Research Article
Photosynthetic Response to the Low Temperature in Elephant Grass (Pennisetum purpureum) and Zea mays
Department of Biology, Faculty of Science, Taibah University,P.O. Box 30002, Almadinah Almunawwarah, KSA
Low temperature is one of the most important factors which limit the growth, the distribution and the productivity of plants (Tambussi et al., 2004; Van Heerden et al., 2004; Hu et al., 2006). Most C4 plants are restricted largely to warmer climates because their physiological processes are adversely affected by low temperatures (Long et al., 1983; Hund et al., 2007). However, few C4 plants have been found to be adapted and naturally grow in the cool climates (Beale and Long, 1995). The ability of these plants to activate or to increase their physiological and photosynthetic processes at low temperatures enabled them to survive in these cooler environments (Long, 1999; Naidu et al., 2003; Sowinski et al., 2005).
The photosynthetic production of C4 crops is 40% higher than C3 plants under the optimum climate (Long et al., 1983). However, this advantage is not realised in C4 plants under low temperature conditions. Low temperature damages thylakoid membranes, breaks down chlorophyll and induces reduction in the photosynthetic capacity of C4 plants such as maize (Tambussi et al., 2004; Sowinski et al., 2005). The reduction in the photosynthetic capacity of Z. mays leaves at cool temperatures is associated with reductions in both the light-saturated rate of carbon dioxide assimilation (Asat) and the maximum quantum yield (φ) (Fryer et al., 1998; Foyer et al., 2002; Sowinski et al., 2005; Hund et al., 2007).
Photoinhibition of photosynthesis may occur when the absorption of light is in excess of that required by the plant photosynthetic demands (Savitch et al., 2000). Many studies have shown that plants subjected to photoinhibition at low temperatures, both in controlled environments and in the field (Haldimann, 1999; Tambussi et al., 2004; Van Heerden et al., 2004; Sowinski et al., 2005; Hu et al., 2006; Hund et al., 2007). The phenomenon of photoinhibition is characterized by a reduction of the quantum yield of CO2 uptake (φ) and the ratio of variable to maximum chlorophyll a fluorescence (Fv/Fm) (Fryer et al., 1998; Hu et al., 2006). The reduction in Fv/Fm of dark-adapted leaves indicates photoinhibition of PSII. In addition, the percentage of reduction depends upon the environmental conditions prior to the photoinhibitory treatment and genotypic variability (Lee et al., 2002; Sowinski et al., 2005; Hund et al., 2007).
The previous results (AL-Shoaibi, 2007) showed that P. purpureum had significantly higher growth rate at 14°C compared to Z. mays. This higher growth rate of P. purpureum could be, in part, is due to the consequence of differences in photosynthetic capacity between the two C4 grasses (AL-Shoaibi, 2007). Therefore, this study was carried out to determine the effect of low temperature on photosynthetic CO2 uptake of both P. purpureum and Z. mays and to compare the responses of photosynthesis of the two grasses to low temperature.
Plants and growth conditions: This research was conducted in Biology Department, Faculty of Science, Taibah University during 2006. Rhizomes of Pennisetum purpureum originally derived from Africa and Zea mays cv. LG 80 were planted in pots containing a peat-based compost (F2, Levington Horticultural Ltd., Ipswich, UK.) and grown in high-light controlled environment chambers (Fitotron SGC066. CHX, Sanyo Gallenkamp PLC, Leicester, UK.), at day/night temperatures of 25/20°C and 14/12°C. Fertilisation was provided once a week by irrigating with Hoagland`s nutrient solution (Arnon and Hoagland, 1940). The Vapour Pressure Deficit (VPD) was kept below 1 kPa. Photosynthetic Photon Flux Density (PPFD) at leaf height was 600 μmol m–2sec–1 and the photo period was 14 h.
