Research Article
Zinc Nutrition of Wheat: I: Growth and Zinc Uptake
Not Available
B.J. Alloway
Not Available
K.H. Shah
Not Available
S.H. Siddiqui
Not Available
M.Y. Memon
Not Available
M. Aslam
Not Available
P. Khan
Not Available
Zinc deficiency was first recognized by Nene (1966) as a problem of rice on an alkaline me soil in Indo-Pak subcontinent. Later on, deficiency was reported in rice areas in Pakistan (Yoshida et al., 1973; Yoshida and Takana, 1969). The occurrence of zinc deficiency has been reviewed by many researchers (Kausar et al., 1976; Hadi et al., 1997) and the survey revealed that 70% of the cultivated soils are Zn deficient in Pakistan.
In general, soils of arid and semi arid regions are most inclined to Zn deficiency. However, crops are not equally susceptible to Zn deficiency. When grown on the same soil, some crops may suffer from Zn deficiency, while others are not affected (Takkar et al., 1976). Zinc deficiency is common in both cold and warm climates and million of hectares soils grown to cereals have been reported as Zn deficient. Among the cereals, rice, sorghum and maize are classified as most sensitive species whereas barley, wheat and rye are classified as less sensitive (Clark, 1990). Despite being graded as less sensitive, wheat is severely affected by Zn deficiency in Pakistan. The study was undertaken to investigate the effects of Zn on growth and growth components of wheat.
A sand culture pot experiment was conducted in glasshouse of the Department of Soil Science, University of Reading, UK. The experiment was laid out in a completely randomized design with five Zn treatments and six replications. Out of six replicates, three were harvested at 22nd day (Zadoks stage 19), after transplantation and designated as stage 1 and the remaining three replicates were harvested after 48 days (Zadoks stage 39) were called stage 2. One kg acid washed sand was placed in a plastic pot for each treatment to avoid any Zn contamination. All pots, lids and plastic bottles used for stock solutions were rinsed in 5% HCl for 48 hours. The wheat seeds (cv. Highbury) were germinated on a moist filter paper (Whatman 542) at a dark room temperature of 20±10C.Three days after germination; seedlings of equal size and vigour were transplanted into pots at equal depth and spacing. Six wheat plants were transplanted into each pot. Zinc was applied @ of 0,5,10,15, 20 μg ml-1 along with a basal dose of Long Ashton complete nutrient solution (Hewitt and Smith, 1975) as per irrigation requirement of the crop. The chemicals were purified from Zn contamination by the method of Piper (1942). Fresh solution for irrigation was prepared every time from the stock solution and the pH was maintained at 5.5±0.1 with 0.1M HCl or 0.1M NaOH. After transplantation, the seedlings were initially irrigated with double deionized water for two days and then supplied with a 50% basic nutrient solution for a further two more days, but no Zn solution was included during this time. For the rest of the experiment, the Zn treatments were supplied together with the full-strength basic nutrient solution. Each pot was kept at constant moisture content following the weighing of the pot. The day-night temperatures were set at 22 and 18C, respectively. Artificial illumination was used to maintain daylight for 16 and the relative humidity was around 65%. Two days prior to harvest, the plants were again irrigated with double deionized water alone.
Three replications out of six were harvested at Zadoks stage 19 (9 or more leaves unfolded) for cereals and the remaining three were harvested at Zadoks stage 39 (flag leaf ligule). The plants were dug out of the pots with their root system and washed with deionized and double deionized water. After separating the roots and shoots, the fresh weights were recorded. The plant parts were dried in aluminum dishes at 80oC for 48 h in a large forced drought oven (Wood Colchester Ltd.). After drying, the samples were weighed to record the dry matter yield of the crop for each Zn treatment. The dried samples were separately ground and stored in air tight-stoppered glass bottles. Sub-samples were taken for the chemical analysis and standard laboratory procedures were adopted for chemical analysis (Jackson, 1962).
Crop growth: In general, the crop growth was lower in control pots than in the Zn-treated pots (Fig. 1). The plants were stunted in the control pots and weaker than the plants treated with Zn. The plants, which received 20 μg ml-1 Zn, also showed lower growth than the other Zn-treated plants, which possibly may be due to toxic Zn concentrations in the plants.
Well-defined Zn deficiency symptoms were not noticed in the crop. However, Cu deficiency symptoms of burning of leaves were observed in wheat plants when Zn was applied at the higher rate for a longer time. Safaya (1976) also found that Zn generally depressed the Cu concentration in maize plants and that Cu deficiency was minimized to some extent by a basal application of Cu. Chaudhry and Loneragan (1970) reported a similar effect of Zn on Cu absorption in wheat.
