Research Article
Enhancement of γ-Amminobutyric Acid Production by Co-Culturing of Two Lactobacilli Strains
Department of Biological and Geological Sciences, Faculty of Education, Ain Shams University, Cairo, Egypt
Gamma-aminobutyric acid (GABA), which is also known as 4-amino-butanoic acid, is a ubiquitous non-protein amino acid that is widely distributed in nature from microorganisms to plants and animals (Li et al., 2010). It is synthesized by glutamate decarboxylase (GAD), pyridoxal-5-phosphate-dependent enzyme that catalyzes irreversible α-de-carboxylation of L-glutamate to GABA. It acts as the major inhibitory neurotransmitter in the mammalian central nervous system (Huang et al., 2007). In addition, GABA has hypotensive, tranquilizing and diuretic effects and can prevent diabetes (Hayakawa et al., 2005). Also, GABA involved in cardiovascular function regulation such as lowering blood pressure and heart rate (Ueda et al., 2007). Furthermore, GABA would also have an inhibitory effect on cancer cell proliferation, stimulate cancer cell apoptosis. Therefore, GABA has potential as a bioactive component in foods and pharmaceuticals (Kim et al., 2009). However, the direction addition of chemical GABA to food is considered unnatural and unsafe as it causes some diseases such as cancer, heart disease and diabetes (Seok et al., 2008). So it is necessary to find a natural method to produce and increase GABA in food.
In the biological method, microbial fermentation is regarded as the effective one because of its convenience and a high ratio of transformation. Several safe microorganisms including Lactic Acid Bacteria (LAB) have been widely applied in GABA production (Park and Oh, 2007). The LAB possess special physiological activities and are generally regarded as safe and have been extensively utilized in food industries for a long time (Karahan et al., 2010). In addition, the biosynthetic production of natural GABA produced by LAB for the manufacturing of food can make full use of the health-promoting properties of GABA and LAB themselves. In recent years, the GABA production by using LAB as bacterial cell factories has therefore been a focus of research (Li and Cao, 2010). Some fermented products enriched in GABA using GABA producing LAB as starters such as dairy products and cheese have been developed (Li et al., 2008).
This study was aimed at studying various fermentative parameters such as pH, temperature, cultivation time and media additives to obtain a high production of GABA by L. brevis NM101-1 and L. plantarum DSM749 isolated from Egyptian dairy products. Effect of co-culturing on growth and GABA production was also evaluated.
Bacterial strains and culture conditions: The Lactobacilli species were isolated from 24 samples of traditional Egyptian dairy products collected from Cairo markets as described by Rushdy and Gomaa (2013). All strains from the samples were screened for their GABA producing ability. For GABA production, the strains were subsequently inoculated into 250 mL Erlenmeyer flasks containing 50 mL of the production media, which consisted of following compositions (g L–1): glucose, 50; soya peptone, 25; MnSO4•4H2O, 0.01 and Tween 80, 2 mL. Unless otherwise emphasized, the initial monosodium glutamate (MSG) concentration in the medium was 500 mM. The seed medium was composed of (g L–1): glucose, 50; soya peptone, 25; MnSO4•4H2O, 0.01; L-glutamate, 150 mM and Tween 80, 2 mL. Nitrogen sources, glutamate and the other compositions were autoclaved separately at 121°C for 20 min and mixed together prior inoculation. Lactobacilli strains were cultured in the seed medium at 30°C for 24 h and then used for seed culture inoculation. The flasks were inoculated with 10% (v/v) seed culture and incubated with shaking at 30°C for 72 h.
