ABSTRACT
The present study focuses on the stomatal characters of 54 species from 6 families of monocotyledons, the majority of which are grasses. The stomatal density, guard cell lengths on the adaxial and abaxial leaf epidermis and the stomatal type in each family are described and the relationship between stomatal density and guard cell size is reviewed. These plants are collected from one of the Irano-Turany protected area. However, studying of stomata characters, particularly herbaceous plants growing in less contaminated zone such as protected areas, could improve basic knowledge on main characters of monocotyledons.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2006.1650.1659
URL: https://scialert.net/abstract/?doi=pjbs.2006.1650.1659
INTRODUCTION
Stomata have a fundamental role in controlling both photosynthesis and transpiration as the most important plant processes. The developing leaves respond by adjusting their stomatal density or stomatal index. Clues to the nature of these interactions and the molecules involved are bringing fresh understanding of the factors that regulate the density and patterning of stomata in response to environmental stimuli. Environmental factors, notably light, also exert a control over stomatal density, (developing under high levels of light has increased stomatal densities). A growing number of studies use the plant species inverse relationship between atmospheric CO2 concentration and stomatal density. Lake et al. (2000), McElwain and Chaloner (1995) have provided evidence that stomatal frequency declines in response to increasing CO2 and may have occurred over geologic time. Stomatal frequency in present day plants can be manipulated by growing them at different CO2 concentrations (Tomlinson, 1961; Woodward, 1987; Beerling and Woodward, 1995), although some species undergo no change (Oberauer et al., 1985). The absence of patterning change may indicate that some species are less sensitive to CO2 and/or normally produce stomata in excess.
Gas exchange can also being flounced by the abundance of stomata on the leaf surface, measured as the number of stomata per unit area of leaf surface (stomatal density) or the proportion of stomata relative to the total epidermal cell number (stomatal index). Regulation of stomatal density can be considered as a means of setting limits on the rate of gas exchange for a given set of conditions.
There are also good correlations between paleaoclimate model-derived estimates of atmospheric CO2 levels and the stomatal density and index of fossil plant and are therefore useful indicators of past atmospheric CO2 levels. It has been known for several years that leave of plants stomatal density and stomatal index as indicators of pale atmospheric CO2 concentration (Van et al., 1993; Poole et al., 1996; Royer, 2002).
Other way to reach such means is to compare data of particular species plants, collected from uncontaminated sites and the data extracted from same species in sites with more CO2 emissions.
Studying of stomata characters, particularly herbaceous plants growing in less contaminated zones such as protected areas, could serve as a tool for developing above ideas and conducting more researches.
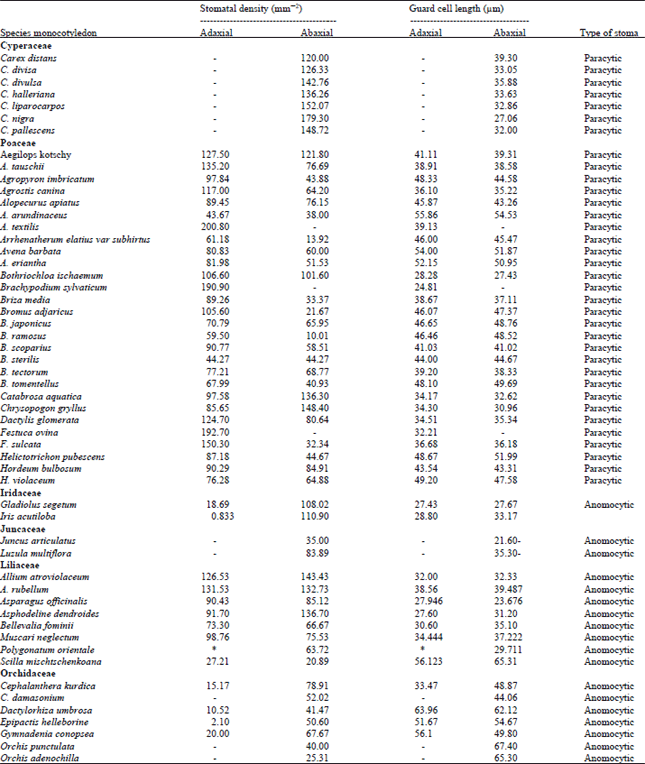
This study aims to collect such data and prepare the ground for further elaboration regarding CO2 emission on internal structure of the leaves including stomatal density and guard cell length (Table 1). It is also of the objectives of this study to elaborate the correlation of stomatal density and guard cell size in different species of monocotyledons and check against some stomatal theories, including Eames theory as well as Willmer theory, regarding arrangements of stomata in superficial surface of the leaves in monocotyledons.
