Research Article
Effect of Fermentation on the Microbial, Nutrient and Anti-nutrient Contents of Melon (Cucumis melo L.) Husk
Federal University of Technology, Akure, Nigeria
M.F. Ekunrin
Federal University of Technology, Akure, Nigeria
The origin of melon is Africa and Asia (Considine and Considine, 1982; Oloko and Agbetoye, 2006) and areas where it is widely cultivated include the Caribean islands, Indonesia and Africa. In Nigeria, the existence of melon dates back to the 17th century. Melon is now cultivated throughout the world, where specific types have been developed for local use as follows: The Cantalupensis group includes cantaloupe, musk melon and persian melon; the Indrorus group includes winter melon, casaba, crenshaw, honey dew, Juan canary and santa Claus; the Flexuosus group includes the snake or serpent melon and the armenian cucumber; the Conomon group includes the oriental pickling melon (Maynard and Maynard, 2000). The fruits are smooth, cylindrical and may be green, white or striped; the Dudaim group includes mango melon, pomegranate melon and queen Anne’s melon; the Momordica group includes the photo and snaps melon.
Melon is a popular fruit in Nigeria, because of the edible seeds, which are commonly used in the preparation of local soup or stew and snacks such as fried melon seed ball known as "Robo" in South-Western Nigeria. Recent statistics show that 100,000 and 488,000 Mt of melon were produced in Nigeria in 1992 and 1997, respectively (FOS., 1998; Oloko and Agbetoye, 2006). In the East, the seeds are sometimes boiled and eaten as snacks too. The seeds are rich in oil (30-50%), which is comparable to other oil plants (Omidiji, 1997; Oloko and Agbetoye, 2006) and the oil contains a high level of saturated fatty acids (Adeniran and Wilson, 1981; Oloko and Agbetoye, 2006). Egusi melon is also an important component of the traditional cropping systems, usually interplanted with such staple crops as cassava, maize, sorghum, etc. (Omidiji et al., 1985; Oloko and Agbetoye, 2006). Melon is a ground creepy plant and thus good for mixed farming systems and its spreading nature covers the ground and as such suppresses weed growth on farm, thereby cutting down on the cost of its production (Bande et al., 2012). Processing of melon involves depodding, fermentation, washing, drying, cleaning and shelling (Oloko and Agbetoye, 2006).
Fermentation in food processing is the conversion of carbohydrate to alcohol, using yeast and/or bacteria, under anaerobic conditions (Ogunshe et al., 2006; Fraizer and Westhoff, 2008). Fermentation offers a number of advantages, including food preservation, improved food safety, enhanced flavour acceptability, increased variety in the diet, improved nutritional value, improved marketability, decreased cooking time and reduction in anti-nutritional compounds (Jimoh et al., 2012; Oyewole and Isah, 2012). Large quantitiesof melon husks are discarded and burnt, which pollute the environment. Information in literature on the nutritional composition and anti-nutrient analysis of melon husks fermented is scarse. In veiw of this, this research aims to study the effect of fermentation on the microbial, proximate and anti-nutrient composition of melon husks.
Source of melon husk: Melon seed (Cucumis melo L.) were purchased from a seller at Oja Oba, in Akure South Local Government area of Ondo State, Nigeria. The seeds were identified and authenticated at the Department of Crop, Soil and Pest Management of Federal University of Technology, Akure, Ondo state.
Processing of melon husk: The sorted seeds were divided into two portions, coded A and B of 100 g each, using electronic weighing balance (Electronic balance, MT-301 Model). The first portion A was analyzed raw, which served as the control while Part B was fermented naturally at ambient temperature for three days. Figure 1 shows the processing method.