Strain identification
16S rRNA sequencing and analysis: The isolates showing the highest GABA production were identified by 16S rRNA sequencing according to the method of Rochelle et al. (1995). The gene sequencing was done by Macrogen (South Korea). The DNA sequences were aligned using Gene Mapper v. 4.1 and data collection v. 3.1 Communication Patch1. To extract the genomic DNA, bacterial colonies were picked up with a sterilized toothpick and suspended in 0.5 mL of sterilized saline then centrifuged at 8000×g for 10 min. After removal of the supernatant, the pellet was suspended in 0.5 mL of Insta Gene Matrix (Bio-Rad, USA), incubated at 56°C for 30 min and then heated to 100°C for 10 min. After heating, supernatant can be used for Polymerase Chain Reaction (PCR). Bacterial 16S rRNA gene was amplified using the following universal primers for eubacteria: forward primer 27F (5’-AGAGTTTGATCMTGGCTCAG-3’) and reverse primer 1492R (5’- TACGGYTACCTTGTTACGACTT-3’). The PCR was performed using kits with AmpliTaq DNA polymerase (FSenzyme) (Applied Biosystems, USA).
One microlitre of template DNA was added in 20 μL of PCR solution. The 27 F/1492R primers were used for bacteria. Thirty five amplification cycles were performed at 94°C for 45 sec, 55°C for 60 sec and 72°C for 60 sec. The PCR amplicon were purified by using Montage PCR clean-up kit (Millipore). The purified PCR products of approximately 1400 bp were sequenced by using 2 primers 518 F (5’-CCA GCA GCC GCG GTA ATA Cg-3’) and 800R (5’-TAC CAG GGTATC TAATCC -3’). Sequencing was performed by using Big Dye terminator cycle sequencing kit (Applied BioSystems). Sequencing products were resolved on an Applied Biosystems model 3730XL automated DNA sequencing system (Applied BioSystems). Sequence analysis was performed with sequences in the National Center for Biotechnology Information (NCBI), USA database using Basic Local Alignment Search Tool for Nucleotides (BLASTN) (Altschul et al., 1997). All identified isolates were kept at -80°C in De Man-Rogosa-Sharpe broth (MRS) with glycerol (30% v/v). The strains were serially transferred at least three times prior to use.
GABA quantification: The contents of extracellular GABA accumulated in the culture medium were extracted according to the method of Yang et al. (2008) with minor modifications. First, the culture broth was separated from cells by centrifugation (8000×g for 15 min at 4°C) and the supernatant was diluted 50 fold with 7% (v/v) of glacial acetic acid. The diluted sample was centrifuged at 8000×g for 15 min at 4°C and the supernatant was filtered using 0.22 μm pore size membranes and then collected for further analysis.
One hundred microlitre aliquot of the extracellular GABA were derivatives to Phenylthiocarbamyl-GABA (PTC-GABA) and analyzed by HPLC according to Rossetti and Lombard (1996) with minor modifications. The dried residue containing PTC-GABA was dissolved in 100 μL of mobile phase, consisting of a mixture of 80% solution A (aqueous solution of 13.608 g sodium acetate-3-hydrate, 0.5 mL triethylamine, 0.7 mL acetic acid and 5.0 mL acetonitrile in 1000 mL deionized water) and 20% solution B (acetonitrile-water (60:40)) adjusted to pH 5.8. Isocratic HPLC separation was performed using HPLC (Shimadzu Asia PTe.) with UV set of 254 nm and PDA detector. The used column was purospher STAR RP-18 LiChroCART (250-4.6 Merck, Darmstadt, Germany) and the column temperature was set at 35°C. The flow rate for the mobile phase of isocratic elution was 1.0 mL min–1. GABA were detected by comparison of their retention time (Rt) values and spectra with known standards and determined by peak areas from the HPLC chromatograms.
Cell growth: Cell growth was monitored by measuring the optical density at 600 nm using spectrophotometer (UV-VIS Double Beam PC, Labomed INC). Ten milliliter culture medium was centrifuged at 10,000 rpm, 4°C for 15 min and the cell pellet was washed with 10 mL distilled water. The cell pellet was harvested by centrifugation and dried at 105°C for 48 h or till constant weight was obtained. Cell mass concentration was determined by the standard calibration curve between OD600 and cell dry weight.
