Research Article
Isolation and Characterization of Acid Phosphatase from Chickpea Seeds (Cicer arietinum)
Not Available
Mohammad Tariq
Not Available
Anwar Khan
Not Available
Naheeda
Not Available
S.F. Mabood
Not Available
Phosphatases that catalyses the hydrolysis of phosphatase monoesters, a reaction of considerable importance in several metabolic processes, have been isolated from animals and plant tissues as well as from bacterial and fungal cells (Schmidt et al., 1963; Besford, 1979; Fatima et al., 1981). Seeds are easily obtainable source of several enzymes including phosphatases (phosphoric monoester phosphohydrolases). Two classes of phosphatases have been reported. Acid phosphatase class that has optimal pH 4.2-7.4 and alkaline phosphatase class that has optimal pH varies from 8.6-9.4. Since NIFA-88 is a new commercial high yielding, blight resistant variety developed by the mutation breeding technique using gamma radiation (Khan and Javed, 1990) having 2-3 times more protein (20-26%) than other food grain (Hassan and Khan, 1991) and so far as we know the detailed kinetics of the enzyme from plant tissues have not been studied as yet, so a study of acid phosphatase isolation and characterization was undertaken.
Preparation of the Enzyme: The seeds of chickpea variety NIFA-88 were cleaned and ground to a fine powder. The crude enzyme was extracted by shaking powder in 0.1 M acetate buffer (1 g/10 ml) of known pH for 1 hour at 30°C. The mixture was then centrifuged at 5000 rpm for 5 minutes. The supernatant thus obtained was used for further work.
Estimation of Enzyme Activity: Acid phosphatase activity was determined by method of Gutman and Gutman (1940) using p-nitrophenyl phosphate as substrate. P-nitrophenol obtained as the result of hydrolysis of substrate was converted into phenolate ions (yellow colour) at alkaline pH. The yellow colour was read at 405 nm. 900 μl of the substrate (0.1 M) was incubated for 5 minutes at 30°C. Then 100 μl of the enzyme was added to it and mixed. The mixture was further incubated for another 5 minutes. The reaction was stopped by adding 4 ml of sodium hydroxide (0.1 M). The yellow product of p-nitrophenolate anions was formed. Spectronic-20 was standardized at 405 nm with blank, which has 100 μl water instead of the enzyme and the intensity of the product was read. To increase the precision the transmittance was recorded and it was then converted in absorbance as:
A = 2- Log % T
The activity was expressed as milli mole of substrate hydrolyzed per minute.
Effect of pH on the Activity: Acid phosphatase was extracted from chickpea seeds. The effect of pH on extraction of acid pohospatase from chickpea seeds was studied. The results are given in Fig. 1, which suggest that maximum extraction of the enzyme is achieved at pH 5.5. However, from maize the maximum activity of the enzyme was released at pH 8.5 (Fatima et al., 1981). Since these workers used a different extraction medium of different ionic strength, the release of the enzyme seems to depend on the nature as well as concentration of the medium/or on its binding in the tissue.
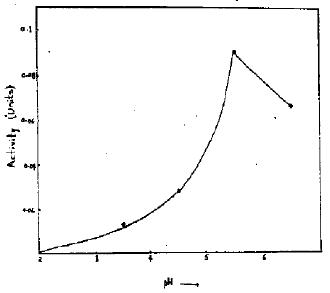 | |
| Fig. 1: | Effect of pH on the activity of the enzyme |
Consequently, a pH 5.5 was used throughout, both for extraction as well as for estimating the enzyme activity because at this pH the enzyme was more stable and gives optimum activity (Fatima et al., 1981; Tahir, 1991).
Effect of Temperature on the Activity: The enzyme activities were determined by incubating the enzyme-substrate mixture at various temperatures varying from 20 to 80°C (Fig. 2). The maximum activity of the enzyme was found at 30°C. Above this temperature, the activity was decreased sharply which may be due to heat denaturation of the enzyme.
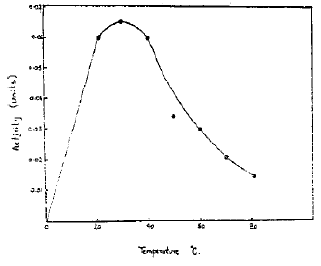 | |
| Fig. 2: | Effect of temperature on the activity of the enzyme |
Effect of Incubation Time on the Activity: Acid phosphatase activity was determined after different incubation times. The results are shown in Fig. 3 which indicate that activity was linear function of time during at least 10 minutes. After this, activity increased but not proportionately. The deviation may be due to product inhibition.
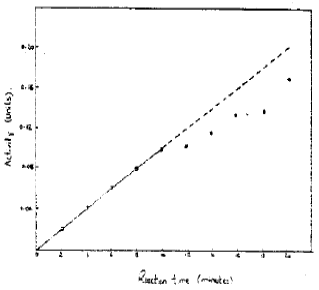 | |
| Fig. 3: | Effect of incubation time on reaction rate |
Effect of Substrate Concentration on the Enzyme Activity: The effect of substrate concentration on the enzyme activity was studied with p-nitrophenyl phosphate as substrate using concentrations between 10 and 400 μM. With increase in concentration of the substrate the enzyme activity increased. The Michaelis-Menten constant (Webb, 1963) Km was determined by Hanes plot as shown in Fig. 4. The value was found to be 8.3 μm.
Effect of Enzyme Concentration on the Activity: The effect of substrate concentration on activity was assayed. The result are shown in Fig. 5. It is evident that the enzyme activity was linear for upto 80 per cent of the enzyme concentration (extract). Hence in further work 80 per cent of the enzyme concentration was used for assaying enzyme activity.
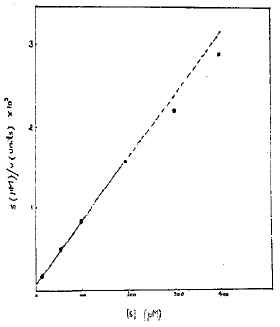 | |
| Fig. 4: | Effect of substrate concentration on s/v versus s hanes plot. |
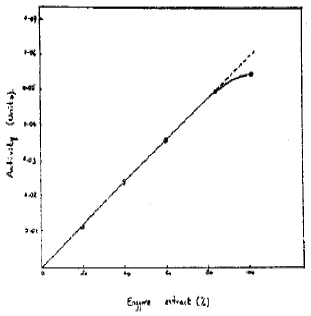 | |
| Fig. 5: | Effect of enzyme concentration on activity |
Effect of in vitro Storage: To investigate the effect of the in vitro storage on the activity of the enzyme, the extract of pH 5.5 was stored at 4°C over a period of 1 month. The results are given in Table 1. It is noticed that there is no obvious change in activity of the enzyme for upto the period of storage. The sort of ability of the enzyme (at 4°C and pH 5.5) suggests that it is a suitable enzyme for advanced kinetic studies.
| Table 1: | The effect of in vitro storage on activity of acid phosphatase from chickpea seeds. |
 | |
Selection of Absorption Maximum: The enzyme was assayed (s 200 μM) at different wavelengths and found that the maximum absorbance was obtained at 405 nm. It is concluded that chickpea is a good source of acid phosphatase and that the enzyme may be a suitable one for advanced kinetic studies.