Determination of total viable count: One gram of macerated melon husk sample (raw and fermented) was put into 9 mL of sterile water and diluted serially to obtain a dilution of 1010 dilution factor. One milliliter was pipetted from dilution tube (10–3) into sterile petri-dishes.
 | |
| Fig. 1: | Flow chart of the processing of melon husk |
Thereafter, 20 mL of nutrient agar, De Man Rogosa Sharpe and potato dextrose agar cooled to 45°C was poured separately, onto each of the plates in triplicate and the plates were gently swirled and allowed to solidify. The nutrient agar plates were incubated in an inverted position at 37°C for 24 h, while Potato Dextrose Agar (PDA) plates were incubated at 25°C for 3-5 days. The plates of De Man Rogosa Sharpe, which is used for the isolation of lactic acid bacteria were incubated anaerobically in a candle jar. After incubation, the bacterial colonies were observed and counted using a colony counter (Gallenkamp). Representative colonies of bacteria were selected and sub-cultured on fresh bacteriological media until pure cultures were obtained.
Identification of bacteria: The identification of bacteria was based on morphological characteristics and biochemical tests carried out on the isolates. Morphological characteristics were observed for each bacterial colony after 24 h of growth. The appearance of the colony of each isolate on agar media was studied and the characteristics observed to include; shape, elevation, edge, optical characteristics, consistency, colony surface and pigmentation. Biochemical characterizations were done according to the method of Fawole and Oso (2004).
Identification of fungi: The fungal colonies were sub-cultured on PDA. The isolates were identified, based on their morphological and microscopic features. Two drops of cotton-blue-in-lactophenol were placed on clean glass slide and small pieces of mycelium free of medium was removed with sterile inoculating needle and transferred on to the specimen on the slide. The mycelium was teased (picked) out with the needles and covered with a clean cover slip carefully, avoiding air bubbles and observed under the microscope for vegetative and reproductive structures (Hunter and Bamett, 2000).
Proximate estimation: The moisture, crude protein, crude fibre, crude fat ash and soluble carbohydrate (by difference) were determined in accordance with AOAC (2012) methods. The proximate analyses were carried out in triplicate and reported in percentage.
Anti-nutrient determination
Phytate determination: Four grams of finely ground samples were soaked in 100 mL of 2% HCl for 3 h and then filtered. The 25 mL of the filtrate was placed in a 100 mL conical flask and 5 cm3 of 0.03% of ammonium thiocynate solution (NH4SCN) solution was then added as indicator. Fifty milliliter of distilled water was then added to maintain proper acidity and this was titrated against 0.00566 g mL–1 of standard iron (iii) chloride solution that contained about 0.00195 g of iron per milligram until a brownish yellow colouration persisted for 5 min. The equivalent was obtained and from this, the phytate content in milligram 100 g–1 was calculated (AOAC., 2012).
Tannin determination: About 0.2 g of finely ground sample was weighed into a sample bottle. Ten milliliters of 70% aqueous acetone was added and properly covered. The bottles were put in an ice bath shaker and shaken for 2 h at 30°C. Each solution was then centrifuged and the supernatant stored in ice. A 0.2 mL of each solution was pipetted into the test tube and 0.8 mL of distilled water was added. Standard tannin acid solutions were prepared from a 0.5 mg mL–1 of the stock and the solution made up to 1 mL with distilled water. 0.5 mL of Folin Ciocaeteau reagent was added to both, the sample and the standard, followed by 2.5 mL of 20% Na2CO3. The solution was then vortexed and allowed to incubate for 40 min at room temperature after which its absorbance was read at 725 nm AJ- IC03 spectrophotometer against a reagent blank concentration of the same solution from a standard tannic acid curve that was prepared (AOAC., 2012).
Oxalate determination: One gram of the sample was weighed into 1000 mL conical flask. The 0.75 M H2SO4 was added and the solution was carefully stirred intermittently with a magnetic stirrer for 1 h and then filtered using Whatman No. 1 filter paper. Twenty five milliliter of sample filtrate (extract) was collected and titrated hot (80-90°C) against 0.1 M KMnO4 solution to the point when pink colour appeared that persisted for at least 30 sec (AOAC., 2012).