Effect of initial pH, temperature and cultivation time on GABA production: The effect of pH, temperature and cultivation time on GABA production were determined by cultivating the strains in a culture medium using the following conditions pH (4, 5, 6, 7 and 8), temperatures (25, 30, 35, 37 and 40°C) and cultivation time (24, 48, 72, 96 and 120 h)
Effect of monosodium glutamate (MSG) concentrations and addition of pyridoxal-5-phosphate (PLP) concentration on GABA production: The effect of different concentrations of MSG (250, 500, 750 and 1000 mM) and PLP addition (50, 100, 150 and 200 μM) on the GABA production were investigated at optimum pH, temperature and cultivation time.
Statistical analysis: All experiments were done in triplicate and the results were presented as Mean±Standard Deviation. The experimental data were analyzed by using SPSS. Statistical significance was accepted at a level of p<0.05.
The consumer attention towards the selection of health beneficial foods makes the significant growth of GABA-enriched foods. GABA is present in small quantities in many plant sources, vegetables, for example spinach, potatoes, cabbage asparagus (broccoli), tomatoes, fruits, for example apples, grapes, in cereals, for example barley, maize. The increased amount is found mainly in fermented products, especially fermented dairy products, soy sauce (Yamakoshi et al., 2007), cheeses (Siragusa et al., 2007) and so on. Natural addition of GABA is in high demand over the addition of chemical nutrient GABA since consumers prefer naturally occurring substances and the fermentation helps to reduce the cost of the foods due to the omission of chemical addition of GABA and also provides attractive foods with better taste and at the same time replaces the chemical GABA by natural GABA (Li and Cao, 2010). Therefore, GABA production by naturally occurring microorganisms during fermentation is getting higher request.
The most interesting and practical group of bacteria for GABA production is LAB, which produce high levels of GABA (Watanabe et al., 2007). Lactic acid bacteria possess special physiological activities and are generally regarded as safe. Therefore, using lactic acid bacteria as cell factories for gamma-aminobutyric acid production is a fascinating project and opens up a vast range of prospects for making use of GABA and LAB. In the present study, twenty three lactobacilli species were isolated from 24 dairy samples of traditional Egyptian dairy products. Out of 23 bacterial isolates, eight strains showed GABA productivity. The strains exhibited the highest GABA production were identified by 16S rRNA gene sequence analysis. The sequence alignment using BLASTN software for the comparison up to 1500 bp of the analysis gave a high homology of 99% homology to L. brevis NM101-1 and L. plantarum DSM749, respectively.
Factors affecting GABA synthesis: Generally, bacterial glutamate decarboxylases are located in cytoplasm, whereas glutamate and GABA can be transported through membrane and exchanged, which results in the extracellular accumulation of GABA. Different fermentation factors affect the rate of extracellular GABA production by microorganisms. Among them, the most common and essential factors are pH, temperature, cultivation time and media additives of culture.
Effect of initial pH: The biosynthesis of GABA in microorganisms is mainly regulated by pH, which usually has the most pronounced effect for a fermentation process (Yang et al., 2008). To investigate the effect of different pH levels on biomass and the production of GABA by L. brevis and L. plantarum, the initial pHs of the media were adjusted to 4, 5, 6, 7 and 8. As shown in Fig. 1a, b, pH has a significant effect on the production of biomass and GABA production. The optimum GABA production by L. brevis and L. plantarum reached 80.47 and 75.82 mM was recorded at pH 5. Further increase in the pH led to a decrease of GABA production. These results accorded with the previous reports about the optimal pH values for maintaining the activity of LAB GAD were in the range of 4.0-5.0 (Huang et al., 2007; Komatsuzaki et al., 2008). The higher or lower pH may lead to the partial loss of the GAD activity. Co-culturing of L. brevis and L. plantarum in a ratio of 1:1 showed more drastic difference on the production of GABA, where the extensively large amount of 96.14 mM was recorded at pH5 (Fig. 1c). The GABA production appeared to correlate with the acidification of the medium, hence, it is probable that two strains collaborated in the production of GABA as a result of acid-resistant response.