For such purposes, the study focuses on the stomatal characters of 54 species from 6 families of monocotyledons, the majority of which are grasses. The stomatal density, guard cell lengths on the adaxial and abaxial leaf epidermis and the stomatal type in each family are described and the relationship between stomatal density and guard cell lengths is reviewed. These plants collected, from an Irano-Turany protected area.
According to the Eames et al. (1925), in the majority of leaves, stomata are arranged apparently without regularity. Salisbury (1975) found that in some broad-leaved plants frequencies were greater at the leaf margins than near the midrib. While in some monocotyledons leaves, which have an intercalary leaf meristem, frequencies increasing from the base, reaching a maximum value approximately in the middle of the leaf lamina, before decreasing in frequency toward the tip. More recent studies on the monocot, maize, show a different pattern of stomatal frequency along the leaf in young developing and mature leaves frequencies initially decreased from the base along the sheath up to the tip and then slowly increased, reaching biggest value towards the leaf tip (Heichel, 1971). Smith et al. (1989) have mapped stomatal frequencies using iso-frequency contours and discovered that highest frequencies were near the leaf margins and lowest frequencies next to the central main.
MATERIALS AND METHODS
Plant material 54 species from 6 families of monocotyledons were selected to represent a broad spectrum of stomatal characteristics. All materials were identified by the Iranian central herbarium (TARI). The main research as well as laboratory study regarding this study including fixed materials was conducted in the Laboratory of Plant Anatomy, Faculty of Basic Science of Tarbiat Modarres University of Iran in 2005.
To minimise misinterpretation, for analysis, the data entry comprises an average of 30 samples taken from 6 plants of the same species. In order to study stomatal density, the diafanization technique was employed (Stritmater, 1973) and the results were observed using a light microscope. The stomatal classification scheme of Metcalfe (1960) was used as a basis to define the various stomatal forms encountered in this study.
RESULTS
Cyperaceae (Fig. 1 a-h): In the Cyperaceae, stomata are present only on the abaxial surface. At the leaf margins stomata are absent. Stomata occur in linear groups with up to 5 rows and occasionally more stomata next to the central vein. The highest frequencies are observed in Carex nigra (179 mm-2) and the lowest in C. distans (120 mm-2). Only paracytic stomata are found in the species studied. Guard cell length ranges between 27.06-39.3 μm (Zarinkamar, 2002).
Poaceae (Fig. 2 (a-f) and 3 (a-g)): In the Poaceae leaf blade, stomata are usually observed on both surfaces. The exceptions are Alopecurus textils, Brachypodium sylvaticum and Festuca ovina, where stomata are completely absent from the abaxial surfaces. Adaxial stomatal densities are higher than those of abaxial surfaces throughout the rest of family, though in Avena eriantha, Bothriochola ischaemum, Catabrosa aquatica and Chrysopogon gryllus these are vice versa. Stomata are usually situated in regular rows in lamina, in Bromus genus that highest frequencies were near the leaf margins and lowest frequencies next to the central vein. Overall densities are highest in A. textilis (averaging at 200 mm-2) and lowest in A. arundinaceus with an average stomatal density of 43 mm-2. The differences in frequency between adaxial and abaxial surfaces show wide variability, without any genus specific phenomena. Guard cells are large and the average cell length on adaxial surfaces (55.86 μm) is greater than those of abaxial surfaces (30.96 μm). The stomatal size and frequency is similar on both surfaces in Bromus sterilis.
