Research Article
Effects of Refrigerated and Frozen Storage on the Survival of Campylobacter jejuni in Ground and Cubed Beef
Not Available
Harun Aksu
Not Available
Ozge Ozgen
Not Available
Muammer Ugur
Not Available
Campylobacter jejuni infection is one of the most common bacterial enteric pathogens. Very few number of microorganisms, about 500/g, can cause infection and this improves its importance for public health (Brownsell et al., 1989). People become infected by consumption of raw and undercooked contaminated food products (Ewans et al., 1998). Restaurants provide opportunities for outbreaks of food-borne disease, because large quantities of different foods are handled in the same kitchen. Failure to wash hands, utensils, or countertops can lead to contamination of foods that will not be cooked.
This organism has been isolated commonly from poultry meat and other meat. (Atanassova and Ring, 1999; Uyttendaele et al., 1999). C. jejuni is known as a microaerophilic organism. For providing the optimum growth conditions 5% O2, 10% CO2, and 85% N2 must be present in the medium (Cliver, 1990). Therefore, the organism on meat surface is inhibited by normal atmosphere (Arwana and Scheibner, 1988). Previous studies showed that C. jejuni survived a long time in refrigerated and frozen ground and cubed meat, but is inactivated in a short time at ambient temperatures (Van Netten et al., 1998). Varadaraj (1993) informed that the inhibition rate of campylobacters at 25 °C were eight times faster than at 4 °C. In vacuum and modified atmosphere packing, C. jejuni may survive longer depending upon gas combination (Tomancova et al., 1991; Finali et al., 1996).
The aim of this study was to determine further the survival characteristics of C. jejuni during refrigerated and frozen storage of ground and cubed beef.
Materials and Methods
Test Organism: In the present study, a strain C. jejuni (biotype I) used as test organism was isolated from poultry carcasses. For isolation, Modified Campylobacter Blood Free Selective Agar (Oxoid) was used. For identification mobility test, gram staining and biochemical tests were performed (FDA, 1995). Biotyping was performed according to Lior’s scheme (Lior, 1984). The test organism was stored in Semisolid Brucella Broth (Oxoid) and Modified Preston Campylobacter Selective Enrichment Broth (Oxoid) at 4 °C.
Inoculation into meat :For preparation of inoculum, test organism was activated and multiplied by plating on selective medium. The plates were washed with sterile physiologic saline and the cell concentration of this suspension was measured by turbidimetric method in spectrophotometer (Collins and Lyne, 1985). The suspension was diluted and then inoculated to ground and cubed beef for approximately 105 cfu per gram organism. For homogeneous dispersion of inoculum, artificially contaminated meat samples were mixed thoroughly. Before inoculation, the ground and cubed meat samples were cultured to detect possible natural contamination with Campylobacter. No positive samples were found. The ground and cubed beef used in trials was from carcasses which had been refrigerated for three days after slaughter.
Storage of ground and cubed beef : After inoculation, the ground beef and cubed beef samples were placed in sterile plates by dividing in portions of 500 g and then wrapped with polythene film. They were divided into two groups. The first group was stored for 9 days in refrigerator (4 °C) and second group for 60 days in deep freezer (-18 °C). These samples were evaluated for number and presence of C. jejuni at 1st, 2nd, 3rd, 5th, 7th and 9th days of refrigerated storage and 10th, 20th, 30th, 45th and 60th days of frozen storage.
Determination and enumeration of C. jejuni: For enumeration of C. jejuni, serial dilutions were prepared from refrigerated and frozen meat samples and were plated onto Modified Campylobacter Blood Free Selective Agar (Oxoid) by the spread plate method. Plates were incubated in microaerophilic conditions (5% O2, 10% CO2, 85% N2), using gas generating kit (Oxoid, BR56) at 43 °C for 48 h., and then evaluated typical C. jejuni colonies. To determine the organism in uncountable level, the same samples were cultivated on Modified Preston Campylobacter Selective Enrichment Broth (Oxoid) and then plated the agar. In suspicion, biochemical tests were performed ( FDA, 1995).
The trials were repeated three times and in each trial 20 kg beef with 20% fat was used.
Results
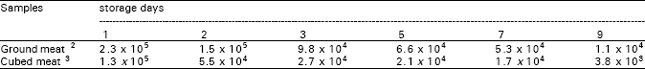
The initial C. jejuni colony counts were 4.1 x 105 cfu/g for the ground meat and 4.3 x 105 cfu/g for cubed meat. During the refrigerated storage at 4 °C, the counts of test organism in the meat samples reduced continuously. On 9th days, when the samples were not acceptable for sensorial, these counts was declined to 1.1 x 104 cfu/g for the ground beef and to 3.8 x 103 cfu/g for cubed beef (Table 1).
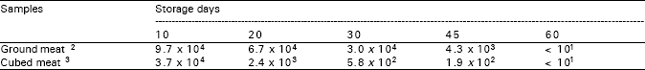
Similar reductions were also observed during the frozen storage at -18 °C (Table 2). On 45th day of storage, the counts of organism in the ground and cubed beef samples were respectively 4.3 x 103 cfu/g and 1.9 x 102 cfu/g. At the end of storage (60th day), the organism was on uncountable level in both ground and cubed meat. But, it could be isolated from enrichment medium.
Discussion
Infected meat is an important source of C. jejuni and the carcasses principally become contaminated with C. jejuni from intestinal contents during slaughter (Gill and Harris, 1982). Nevertheless, studies have shown that, at post-slaughter stage, C. jejuni on meat surface was easily inactivated (Arwana and Scheibner, 1988). But it can be resistant to certain conditions in meat.
