles’ or Nisenhiamer’s salt soultuion (Nettles et al., 1983). Volkoff et al. (1992) by adding 25% salt solution in a diet could increase the egg hatching and larval growth compared to diets lacking that. Dahlan and Gordh (1997) reported the improvement of T. australicum growth by adding 10% Neisenheimer’s solution. Heslin et al. (2005b) discussed that removal of Grace’s medium which contains minerals as well as other nutritional factors improved development of T. pretiosum. Yazgan (1981) reported that Itoplectis conquisitor (Say) (Hymenoptera: Ichneumonidae) needed 0.63% mineral salts in its diet to grow normally but for Pimpla turionellae L. (Hymenoptera: Ichneumonidae) more than 0.39% was toxic. The role of salt solution, in addition to be a source of nutrition (House, 1972), is providing a proper pH in diet as well as osmotic pressure. Moreover, it was reported to induce oviposition in female parasitoids (Nettles et al., 1985). The rate of 25% of Nettles’ salt solution (~ 0.006 concentration) used, gave best results in harvesting larval stages.
Significant changes did not occur by adding 10% host eggs contents to diet M12 compared with diet M11 taking larval growth into considerations. However, higher mean of larvae was observed on M12 in total (Table 5). There are reports that adding parasitized egg contents to artificial diets may give rise to enhanced larval growth mainly because of teratocytes which are inserted into host eggs by female parasitoids while parasitizing (Volkoff et al., 1992). The role of teratocytes in growth and development of immature parasitoids has been discussed in details by Dahlman (1991), Falabella et al. (2000) and Consoli et al. (2001).
Among diets containing milk with C. suppressalis larval hemolymph, M3 was significantly better than M2 at 3 h but both were the same at 24 and 48 h and in those with that of P. unipuncta, M6 preformed significantly better than M7 at 3, 24 and 48 h. No record of survived larvae after 48 h was registered on diets containing milk (Table 3). Volkoff et al. (1992) obtained some results and concluded that milk might have negative effects on genus Trissolcus in artificial diets.
Present study showed that, with respect to ever increasing knowledge of nutritional ecology of parasitoids and their food requirements, it is possible to enhance and improve in vitro rearing of species in Scelionidae family. Efforts to rear T. grandis on artificial diets resulted in rearing them up to 168 h (second larval instar). The reasons for having no pupae or adults on artificial diets could be because of:
| • | The lack of essential materials for formation of pupal stage which should have been absorbed by larvae before second larval instars (Volkoff et al., 1992). |
| • | Extra liquid diet in the medium after completing second instar larvae (Grenier and Liu, 1990). |
| • | Impossibility of making a defined artificial diet based on the compositions of host egg (E. integriceps) (Daskalova and Dimitrova, 1994). |
| • | Poor gas exchange in rearing cells (Volkoff et al., 1992). |
Efforts to remove obstacles in artificial rearing of natural enemies specially parasitoids will be a great achievement in augmentative biocontrol programs.
This study is dedicated to the soul of Dr. H. Bayat-Assadi, late HOD, Biological Control Research Dept., PPDRI, who passed away recently after over 3 decades of intensive research and extension of biological pest control in IR Iran. I would like to thank Plant Pests and Diseases Research Inst. (PPDRI), Agricultural Research and Education Org. (AREO), IR Iran, to grant the project No. 107-11-75-141, which this article is a part of its results. I would also like to thank Mr. A. Nasrollahi, IROST, for providing Antheraea pupae. The help of Dr. M.A. Baghestani, HOD, Weed Science Dept., PPDRI and Mr. B. Imani, IROST, in analysis of data is appreciated.