Cyanogenic glycosides: Five gram of the samples were ground into a paste and were soaked in distilled water for 4 h for the liberation of cyanide. The liberated cyanide was steam distilled and 4 mL of 6 N NH4OH and 5% (w/v) KI were added to the distillate portion before titration with 0.02 N AgNO3 to a faint but permanent turbidity (0.02 N AgNO3 = 1.08 mg HCN) (Onwuka, 2005).
Estimation of total phenols: Standard method of AOAC (2012) in which total phenol is expressed as chlorogenic acid equivalent, was used for the estimation of total phenols.
Phytic acid determination: Phytic acid was extracted from each 3 g of the melon husk flour sample with 3% trichloro-acetic acid by shaking at room temperature, followed by high speed centrifugation as described by Steve (2012). This method depends on an iron to phosphorus ratio of 4:6. Five grams of the test sample was extracted with 3% tri-chloro acetic acid. The phytate was precipitated as ferric phytate and converted to ferric hydroxide and soluble sodium phytate by adding sodium hydroxide. The precipitate was dissolved in hot 3.2 N HNO3 and the colour read immediately at 480 nm. The standard solution was prepared from Fe[NO3]3 and the iron content was extrapolated from a Fe(NO3)3 standard curve. The phytate concentration was calculated from the iron results assuming a 4:6 iron:phosphorus molecular ratio. The phytic acid was estimated by multiplying the amount of phytate-phosphorus by the factor 3.55 based on the empirical formula C6P6O24H18.
Statistical analysis: All experiments were carried out in triplicate. Data obtained were analyzed by one-way analysis of variance (ANOVA) and means were compared by Duncan’s new multiple range test (SPSS 21.0 version). Differences were considered significant at p<0.05.
Total viable count obtained from fermenting melon husk: The microbial load of fermenting melon husk as shown in Fig. 2 revealed that the raw husk had no lactic acid bacteria before fermentation. After 24 h of fermentation, the highest bacterial count was observed on nutrient agar medium, which significantly increased (p<0.05) from 12x103- 37x103 CFU g–1 after 72 h of fermentation.
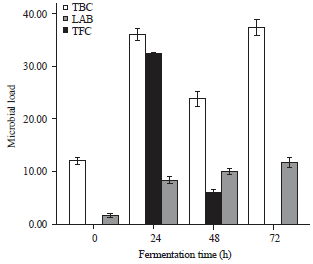 | |
| Fig. 2: | Microbial load (103x) of melon husk during fermentation bars are presented as Mean±SE of replicates (n = 3), TBC: Total bacterial count, LAB: Lactic acid bacteria, TFC: Total fungal count |
Lactic acid bacteria revealed a total colony count of 32.3x103 CFU g–1 after 24 h of fermentation. Fungal count was observed to increase significantly (p<0.05) from 1.67x103-11.67x103 SFU g–1 after 72 h of fermentation.
Effect of fermentation methods on the proximate composition of raw and fermented melon husk: The proximate composition of raw and fermented melon husk as shown in Fig. 3 revealed that ash content of the raw sample was 0.05% but increased to 4.15% after 72 h of fermentation. The moisture content of the raw sample was 2.46% and it increased significantly (p<0.05) to 5.84% after fermentation for 72 h. The fat content of the raw sample was 6.22%. There was a significant increase (p<0.05) to 16.23% after fermentation. The crude fibre of the raw sample was 20.22% while there was a significant (p<0.05) increase to 35.60% in the fermented sample. The protein content of the raw sample was 2.61%. Fermentation increased this significantly (p<0.05) from 2.61-6.54%. Soluble carbohydrate content of raw sample was 13.88% but increased to 31.66% after the 72 h of fermentation.
Effect of fermentation methods on the Anti-nutrient composition of raw and fermented melon husk: The anti-nutrient content of both raw and fermented samples of melon husk is presented in Fig. 4. Cyanogenic glycoside was the most abundant anti-nutrient detected in the raw sample with a value of 65.57 mg g–1. Fermentation process was observed to significantly (p<0.05) reduce the glycosides content to 6.06 mg g–1 after 72 h. Tannin was the lowest anti-nutrient detected in the raw sample with a value of 0.05 mg g–1.
