Research Article
Production of Chitosan by Fungi
Not Available
J. Kader
Not Available
Mohd. Mazmira
Not Available
D.C.Paul
Not Available
Chitosan (β-1,4-D-glucosamine) is produced by the chemical or spontaneous deacetylation of chitin (β-1,4-N-acetyl-D-glucosamine). This molecule is rarely found in living organisms and is abundant in the cell wall of certain fungi such as zygomycetes (Arcidiacono et al., 1988). Crab and shrimp shell derived chitosan has a wide and varied scope of applications in industries such as agriculture and pharmaceuticals due to its high amine content and polycationic nature (Knorr, 1991; Rane and Hoover, 1993). The crustacean chitosan is inconsistent in its physico-chemical properties due to the variability in raw materials, caustic effects of the chemicals used in the isolation process, variability in the levels of deacetylation and protein contamination (White et al., 1979). Due to these inconsistencies the zygomycete has been considered as an alternative source of chitosan as it has more of a constant physico-chemical nature. This is achieved by careful manipulation of the growth parameters such as pH and composition during the fermentation process. These manipulations result in chitosan of a varying molecular weight (Arcidiacono and Kaplan, 1992). A variation in the molecular weight will alter its application (Rane and Hoover, 1993).
There are several attributes that make fungi a very amenable source for chitosan production. The most important being the lack of protein that could cause an allergy reaction in individuals with allergies towards shellfish. The extraction process from fungi is simple and cheap and therefore results in the reduction in time and cost required for production (White et al., 1979). In addition fungi can be grown easily on any simple medium or industrial by products therefore liberating the production of chitosan from the dependence on the seasonal shellfish industry (White et al., 1979; McGahren et al., 1984). Due to the ability of fungi to grow on industrial by-products and waste this will also free the environment of pollution concerns.
In this study fungal isolates were screened for their ability in producing chitosan. Previous researchers have stipulated that the highest levels of chitosan is obtained during the active growth phase, specifically the late exponential phase (McGahren et al., 1984; Hang, 1990; Hansson et al., 1992). Therefore in this report we choose to determine the optimum harvest time, the most suitable isolate and the glucose concentration necessary for the highest chitosan yield.
Materials and Methods
Isolation, cultivation and extraction: Fifteen fungal isolates were obtained from soil, fermented food and the empty fruit bunch (EFB) of oil palm. All the isolates were studied for their ability to produce chitosan. The results presented here however are of the isolates that produced yields comparable or higher than isolates previously used in the chitosan fermentation. The isolates were maintained and subcultured on potato dextrose agar (Oxoid). The cultures were allowed to grow for five to ten days and the spores were harvested in distilled water with 2% Tween 20. The fungi were cultured in 500ml flask containing 300ml of Yeast-Peptone Glucose (YPG) broth (glucose [1 to 5%], 1% peptone, 0.1% yeast extract, 0.5% (NH4)2SO4, 0.1% NaCl, 0.5% MgSO4.7H20 and 0.01% CaCl2 in distilled water at pH5.0) and incubated with shaking at 300C with continuous shaking at 150rpm. The mycelium cell mass obtained from the growth was separated from growth medium through a vacuum filter using Whatman No. 4 filter paper. The mycelium was washed a few times with deionized water before proceeding with the acid extraction protocol (White et al.,1979; McGahren et al., 1984).
Determination of growth curve of isolates: The growth curves of the isolates were determined for each glucose concentration in YPG broth. The inoculum size was fixed at 1x 107 spores per flask. This was done by adding 5 ml of distilled water containing the above inoculum size into 295ml of YPG broth. The mycelial was harvested at intervals of 12, 24, 36, 48, 60, 72, 84 and 96 hours. The mycelial mass was then washed and freeze dried before the mycelial dry weight was determined. All the above time points were done in replicates.
Determination of chitosan levels in isolates: The chitosan content within the isolates were determined using the above mentioned extraction protocol. The amount of extractable chitosan of all fungi was evaluated for all time points mentioned above. This was done to draw a correlation between the growth phase, AIM (alkaline insoluble material) and chitosan produced throughout the experiment.