Gas exchange and chlorophyll fluorescence measurements: The CO2 uptake was measured with an open gas-exchange system incorporating open path infrared, CO2 and water vapour analysers (LI-6400, LI-COR Inc, Lincoln, USA). A Peltier cooling system maintained the leaf temperature at 20°C as described by Long et al. (1996) and VPD was controlled between 1 and 1.5 kPa. The most recent fully expanded leaf was used to measure photosynthetic CO2 uptake at PPFD of 0-2000 μmol m–2 sec–1. Net photosynthesis per unit leaf area and intercellular CO2 concentration (ci) were determined using the equations of Von Caemmerer and Farquhar (1981). The light saturated photosynthesis (Asat) was determined at saturating PPFD (1500 μmol m–2 sec–1) and at the ambient CO2 concentration of 360 μmol mol–1. Carbon dioxide response curves were made over the range of 50-550 μmol mol–1 using PPFD of 1500 μmol m–2sec–1, at a leaf temperature of 20°C. These curves were analysed according to the model of Collatz et al. (1992).
Chlorophyll fluorescence was measured using a portable fluorimeter (PEA, Hansatech, Kings Lynn, Norfolk). The initial (F0) and maximum (Fm) fluorescence emissions were measured after 20 min of dark adaptation and the ratio of variable to maximum fluorescence (Fv/Fm) was calculated as (Fm-F0)/Fm as described by Öquist and Wass (1988). The Fv/Fm ratio was measured weekly for four replicates of each plant using the youngest fully expanded leaves.
Statistical analyses: The data obtained from various analyses and measurements were statistically analysed using analysis of variance (Systat, Inc., Evanston, Illinois, USA).
Leaves of Z. mays growing at 14°C showed a significant reduction in Fv/Fm (p<0.001) compared to those of Z. mays and P. purpureum grown at 25°C (Fig. 1).
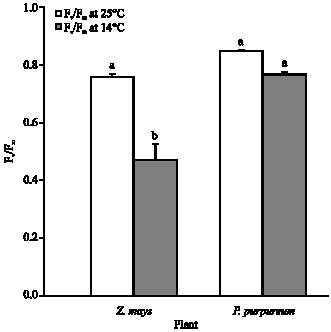 | |
| Fig. 1: | The dark adapted Fv/Fm of P. purpureum and Z. mays. The data represent the mean of n = 3-6 replicate shoots (±SE). Different letter(s) shows the significant different between low temperature treatments (p<0.05) |
Furthermore, the Fv/Fm of P. purpureum leaves grown at 14°C was 64% greater than that of Z. mays leaves (p<0.001) growing at the same temperature (Fig. 1).
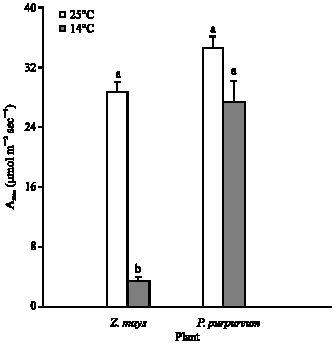
Growth at chilling temperature (14°C), compared to 25°C, had no significant effect on the light-saturated (Asat) or light-limited photosynthetic capacity (φ) of P. purpureum, but caused 78% loss (p<0.001) of both in Z. mays (Fig. 2-4). For leaves of P. purpureum grown at 14°C, the Asat and φ were significantly greater than those of Z. mays grown at the same temperature (Fig. 3-4).
The plateau of the A/ci curve (Amax) is co-limited by the amount of in vivo Rubisco or/and PPDK activity (Collatz et al., 1992). Decreasing temperature from 25°C to 14°C did not significantly affect the Amax of P. purpureum, but caused 78% loss (p<0.001) of Z. mays leaves compared to those grown at 25°C (Fig. 5-6). In addition, the Amax of P. purpureum leaves grown 14°C was more than 7 times greater than that of Z. mays leaves growing at the same temperature (Fig. 5-6).
