Research Article
Blood Chemistry of Domestic Rabbits Exposed to an Oxidative Hair Dye
Department of Zoology, College of Science, King Saud University, P.O. Box 2455, Riyadh 11451, Saudi Arabia
Nowadays, millions of consumers use hair daye, all of them have the desire to improve their appearance. Taking into consideration the frequency of using hair colouring products, the ingredients of these products must be highly safe (IARC, 1993). In general, cosmetics and their ingredients must have no harmful effects to human health (Altekruse et al., 1999; Henley and Thun, 2001). This property of cosmetics contradicts to drugs which when assessed a risk-benefit relationship is considered.
Human skin is not impermeable for the topically applied substances, which means that some of these substances or ingredients can be absorbed (Goetz et al., 1988; Burnett and Goldenthal, 1988; Dressler, 1999). This might be the situation in case of hair dyes ingredients. The important hair dye ingredients include para-phenylenediamine (PPD), para-toluenediamine (PTD), para-aminophenol (PAP) and lawsone (Corbett, 1999; Meuling et al., 2003). Some of the investigations conducted on the safety of hair dyes ingredients have indicated some unfavorable results (Altekruse et al., 1999; Corbett et al., 1999; Czene et al., 2003). Here we test the hypothesis that oxidative hair dye may bear some health hazards. To reveal some of these health hazards which could be induced by commercially available hair dyes, the present study was conducted to investigate the effect of an oxidative hair dye on domestic rabbits. The blood chemical parameters of the exposed rabbits were employed to investigate the probable damaging effects.
Animals: Ten adult male domestic rabbits (white haired), of the same age and weighing 1050-1150 g were used. The animals were maintained at the animal facility at the Department of Zoology, College of Science, King Saud University, Riyadh, Saudi Arabia. Acclimatization period extended for two weeks during which the animals were maintained separately in cages under standard laboratory conditions, including ambient temperature (25°C). Standard ration and clean water were supplied ad libitum. Additional group of age-matched rabbits (n = 6) served as a control one.
Hair dye: A commercially available oxidative hair dye was used. The dye consists of two components which differ in consistency, the first component is pasty and of yellowish color and the other is jelly-like and semi-transparent. The two components of the dye were mixed in equal proportions just before application.
Experimentation: Using the brush supplied with the dye package, the dye was weekly applied to the hair on back and scalp regions of the exposed animals. The application process was adequate to paint deeply the hair of rabbits. Experimentation period extended for nine months (March, 2004-November, 2004) and at each time interval the dye was left to dry and then the hair was washed with normal water. Control animals were not exposed to hair dye.
Blood chemistry: Blood samples were collected from all experimental animals at the 1st, 3rd, 5th, 7th and 9th months post-exposure for estimation of plasma total proteins. Blood samples collected at the last interval (9 months post-exposure) were also used to estimate the various blood chemical parameters. The total plasma proteins and blood chemistry parameters were assessed by the analyzer apparatus (Reflotron plus, Roche, Germany) using Reflotron kits (Roche, Diagnostic GmbH D-68298 Mannheim, Germany).
Statistical analysis: The obtained data were expressed as mean value±SD of 10 rabbits (exposed group) and 6 rabbits (control group). The statistical differences between the two groups were evaluated by student's t-test.
Gross findings: Hair on the scalp and back regions of the exposed rabbits was discolored black after the first exposure to the applied hair dye. The blackish discoloration persisted over the experimentation period.
Blood chemistry: Compared with control animals, the serum levels of alkaline phosphatase, alanine aminotransferase and gamma-glutamyl transferase in exposed rabbits were increased significantly (Table 1). The levels of the estimated other two enzymes, aspartate aminotransferase and amylase were increased but insignificantly. Cholesterol level was increased significantly while triglycerides level showed insignificant increase. Glucose levels showed insignificant decrease.
| Table 1: | Blood chemical parameters of rabbits exposed to the oxidative hair dye for 9 months (Exposed, n = 10; control = 6) (values are expressed as means±SD) |
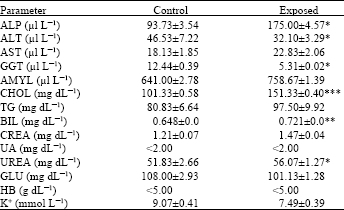 | |
| *p<0.05; **p<0.01 and ***p<0.001; when compared to control level as evaluated by Student's t-test. ALP, Alkaline Phosphatase; ALT, Alanine Transaminase; AST, Aspartate Transaminase; GGT, Gamma-glutamyl Transferase; AMYL, Amylase; CHOL, Cholesterol; TG, Triglycerides; BIL, Billirubin; CREA, Creatinine; UA, Uric Acid; GLU, Glucose; HB, Haemoglobin; K+, Potassium | |
The level of creatinine was insignificantly increased but urea level was significantly increased. K+ level was insignificantly decreased compared to that of the control level.
At all time intervals, the level of total plasma proteins was increased compared to the control level, however the significant increase was recorded from the second interval (3rd month) and continued to the last one (9th month) (Table 2).