In the artificially contaminated ground and cubed meat samples, the cell counts/g of the test organism decreased slowly throughout the refrigerated storage period. Even on 9th day of storage, it was present in high level (1.1 x 104 cfu/g in ground meat; 3.8 x 103 cfu/g in cubed meat). These results agree with those of Koidis and Dolye (1983), who studied the ability of C. jejuni to survive in fresh ground beef during refrigerated storage and to identify time-temperature treatments needed to inactivate Campylobacter in ground and cubed red meat. They determined that C. jejuni survived well in refrigerated ground beef containing large numbers of indigenous bacteria and relatively little death (less than 1.2 log10 reduction) occurred during 14 days at 4 °C. In study of Refaie and Galal (1992), the cubed fresh beef were inoculated with C. jejuni isolates and then stored at different holding temperatures. Their results suggested that C. jejuni survived for 14 days at 4 °C. Stern and Kotula (1982) reported that in the contaminated ground beef samples stored at 4 °C, no changes in viability of C. jejuni were observed over 14 days of storage. Hazeleger et al. (1998) studied the physiological activity of C. jejuni far below the minimal growth temperature and observed that the vital cellular processes were still functioning at temperatures as low as 4 °C. In addition to cold storage, vacuum packing and modified atmosphere packing of meat products prolonged the survival of C. jejuni (Finali et al., 1996). Although the organism may survive for a long time in refrigerated ground meat, it has lower incidence in refrigerated red meat carcasses (Vanderlinde et al., 1998). C. jejuni is fairly sensitive to normal atmosphere and therefore, can not survive on meat surface. Tomancova et al. (1991) suggested that low aw and O2 reduced the C. jejuni counts on surface of refrigerated meat. These results show that, depending on particle size, cutting contaminated meat into pieces may serve to create micro environments that may favour survival of C. jejuni. In the present study the test organism counts in the ground meat were higher than in cubed meat at all stages of storage.
In contrast to other pathogens, C. jejuni inactives at ambient room temperature faster than refrigerator conditions. Van Netten et al. (1998) reported that increase in storage temperature from 4 to 12.5 °C delayed decrease in numbers of Salmonella typhimurium, Escherichia coli and Staphylococcus aureus except C. jejuni and deaths in C. jejuni at 12.5 °C slightly exceeded those at 4 °C. Refaie and Galal (1992) determined that C. jejuni in cubed meat survived only three days at 20-25 °C and 14 days at 4 °C. Tomancova et al. (1991) also concluded that the low storage temperature prolonged survival of the C. jejuni to 2 days.
Freezing of foodstuff affects the type and number of present flora. In deep-frozen products, no microbial growth is observed but some of microorganisms may be reduced or destroyed during freezing process. The destruction effect of slow freezing is more than that of rapid freezing. In the present study meat samples were frozen by rapid method. Results showed that the test organism could maintain its viability throughout the storage period at –18 °C. Besides a significant decrease in C. jejuni count was observed during frozen storage and no growth was determined from 10-1 dilution on 60th day, it could be still detected from enrichment broth. But the longer storage may probably result in total inactivation.
These findings about frozen meat are in agreement with the findings of other studies which investigated the survival of C. jejuni in frozen meat. Refaie and Galal (1992) observed a decrease in C. jejuni counts in cubed frozen meat and determined that this organism survived by 42 days at -20 °C. In raw ground meat stored at –19 °C, Barrell (1984) determined only 1 log unit decrease for C. jejuni count on first week. Tomancava et al. (1991) reported that freezing had only partial effects on C. jejuni devitalization and C. jejuni in meat survived for 2-8 weeks.
| Table 1: | C. jejuni colony counts ( cfu/g)1 during refrigerated storage (4 °C) |
 | |
| Table 2: | C. jejuni colony counts ( cfu/g)1 during frozen storage (-18 °C) |
 | |
Stern and Kotula (1982) reported that in the contaminated samples (107 cells per g) held at -15 °C over 14 days of storage, the numbers of C. jejuni declined by 3 log10. Brownsell et al. (1989) indicated that C. jejuni survives on freezing well and the increasing use of frozen meat may have contributed to an increase in its incidence. Lee et al. (1998) investigated the role of packing and storage conditions on the replication of C. jejuni on chicken. In this study, chicken skin pieces were artificially inoculated with C. jejuni and stored at different temperatures and under various packaging conditions. They reported that C. jejuni remained viable at –20 and –70 °C and quickly replicated after the sample thawed.
This survival capacity of the organism during cold storage constitutes a risk for ground and cubed red meat have a short shelf-life. The infectious dose can be as low as a few hundred bacteria and insufficient cooking may result in the survival of the organism at level that is able to cause the infection (Flynn et al., 1994). Especially, a combination of inadequate cooking time and use of large meat pieces increase the risk of campylobacteriosis (Evans et al., 1998). Whereas properly performed cooking process inactivates C. jejuni, Boer and Hahne (1990) indicated that the organism can easily contaminate the cutting boards and other utensil from contaminated raw meat and reported that C. jejuni was isolated from cooked products and raw vegetables contacted with plates which raw meat were just put in. In an outbreak of C. jejuni infection that occurred in USA, it was determined that the most likely source of infection was eating lettuce or lasagne cross-contaminated from raw chicken in a restaurant kitchen (Graves et al., 1998).
Data of this study showed that C. jejuni were reduced depending upon time in ground and cubed meat during cold storage but survived at level which can cause the infection. Even meat was not acceptable for sensorial.
This work was supported by the Research Fund of the University of Istanbul (Project number: 769/131295).