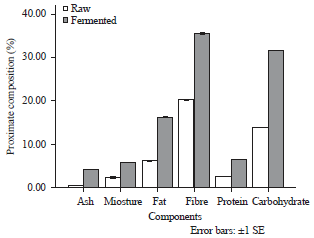 | |
| Fig. 3: | Proximate composition of Melon husk during fermentation, bars are presented as Mean±SE of replicates (n = 3) |
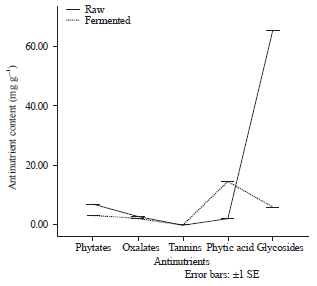 | |
| Fig. 4: | Anti-nutrient content of raw and fermented melon husk |
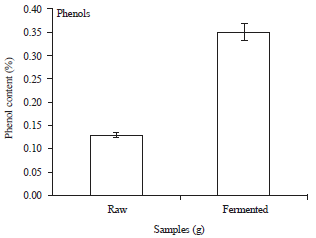 | |
| Fig. 5: | Phenol content (%) of raw and fermented melon husk |
Phenol content slightly increased from 0.13-0.35% after 72 h of fermentation (Fig. 5).
Microbial analysis in this research revealed the presence of Staphylococcus aureus, Bacillus subtilis, B. megaterium, B. licheniformis, Leuconostoc mesenteroides, Lactobacillus plantarum, Rhizopus stolonifer and Saccharomyces ludwigii. These organisms have been reported to be responsible for the fermentation of melon seed (Ogunshe et al., 2007; David and Aderibigbe, 2010). Bacillus spp. had the highest population while L. plantarum had the least count. This is in agreement with the findings of David and Aderibigbe (2010) who reported high population of Bacillus spp. during the fermentation of melon seed.
Ash content of the melon husk significantly (p<0.05) increased after 72 h of fermentation. A similar result was observed by Yabaya (2006), during fermentation of Linn seed for the production of dadawa condiment. The changes observed in the ash content during fermentation could have been facilitated by the enzymatic activities of the fermenting organisms. Fermentation significantly (p<0.05) increased the moisture content of the husk from 2.46-5.84%. The increased moisture content may be as a result of the soaking of the husk. It may also be as a result of decomposition of the fermenting bacteria on the products. This agreed with the report of David and Aderibigbe (2010) on fermentation of melon seed. The increase in crude fat could be a result of extensive breakdown of large molecules of fat into simple fatty acids (Igbabul et al., 2014).
The increase in crude protein values could be attributed to increased microbial mass during fermentation, causing extensive hydrolysis of the protein molecules to amino acid and other simple peptides. The increases observed in protein content agreed with the findings of Akinfemi (2010) during the fermentation of melon husk with the fungi Pleurotus ostreatus and P. pulmonarius. The increase observed in soluble carbohydrate may be due to enzymatic hydrolysis of complex oligosaccharides.
The reduction observed in the anti-nutrient content of the melon husk may be as a result of the enzymatic activities of the fermenting organisms. This is in agreement with the findings of Ibukun and Anyasi (2013) with the fermentation of sesame, musk melon and white melon. The increases observed in total phenolic content is in accordance with the findings of Ibukun and Anyasi (2013) with the fermentation of sesame, musk melon and white melon.
This research has established the effect of fermentation on the nutrient and anti-nutrient contents of melon (Cucumis melo L.) husk. The results of the microbial assays revealed Bacillus spp. had the highest population while Lactobacillus plantarum had the least count. Fermentation process also enhanced the protein and carbohydrate content of melon husk and significantly reduced the anti-nutrient contents. The level of its nutritive value indicates that it can be used as agricultural feed for herbivores, which would solve a waste disposal problem and help in adding value to a waste otherwise, around us.