Iridaceae (Fig. 4 a-f): In the two species of Iridaceae, examined in this study, stomata are found on both surfaces, with overall higher abaxial than adaxial densities. The differences between the two leaf surfaces is considerable; the abaxial stomatal density (at around 110 mm-2) is high in both species, with the adaxial density very low in Gladiolus segetum (18.69 mm-2) and even lower in Iris acutiloba (0.833 mm-2). This is similar to Orchidaceae and in contrast with the Liliaceae and Gramineae. Stomata are arranged apparently uniform without regularity in whole leaves. The stomatal type is anomocytic. Abaxial guard cell lengths (average 33.17 μm) are greater than adaxial guard cells (average 27.43 μm). Longer guard cells are associated with higher stomatal frequencies (Zarinkamar, 2005).
Juncaceae (Fig. 5 a-f) : In the Juncaceae, stomata are only found on abaxial surfaces. Stomata are regularly ordered usually situated in linear groups. The stomatal density in both species studied is between 35-83 mm-2. The stomatal type is anomocytic and the guard cell lengths vary between 21.6-35.3 μm.
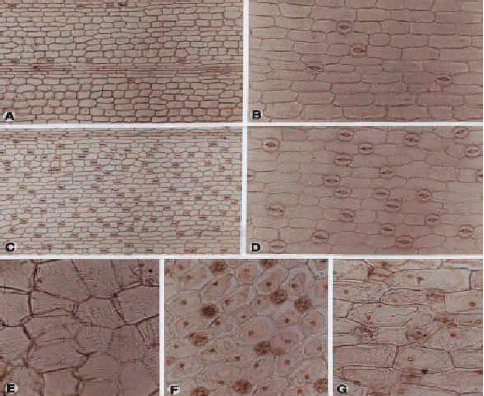
Liliaceae (Fig. 6 (a-f) and 7 (a-h)): In the Liliaceae, stomata are present on both surfaces, with the exception of Polygonatum orientale, where stomata are completely absent from the adaxial surface. Special arrangements of stomata are evident in a genus specific manner; e.g., in Asparagus, Bellevalia, Muscaria and Scilla stomatal frequencies on adaxial surfaces are greater than those of abaxial surfaces, whilst in Allium and Asphodeline, these are vice-versa.
 | |
| Fig. 1: | A-H, observation of abaxial epidermis in superficial view of the Cyperaceae: A, B Carex divisa; C, D, C. liparocarpos; E, F, C. distans; G, H, C. divisa; A, C, E, G, (×150); B, D, F, H, (×300) |
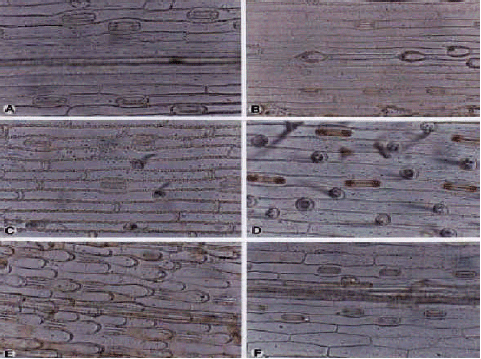 | |
| Fig. 2: | A-F, epidermis in superficial view of the Poaceae; A, B, Agrostis canina; A, adx; B, abx; epidermis; C, abx epidermis of Agropynun imbricatum; D, abx epidermis of Bromus tomentellus; E, adx epidermis of Catabrosa aquatica; F, adx Epidermis of Dacrylis giomerata; A-F, (×300) |
 | |
| Fig. 3: | A-G, epidermis in superficial view of the Poaceae; A-C, Bromus adjaricus; A, adx; B, C, abx; D,E, Helictotrichon pubescens; D, adx; E, abx epidermis with trichomes; F, G, Hordeasn violaceunc; F, adx; A, B, F, G, (×300); C-E, (×150) |
 | |
| Fig. 4: | A-F, epdermis in superficial view of the Iridaceae; A-D, Iris acutiloba; A,B, adx; C, D, abx; E, F, Gladivlus segenon; E, adx; F, abx; A,C,(×150); B, D-F, (×300) |
 | |
| Fig. 5: | A-F,epidermis in superficial view of the Juncaceae; A, B, abx epidermis of the Juncus articlanes; C-F, Laszula mutiflora; C, D, adx; E, F, abx; A, D, E, (×300); B, F, (×600); C, (×150) |
 | |
| Fig. 6: | A-F, epdermis in superficial view of the Liliaceae; A, B, Allium atroviolaceun; A, adx; B, abx epidermis of Allium rubellum; E, F, Bellevalia fominii; E, adx; F, abx; A-C, E, (×300); D, F, (×600) |
 | |
| Fig. 7: | A-H, epidermis in superficial view of the Liliaceae; A, B, abx epidermis of Muscari neglectum; C-E, Polygontum orientale;C, adx; D, E, abx; F-H, Scilia mischischenkoana; F, G, adx; H, abx; A, D, (×150); B, C, F, H, (×300); G, (×600) |