The present results indicate that the use of oxidative hair dyes provokes blood chemical changes in the exposed animals. In the present study, the level of some serum enzymes was increased significantly, this included ALP, ALT and GGT. Injury of liver cells that results in cytolysis and necrosis causes elevation of various serum enzymes. The measurement of hepatic enzymes in the serum is useful to assess the extent of liver damage and to differentiate hepatocellular (functional) from obstructive (mechanical) diseases (Batsakis et al., 1974; Zimmerman and Henry, 1979; Chang, 1983). The present result showed that the level of the most common enzymes characteristic for hepatobiliary disorders was increased. As regard to ALP, it is used most often in clinical diagnosis of liver disease. This enzyme may be the only altered serum parameter on routine liver function test (Kaplan, 1986; Davidson and Henry, 1991). Aminotransferases (transminases) include ALT (GPT) and AST (GOT), these two enzymes are commonly used to assess hepatocellular damage. AST is found in all tissues, especially the heart, liver and skeletal muscle while ALT is present primarily in the liver and to a lesser extent in kidney and skeletal muscle. Hence, ALT is more indicative to liver damage (Burnt et al., 1984; Chapra and Griffin, 1985).
GGT is found in high concentrations in kidney and liver and its serum level is elevated in hepatobiliary disorders but it is not specific for certain disease (Schiff and Schiff, 1987; Reichling and Kaplan, 1988).
Regarding the cholesterol serum level, liver is the major site of cholesterol synthesis (Russel, 1992). Increased level of cholesterol, as recorded here, may indicate the compromised ability of the liver to reutilize or excrete cholesterol (Nicolosi et al., 1990; Schaefer and McNamara, 1994). Similar conclusion could be applied to the recorded increased level of triglycerides.
The present exposed cases showed significant increase of urea serum level. Urea constitutes nearly half the non-protein nitrogenous substances in the blood, it is synthesized in the liver and mainly excreted through the kidney (Morgan et al., 1977; Bakerman, 1983).
| Table 2: | Total plasma proteins concentration (g L-1) in exposed rabbits (Exposed, n = 10; control = 6) (values are expressed as means±SD) |
 | |
| *p<0.05; ** p<0.01 and *** p<0.001; when compared to control level as evaluated by Student's t-test | |
Therefore, blood urea is traditionally used to monitor renal function. In our cases, increased urea level may be the result of renal dysfunction which leads to an increase in plasma urea concentration.
Creatinine serum level was increased in the presently exposed animals. Creatinine is synthesized mainly in the liver from arginine, glycine and methionine and when transported to other tissues, such as muscle it is converted to phosphocreatine. Creatine or creatine phosphate loses water or phosphoric acid, respectively to form the anhydride form, creatinine which is excreted into the plasma (Bleiler and Schedl, 1972). Elevated levels of creatinine are merely associated with abnormal renal function, more specifically with reduced glomerular filtration rate (Morgan et al., 1978; Narayabnan and Appelton, 1980). Our exposed cases manifested a mild hypoglycemia (decreased glucose level) which is probably related to lower metabolic activity due to organ failure (Cryer, 1992; Radziuk and Zhang, 1993).
The present exposed cases revealed decreased K+ level. Potassium is the major intracellular cation in the body and most of cellular functions require a low concentration of K+ in extracellular fluid (Maxwell et al., 1987; Rose, 1989). Potassium functions to regulate neuromuscular excitability, cardiac muscle contractility, intracellular fluid volume and hydrogen ion concentration (Maxwell et al., 1987). In hypokalemia, sodium and hydrogen ions move into the cell, subsequently hydrogen ion concentration is decreased in the extracellular fluid resulting in alkalosis (Toffalett, 1993). The cause of hypokalemia in the present hair dye-exposed animals is possibly the resultant renal damage.
The level of the estimated plasma total proteins in the presently exposed animals between 2nd and 5th intervals was significantly lower than that of control animals. At the first interval, the level was comparatively lower but not significantly different. Estimation of plasma proteins is important as it reflects the abnormalities of different organs (Doumas et al., 1981). Hypoproteinemia indicates a condition of negative nitrogen balance and usually it is a consequence to renal diseases (Richie, 1979). In the presently hair dye-exposed animals, renal or hepatic disorders are the suggested causes of hypoproteinemia.
The presently used hair dye is an oxidative (permanent) one, oxidative hair dyes have been found of higher percutaneous penetration absorption rates than the direct hair dyes (Dressler, 1999). The highest percutaneous absorption rate among the hair dye ingredients has been recorded for PTD ingredient of hair dyes (Czene et al., 2003). It was found that the agents responsible for the systemic effects (systemic exposure agents) after the topical application of hair dye ingredients are the acetylated and/or bi-acetylated metabolites of these ingredients molecules (Kawakubo et al., 2000). The present blood chemical changes may point to the ability of the used hair dye to exert systemic effects. Moreover, one or more ingredients of the applied hair dye are supposed to be able to penetrate the skin of the exposed animals to provoke these effects. Future work should be focused on the exact mechanism(s) that leads to the presently manifested blood chemical changes and to assure that tissue damage is among the contributing factors.
This research was supported by the Research Center, College of Science, King Saud University, Riyadh, kingdom of Saudi Arabia (project No. Zoo/24-25/05). This support is gratefully acknowledged.