The mycelial biomass of all the isolates showed a tendency to increase rapidly during the first 72 to 84 hours. Beyond this time point the dry weight measured did not increase and therefore this indicated that the isolates had entered the stationary phase. The AIM (alkaline insoluble material) and the chitosan obtained increased for the first 60 to 72 hours, after which both declined or remained unchanged with time. The decline in the amount of extractable chitosan obtained may be due to physiological changes in the cell wall of the fungi.
| Table 1: | Mycelial, AIM and extractable chitosan obtained from zygomycetes |
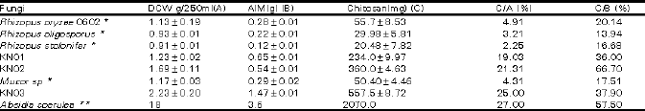 | |
| * Data obtained from Tan et al., 1996. DCW obtained from 250ml culture filtrate ** Data obtained from McGahren et al., 1984.DCW obtained from 1L culture filtrate | |
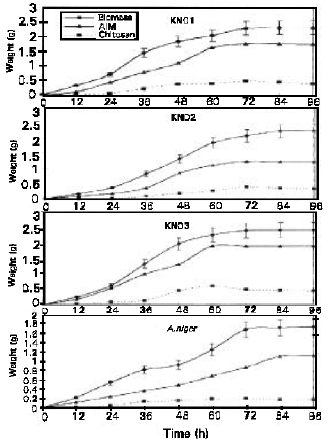 | |
| Fig. 1: | The biomass, AIM and chitosan obtained from the four isolates. The replicate data of biomass is presented in the graph. The AIM and chitosan values are representatives of the highest values obtained. Values obtained are at 1% glucose concentration |
It is believed that as the fungi enters a non active growth phase the chitin (precursor of chitosan) becomes more securely to the lipid, proteins and carbohydrates components in the cell wall (Bartnicki-Garcia, 1968; McGahren et al., 1984; Tan et al., 1996) making it more difficult to extract.
The results presented in Figures 1 indicate that the highest amount of extractable chitosan was obtained at the late exponential growth phase of the fungi. Shimara et al. (1988) in his research using Absidia butleri HUT 1001 showed that though the amount of AIM obtained from this isolate continued to increase until day 10, but the highest yield in chitosan was observed on day 4. This coincides with the late exponential phase of this fungi. In our study (Fig. 1) and others the late exponential phase seems to be the ideal phase for chitosan extraction. This suggests that growth profiles of fungal isolates need to be studied prior to determination of harvest and extraction period. It is not ideal to fix an extraction time for all fungal isolates for fear that the amount of extractable chitosan obtained may not be maximum.
The four isolates showed clear distinguishable growth phases. Mucor sp KN03 arrived at the late exponential stage at 60 hours. Whilst A.niger, Rhizopus sp KN01 and Rhizopus sp KN02 reached their late exponential stages at 72 hours. As in all previous research, the highest levels of extractable chitosan for all isolates were at their late exponential stage (Fig. 1). Glucose concentration seemed to influence the rate of growth. Concentrations of glucose from 1 to 3% showed an increase in the biomass obtained. Whilst concentrations of 4 and 5 % had a detrimental effect on the growth of the fungi. Both these concentrations (4 and 5%) only sustained growth for approximately 48 hours. The cultures died thereafter. Therefore the results obtained with 1 to 3% glucose was analysed statistically to determine the effect of concentration on fungal growth. The statistical analysis conducted showed that there was a significant statistical difference (α=0.05) in biomass produced with the above glucose concentrations. There was however no significant statistical difference observed in extractable chitosan obtained in these glucose concentrations. Though glucose is a precursor in the biosynthesis of chitin (Gooday, 1994), increasing the glucose concentration did not result specifically in increased chitin biosynthesis but rather in an overall increase of growth. The glucose supplied could have been used in the synthesis of other cell wall components as well as intracellular substances such as the enzymes. It is therefore unnecessary to increase the concentration of glucose used as it possible to increase chitosan yield through careful manipulation of other growth parameters and the extraction protocol. Therefore a concentration of 1% glucose is sufficient in the production of chitosan.
Table 1 presents the extractable chitosan produced in this research in comparison with those obtained elsewhere. The data presented in Table 1 is at the optimal harvest time for each isolate. The extractable chitosan content varied widely among the fungi evaluated and the variations were observed even between species and genus. In Table 1 the highest yield of chitosan per dry cell weight (DCW) is highest for Absidia coerulea (McGahren et al., 1984) with 27 % yield of chitosan from DCW. This is followed closely by an isolate used in this research Mucor sp KNO3 which produced 25% yield of chitosan from DCW. Both Rhizopus sp KN01 and Rhizopus sp KN02 produced 21.31 and 19.03% of chitosan per DCW respectively. The lowest yield per DCW was obtained with A.niger that produced only 11.2% chitosan per DCW. The highest level of extractable chitosan per ml substrate used in this research was obtained from Mucor sp KN03 which produced 0.587g of chitosan per 2.265g of DCW (per 250ml cultures). Therefore beside Absidia coerulea (McGahren et al., 1984; Rane and Hoover, 1993), Mucor sp KN03, Rhizopus sp KN01 and Rhizopus sp KN02 may also be considered as good candidates for use in chitosan production. However for us to ascertain the application of the chitosan produced from these isolates it is important to characterize its physico-chemical nature through the determination of molecular weight and degree of deacetylation.