Zinc application had a pronounced effect on the growth of the plants. Dry weight of wheat plants considerably increased with increasing rates of Zn up to 15 μg ml-1. The higher rates of Zn application (20 μg ml-1, above these optimum rates of Zn for wheat crop, depressed the dry weight. The maximum dry weights were 2.02 and 6.54 g pot-1, respectively for stages S1 and S2. The root dry weights of wheat were not affected significantly by Zn applications during stage S1. However, with the passage of time the higher Zn concentration (20 μg ml-1 reduced the dry weight significantly (P≤ 0.05), while all other treatments were non-significant with each other. These results are in agreement with the findings of Safaya (1976), who also obtained a maximum dry matter yield in maize with an application of 10 μg Zn g-1 soil. However, Shukla and Raj (1974) observed either a decrease or no change in the magnitude of the response to Zn applications of 5 to 20 μg g-1 in different wheat genotypes. This may be due to the wide gap in Zn application between 5 and 20 μg g-1. Singh et al. (1986) stated that 20 μg g-1 Zn was the optimum concentration in the soil for obtaining maximum wheat dry matter yield. It seems to be a varietal difference as the responses to Zn fertilization may vary from variety to variety within a crop (Shukla and Raj, 1974).
Reduction in yield at higher Zn applications may be attributed to nutrient imbalance in the plant root system (Pathak et al., 1975), where the higher Zn concentration in plant tissue possibly induces Cu deficiency.
Zinc concentration and uptake: Zinc concentration and uptake were affected significantly (P ≤ 0.05) by the application of Zn (Figs. 2, 3). Zinc concentration in roots and shoots increased linearly with increasing Zn levels in the substrate. The plants of wheat had lower Zn concentrations from the control pots, which ranged between 15-16 μg g-1. At these Zn concentrations in the plants, only a yield response was observed, but with no Zn deficiency symptoms. These levels of Zn in cereals could be low enough to reduce the yield, but not so low to produce visual Zn deficiency symptoms (Ward et al., 1973). Total uptake of Zn in the crops increased linearly, as it is obviously related to yield and concentration.
The data also revealed that Zn concentration in the second growth stage (S2) was lower compared to the first growth stage (S1), but the total uptake was higher. This could be explained as a dilution effect (Fig. 3). Generally, when the rate of plant growth exceeds the rate of uptake of a particular nutrient, the concentration of that nutrient in the tissue decreases or it is diluted in the plant tissue. Singh et al. (1986) also described these decreases in the Zn concentration of the above ground dry matter as a dilution effect, when yield increased from 27 to 35% from the corresponding P treatments.
The decrease in Zn concentration with the passage of time also confirms the dilution effect as discussed above. Zinc uptake increased significantly (P ≤ 0.05) with the application of Zn up to 15 μg ml-1 but further Zn application (20 μg ml-1 decreased it, which may be due to the lower yield at this rate of Zn application.
In the nutrient solution, all Zn was in an available form and the uptake of Zn has been reported to be linear with concentration in the nutrient solution and yield. When given luxury supplies of Zn, appreciable quantities of Zn were transported as a mobile ion from the old leaves to the generative organs (Pendias and Pendias, 1992).
Roots have higher Zn concentrations than shoots at all times and were affected significantly (P≤ 0.05) by the Zn treatments. Pendias and Pendias (1992) also stated that roots often contain much more Zn than tops, particularly if the plants are grown in a Zn- rich medium.
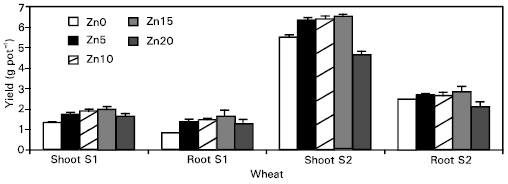 | |
| Fig. 1: | The effect of applied Zn on the dry matter yield of wheat |
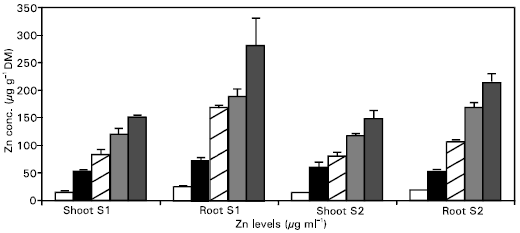 | |
| Fig. 2: | The effect of applied Zn on Zn concentration in wheat |
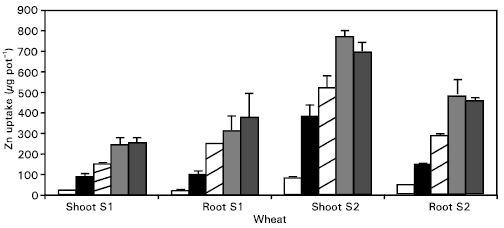 | |
| Fig. 3: | The effect of applied Zn on uptake of Zn by wheat crop |
With luxury levels of substrate Zn, this element may be translocated from the roots and accumulated in the tops of the plant under favourable growth conditions.
Zn is essential for normal plant growth and its deficiency causes stunting in wheat. Zinc and Cu interacted more strongly and markably than the other micronutrients and Cu deficiency symptoms were observed in wheat at higher Zn levels. A dilution effect operated in plants with the passage of time as lower concentrations of elements were observed during the second growth stage.
Iyanu Reply
It was really helpful alot
Editor
Thank you, Iyanu, for your comment on our research article "Zinc Nutrition of Wheat: I: Growth and Zinc Uptake". We are glad to hear that our research has been helpful to you. Our team is committed to conducting research that contributes to the advancement of knowledge in various fields, and we appreciate your support. We hope that our research can continue to be a valuable resource for you and other researchers in your studies. Thank you again for your feedback.