ABSTRACT
The study was conducted to optimize in vitro plantlets production from callus in two sugarcane cultivars. Sugarcane (Saccharum officinarum cv. SPF-213, CPF-237) embryogenic calli were induced from young leaves cultured on MS basal medium supplemented with 3.0 mg/l 2,4-D and 0.1 mg/l NAA. Three concentrations of two different growth regulators (6, benzylaminopurine and kinetin) were tested with and without NAA to compare their ability to induce regeneration from embryogenic calli. After 4 weeks of culture, the percentage of shoot induction was evaluated while after 6 weeks, the total number of shoots produced was checked. Medium containing BA @1.0 mg performed better than Kinetin with the highest percentage of shoot induction.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2002.1081.1083
URL: https://scialert.net/abstract/?doi=pjbs.2002.1081.1083
INTRODUCTION
Sugarcane is one of the most important cash crop of Pakistan. The entire sugar requirement of Pakistan along with the multi billion sugar industry is solely dependent on the fate of this crop. Considerable difficulties have been encountered in the improvement of sugarcane through hybridization (Maqbool and Akhter, 2000). High yielding varieties with built in pest and disease resistance have been developed during the past five decades through conventional breeding and selection programme. However this method of breeding requires 10 to 15 years to release a variety because of greater genetic complexity of the crop. Tissue culture techniques play an important role in the genetic improvement of vegetatively propagated crops like sugarcane (Liu and Chen, 1984; Krishnamurthi, 1986; Siddiqui et al., 1994). The in vitro plants regenerated from callus are capable of producing somaclonal variants for different traits like high yield, more sugar recovery, disease resistance, drought tolerance and early maturity etc.
Rapid callus formation has been obtained mostly from young leaves (Brisibe et al., 1994; Niaz and Quraishi, 2002). To promote regeneration, callus was transferred to medium with different growth hormones (Irvine et al., 1991).
The objective of the present research was to study the regeneration potential of two sugarcane varieties with supplementation of cytokinins and auxin in sugarcane germplasm on defined MS media. It has become increasingly clear that under the appropriate culture conditions, a great deal of genetic variability can be recovered in regenerated plants.
Materials and Methods
Sugarcane varieties SPF-213 and CPF-237 selected in this study were taken from sugarcane germplasm field at the National Agricultural Research Center (NARC) in collaboration with Co-ordinated programme on sugarcane during Dec-February. Embryogenic calli were induced from young leaves as described by Niaz and Quraishi, 2002. Calli which were 3 months old were cultured on MS (Murashige and Skoog, 1962) medium supplemented with either BA, Kinetin at three different concentrations i.e. 0.1, 1.0, 2.0 with and without 0.2 mg NAA. Sucrose was added at the rate of 2% w/v. pH of the medium was adjusted to 5.7 before autoclaving. Total number of regenerated calli and average number of shoots were recorded after four weeks of culture. Each treatment was replicated 10 times. Cultures were incubated at 25±2°C under 2000 lux of light for 16/8 h light/dark photoperiod.
RESULTS AND DISCUSSION
Regeneration is the ability of protoplast to develop the highly structured morphology of a whole plant. When calli of certain size are transferred from culture medium to regeneration medium, the further regeneration process is not fundamentally novel. It follows either the organogenic or embryogenic pathway depending on genotype, donour tissue and culture medium used (Skarzhinskaya, 1996).
Different experiments were conducted in an attempt to find the optimum culture conditions for shoot regeneration from embryogenic calli. Vigorous calli were induced from leaf explants of Saccharum officinarum on MS media supplemented with 2.0 mg 2,4-D in SPF-213 while CPF-237 produced callus on media supplemented with 3.0 mg /l 2,4 D (Niaz and Quraishi, 2002). It was noted that auxin (2,4-D) had already shown a good response to callus induction and proliferation in both the cultivars tested (Chen et al., 1988; Fitch and Moore, 1990; Oropeza and Garcia, 1996). This may be due to its chemical stability (Jha and Roy, 1982). The effect of different concentrations of BAP and Kinetin with and without NAA in MS were studied in terms of shoot initiation from callus of two cultivars. The type and concentration of growth regulators used in culture medium had a significant effect on induction of shoots.