 | |
| Fig. 8: | A-G, epdermis in superficial view of the Orchidaceae; A, B, Cephalanthera damasonium; A, adx; B, abx; C, D, C. kurdica; C, adx; D, abx; E-G, Epipactis helleborine E, adx; F, G, abx A-F, (×300); G, (×600) |
 | |
| Fig. 9: | A-G, epidermis in superficial view of the Orchidaceae: A-D, Gymnadenia conopsea; A, B, adx; E, F, Orchis punctlata; E, adx; F, abx G, abx epidermis of O. Steveni; A, C, (×60); B, D-G, (×150) |
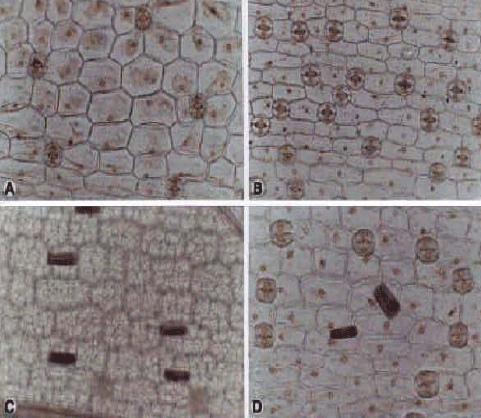 | |
| Fig. 10: | A-D, epidermis in superficial view of the Dactyirrhiza umbrosa; A, D, adx; B,C, abx; A-D, (×150) |
Allium has the highest stomatal frequencies of all species in this study, with an abaxial average density of 143 mm-2. The lowest densities are found in Scilla mischtschenkoana with 27 mm-2 on adaxial and 20 mm-2 on abaxial surfaces, respectively. The difference in frequency between both surfaces is often minor. Stomata are arranged without regularity except in Muscaria tending to situate in parallel rows. The stomatal type is anomocytic occasionally stomata observed in twin. Longer guard cells are always associated with higher stomatal frequencies, (with the exception of S. mischtschenkoana), for e.g., in the genera Allium and Asphodeline, the largest guard cells are always found in abaxial surface, whilst in Asparagus, Bellvalia and Muscaria abaxial cells are greater in size. Guard cell lengths vary from 23.64 to 39.48 μm. the size of stomata is similar on both surfaces in Allium atroviolaceum (32 μm).
Orchidaceae (Fig. 8 and 9 (a-g) and 10 (a-d): In the Orchidaceae, stomata are present on both surfaces, with the exceptions of Orchis and Cephalanthera damasonium, where stomata are absent from the adaxial surfaces. The differences in stomatal density between surfaces are considerable, with much higher frequencies on abaxial surfaces. Abaxial stomatal density for the family is (78 mm-2), this is greater than the adaxial average (25 mm-2). Stomata are arranged without regularity on a whole leaf. The stomatal type is anomocytic. Occasionally stomata observed unpaired guard cells as well as result in stomatal twinning. Average Guard cell lengths vary between 33.47-63.96 μm for adaxial surfaces and 67.4 μm for abaxial surfaces. The presence of calcium oxalate raphides occur in Dactylorhiza umbrosa. The presence or absence of the different crystal types may represent useful taxonomic characters.
DISCUSSION
Existing stomatal theories observe that stomatal density has correlation with guard cell lengths, when stomata are presents on both leaf sides in herbaceous plants (Willmer, 1993). Eames et al. (1925) believed that in the majority of monocotyledons leaves, stomata are arranged apparently without regularity. However in some monocots, for example, in the grasses, stomata are situated in regular rows, each stoma oriented in the someway.
To elaborate such theories and prove the correlation of stomatal density and guard cell lengths and address any exceptions, the information gathered in Table 1.