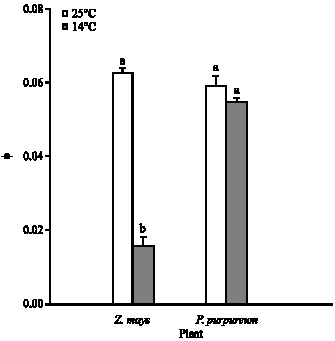
The carboxylation efficiency was calculated from the initial slope of A/ci curve (Fig. 5). Growth at 14°C, relative to 25°C, had no significant effect on the carboxylation efficiency of P. purpureum, but caused 83% loss (p<0.001) of Z. mays (Fig. 7). For leaves of P. purpureum grown at 14°C, the carboxylation efficiency was significantly greater (p<0.001) than those of Z. mays grown at the same temperature (Fig. 7).
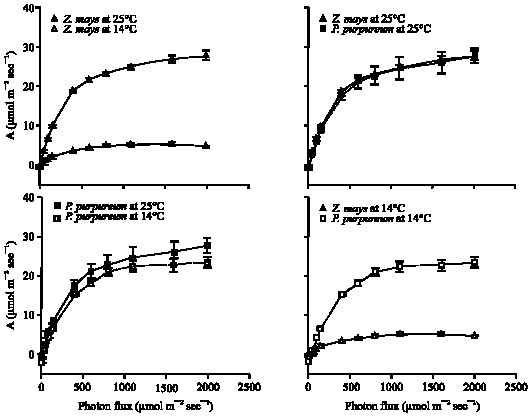 | |
| Fig. 2: | The response of photosynthetic CO2 uptake (A), per unit leaf area, to photon flux (Q) for P. purpureum and Z. mays leaves. Measurements of CO2 uptake were all made at 20°C and a ca of 360 μmol mol–1. The data represent the mean of n = 3-6 leaves (±SE) |
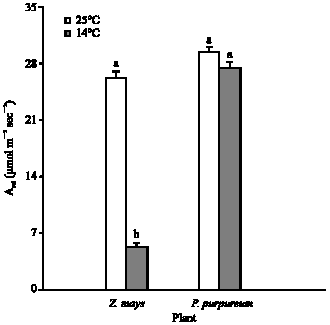 | |
| Fig. 3: | The light-saturated photosynthetic rate (Asat), per unit leaf area, measured at 20°C and a photon flux of 1500 μmol mol–2 sec–1, for P. purpureum and Z. mays. The data represent the mean of n = 3-6 leaves (±SE). Different letters shows significant difference between temperature treatments at p<0.05 |
 | |
| Fig. 4: | The mean quantum yield (φ), measured at 20°C for P. purpureum and Z. mays. The data represent the mean of n = 3-6 leaves (±SE). Different letters shows the significant difference between temperature treatments at p<0.05 |
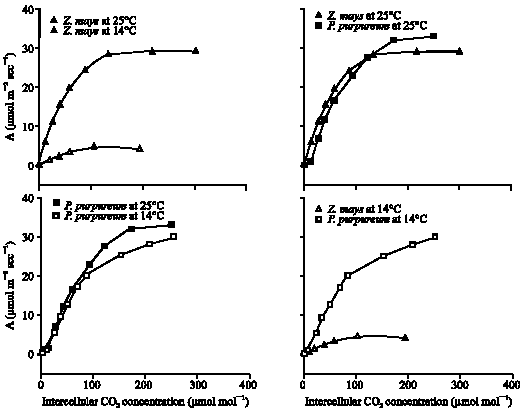 | |
| Fig. 5: | The response of photosynthetic CO2 uptake (A), per unit leaf area, to changes in intercellular CO2 concentration (ci) for P. purpureum and Z. mays leaves. Measurements of CO2 uptake were made at 20°C and a photon flux of 1500 μmol mol–1. Data illustrated are for one selected leaf of each plant |
 | |
| Fig. 6: | The mean plateau (Amax), per unit leaf area, measured at 20°C and a photon flux of 1500 μmol mol–2 sec–1, for P. purpureum and Z. mays. The data re present the mean of n = 3-6 leaves (±SE). Different letters shows significant differences between temperature treatments at p<0.05 |
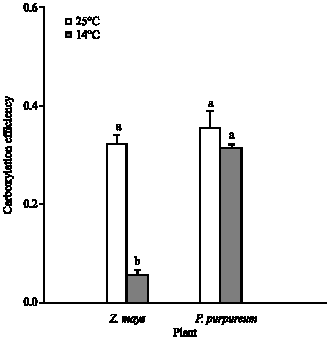 | |
| Fig. 7: | The mean carboxylation efficiency, measured at 20°C and photon flux of 1500 μmol mol–2 sec–1, for P. purpureum and Z. mays. The data represent the mean of n = 3-6 leaves (±SE). Different letters shows significant differences between temperature treatments at p<0.05 |
The results of this research provide a clear evidence that P. purpureum showed significantly greater resistance to photoinhibition at low temperatures than Z. mays. The low Fv/Fm in Z. mays grown at low temperature may not be related to the photoinhibition, but to the impaired development of the photosynthetic apparatus. Nie and Baker (1991) and Nie et al. (1995) showed that several chloroplast polypeptides were poorly expressed in Z. mays leaves developed at 14°C. These lesions, which included the D1 protein, could account for the low Fv/Fm even in the absence of direct photoinhibition.