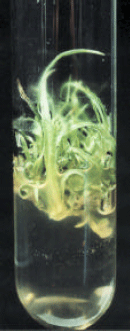 | |
| Fig. 1: | Saccharum officinarum cv. SPF-213 showing multiple shoots on MS medium containing BAP @ 1.0 mg. |
| Table 1: | Effect of different concentration and combination of BA, NAA and Kin on shoot initiation from embryogenic calli of SPF-231 |
 | |
| Table 2: | Effect of different concentration and combination of BA, NAA and Kin on shoot initiation from embryogenic calli of CPF-237 |
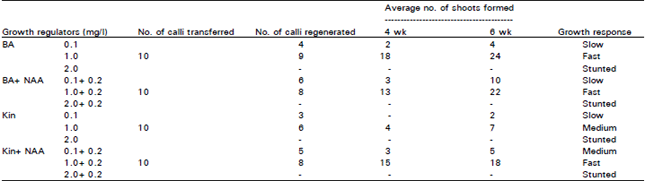 | |
Medium containing BAP (1.0 mg/l) had the highest percentage of shoot initiation in all the concentrations in SPF-213 (Fig. 1) and for callus proliferation but showed similar response towards regeneration. However addition of 0.2 mg NAA to the media did not alter shoot induction when combined with BA. Effectiveness of BAP alone for in-vitro shoot regeneration and multiplication was also reported in some other plants (Conover and Litz, 1987).
After 6 weeks of culture, the total number of shoots was noted. Highest number of shoots was observed on media containing BAP (1.0 mg/l) followed by Kinetin + NAA. Same results were reported by Chengalrayam and Gallo-Meagher, 2001, Prajapati et al., 2000). Minimum number of shoots was observed on medium containing Kinetin (0.1 mg/l) without NAA (0.2 mg/l) followed by BA. However no shoots were observed on media containing BA (2.0 mg/l) and Kinetin (2.0 mg/l) with and without NAA which confirmed that high concentration of growth hormones may hinder the regeneration ability (Siddiqui et al., 1994).
It has surfaced that in media containing Kinetin, addition of NAA significantly increased the number of shoot production. However, with their positive effect on shoot production, these treatments still produced fewer shoots than BAP. This supports the fact that NAA may enhance the number of shoots produced depending upon the cytokinin used, but BAP alone remain the most effective treatment. It was also found that growth rate of callus multiplication and regeneration was dependent upon the concentration of growth hormones and type of explants used (Mujib, 1992). It has been concluded that plantlets formation could be increased after the addition of BAP alone. BAP and Kinetin in combination with NAA induced shoot formation in the same concentration with difference in number of shoots. However high concentration of growth regulators beyond 1.0 mg stunted the shoot formation.
It has also been noted that callus regeneration was also visible on media containing 2, 4-D in various concentration in both the cultivars but a sustained multiplication of callus was not possible on the same medium. It was earlier reported that 2, 4-D had the ability to perform to some extent the function of both auxins and cytokinin. However it is not yet known as to how it duplicate the function (Bhattacharya et al., 2000).
Similar results were also observed on MS media containing Kinetin with NAA (Table 1). It has been observed that BAP and Kinetin had similar intermediate effects. Earlier reports showed that combination of NAA with Kinetin promoted rapid regeneration from sugarcane callus (Irvine et al., 1991). Similar results have been achieved in both cultivars (Table 2).
ACKNOWLEDGMENT
The authors wish to thank Dr. Maqbool Akhtar for providing the source material.
REFERENCES
- Chen, W.H., M.R. Davey, J.B. Power and E.C. Cocking, 1988. Control and maintenance of plant regeneration in sugarcane callus cultures. J. Exp. Bot., 39: 251-261.
CrossRefDirect Link - Chengalrayam, K. and M. Gallo-Meagher, 2001. Effect of various growth regulators on shoot regeneration of sugarcane. In vitro Cell Dev. Biol. Plant, 37: 434-439.
CrossRef - Fitch, M.M.M. and P.H. Moore, 1990. Comparison of 2,4-D and picloram for selection of long-term totipotent green callus cultures of sugarcane. Plant Cell Tissue Organ Culture, 20: 157-163.
Direct Link - Niaz, F. and A. Quraishi, 2002. Studies on somatic embryogenesis in sugarcane. J. Biol. Sci., 2: 67-69.
CrossRefDirect Link - Murashige, T. and F. Skoog, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant., 15: 473-497.
CrossRefDirect Link