The distribution of stomata is irregular in intercostals areas in Iridaceae, Liliaceae, Orchidaceae for they are usually absent from the coastal regions and appear less numerous along the periphery of the lamina than elsewhere. In Cyperaceae, Poaceae and Juncaceae stomata are in linear groups. The orientation of stomatal pores is most often parallel to the long axis of the leaf.
In the considerable species of Poaceae, stomata present on both surfaces of the lamina and stomatal densities are less numerous on the abaxial compare with the adaxial surface. The highest recorded adaxial stomatal densities are found in Alopecurus textiles with 200.8 mm-2 and Chrysopogon gryllus have been reported with as high as on abaxial 148.4 mm-2, under ordinary mesophytic conditions. Although in studied species of Iridaceae, Liliaceae and Orchidaceae stomata observe on both surfaces of the lamina, in the majority of these species stomatal density is less on the adaxial than on the abaxial surface.
As observed, in Cyperaceae and Juncaceae families stomata are present only on the abaxial surface of the lamina. In some species of Liliaceae stomatal density is higher than adaxial, whilst other species show opposite.
The size of stomata in Table 1 shows some degree of uniformity with species. The common size is about 30-50 μm, with the exception of Orchidaceae family, which has the longest guard cell in species under studied (67.4 μm in the Orchis punctulata). The smallest guard cells length is 21.6 μm in Juncus articulatus (Juncaceae). These sizes are all about abaxial surfaces.
Table 1 shows that in the majority of the studied species of monocotyledons, where stomata are present on both surfaces (e.g., Poaceae), there is correlation between increasing guard cell size and increasing stomatal density. It shows that while stomata are found on both surfaces of the leaf, the correlation between increasing the guard cell size and increasing the stomatal density is proved. This behaviour with in families of herbaceous species of monocotyledons did not confirm the original stomatal theory and could be considered as a significant finding of this study. The exception to this new finding is about Bromus jenous, Helictotrichon pubescens, Bellevalia fominii, Muscari neglectum, Scilla mischtschenkoana and Dactylorhiza umbrosa species which the lack of correlation of stomatal density and guard cell size is observed.
Further study is needed to realise correlation of stomatal density and guard cell size in dicotyledones.
| Table 1: | Stomatal density, guard cell length for adaxial and abaxial epidermis and type of stomata |
 | |
However, initial findings show Willmer theory covered main families of dicotyledones while some of them show the same behaviour of majority of monocots particularly Poaceae family (Zarinkamar, in preparation).
Realize stomatal type, Table 1 shows they are anomocytic in Orchidaceae, Liliaceae, Juncaceae and paracytic (guard cells alone, those with two subsidiary cells) in Cyperaceae and Poaceae. The type of stomata in the lamina of the grasses is surprisingly uniform, considering the diversity of habitat in which grasses are growing.
REFERENCES
- Heichel, G.H., 1971. Stomatal movements, frequencies and resistances in two maize varieties differing in photosynthetic capacity. J. Exp. Bot., 22: 644-649.
Direct Link - McElwain, J.C. and W.G. Chaloner, 1995. Stomatal density and index of fossil plants track atmospheric carbon dioxide in the Palaeozoic. Ann. Bot., 76: 389-395.
Direct Link - Oberauer, S.F., B.R. Strain and N. Fetcher, 1985. Effect of CO2 enrichment on seedling physiology and growth of two tropical species. Physiol. Plant., 65: 352-356.
Direct Link - Poole, I., J. Weyers, T. Lawson and A. Raven, 1996. Variations in stomatal density and index: Implications for paleaoclimate 1233-8. Reconstructions. Plant Cell Environ., 19: 705-712.
Direct Link - Smith, S., J. Weyers and W.G. Berry, 1989. Variation in stomatal characteristics over the lower surface of Commelina communis leaves. Plant Cell Environ., 12: 653-659.
CrossRefDirect Link - Zarinkamar, F., 2002. Foliar anatomy of Carex in Arasbaran, NW Iran. Iranian J. Bot., 9: 261-270.
Direct Link - Van Der Burgh, J., H. Visscher, D.L. Dilcher and W.M. Kurschner, 1993. Pale atmospheric signatures in Neogne fossil leaves. Science, 260: 1788-1790.
CrossRefDirect Link