The photosynthetic rates for all leaves grown at 25°C of both P. purpureum and Z. mays were similar and close to the rates recorded previously for healthy leaves of a range of NADP-malic enzyme type C4 grasses (Ehleringer and Pearcy, 1983). This indicates that these plants were unstressed and not suffering any photoinhibition at the optimal growth temperatures and as indicated by high Fv/Fm. When grown at 25°C, P. purpureum had similar Asat, φ, Amax and carboxylation efficiencies to P. purpureum grown at 14°C, but Z. mays showed a very substantial decrease in all these measures. Moreover, rates of CO2 uptake of P. purpureum grown at 14°C were in excess of Z. mays grown at 14°C at all light levels. The decrease in the photosynthetic performance in Z. mays grown at chilling temperatures was observed previously in controlled environments and in the field (Nie and Baker, 1991; Nie et al., 1992; Haldimann et al., 1996; Prioul, 1996; Fryer et al., 1998).
The sensitivity of C4 photosynthesis to low temperature is frequently associated with the reduction in the rate of enzymatically controlled reactions (Long et al., 1983). This was investigated in vivo by A/ci analysis. The Amax and carboxylation efficiency are controlled by activity of Ribulose 1,5-bisphosphate carboxylase/oxygenase (Rubisco) and activity of Phosphoenol pyruvate carboxylase enzymes (PEPC), respectively (Collatz et al., 1992). Also, the regulatory enzyme Pyruvate, Pi dikinase (PPDK) is another limitation for Amax decreased (Furbank et al., 1997). PPDK is known as a cold sensitive enzyme (Usami et al., 1995; Du et al., 1999). Research work with Flaveria bidentis and Amaranthus edulis indicates that, at 25°C, Rubisco accounts for 50-70% of the metabolic control over A at 360 μbar CO2, while PPDK and PEPC account for only 20-30% (Dever et al., 1997; Furbank et al., 1997; Von Caemmerer et al., 1997). Results of the Amax and carboxylation efficiency of the 14°C grown P. purpureum leaves were similar to leaves grown at 25°C (Fig. 5-7). In contrast, the Amax and carboxylation efficiency of the 14°C grown Z. mays leaves suggested a reduction of 84% in the in vivo activity of these enzymes compared to leaves grown at 25°C. This suggests that in contrast to Z. mays, P. purpureum has similar activity of PEPC, PPDK and Rubisco regardless of whether it is grown at 25 or 14°C.
In conclusion, P. purpureum has superior photosynthetic rates at chilling temperatures than Z. mays. The values of Fv/Fm were significantly reduced only in Z. mays at 14°C compared to 25°C and as a result, the photosynthetic apparatus of P. purpureum was more resistant to chilling temperature than that of Z. mays. This high capacity of photosynthetic rates of P. purpureum at chilling temperatures may be responsible for the high growth rates of this grass at chilling temperatures.
The author is indebted to Professor Dr. Moustafa EL-Naggar for his critical reading and revision of the manuscript.