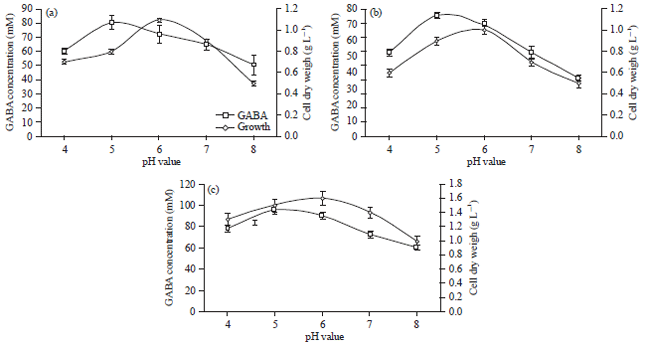 | |
| Fig. 1(a-c): | Effect of the initial pH on growth and extracellular production of GABA of (a) L. brevis, (b) L. plantarum and (c) Co-cultured of both L. brevis and L. plantarum, results are means of three independent determinations and error bars represent the standard deviation |
It was reported that GABA production can be effectively improved by maintaining cellular pH of the medium to acidic conditions because GAD must consume an H+ ion for GABA production (Yang et al., 2008). In addition, pH-regulated genes encoding a GABA transporter was shown to have an acidic pH optimum and preferentially expressed at acidic ambient pH (Espeso and Arst, 2000). In microorganisms, GABA can be decomposed through this pathway: GABA transaminase catalyses the reversible conversion of GABA to succinic semi aldehyde using either pyruvate or α-ketoglutarate as amino acceptor and succinic semi aldehyde dehydrogenase catalyses the reversible conversion of succinic semialdehyde to succinate. Since the above mentioned enzymes have the alkaline pH optimum (Kumar et al., 2000), therefore, acidic condition can block the enzymes that cause GABA to decompose resulted in enhanced accumulation of GABA in the medium.
Initial pH of the culture medium can influence the growth rate of Lactobacilli. The initial pH value of 6.0 caused the highest growth rate of L. brevis and L. plantarum. In the same regard, Zacharof and Lovitt (2010) reported that the maximum growth rate of L. plantarum occurs at moderately acidic initial pH of 6.5. High cell density, in general, is also required for efficient GABA biosynthesis (Li and Cao, 2010). The GAD is the enzyme to make GABA from L-glutamate and utilizes a single proton during the catalysis. This consumption of the intracellular proton is responsible for shifting the intracellular pH toward neutral, hence, it helps microbes to survive under the acidic environment. Since growth and subsequently expression of the GAD gene requires this type of pH changing therefore, stabilization of pH e.g., using buffer system would suppress the gene expression and subsequently GABA production.
Effect of temperature: The incubation temperature is also a major factor affecting maximum GABA yield by fermentation.
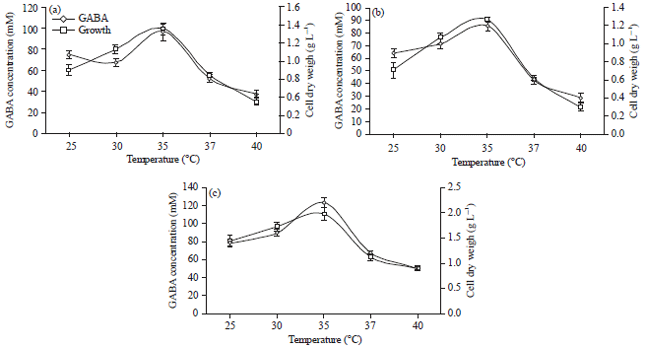 | |
| Fig. 2(a-c): | Effect of cultivation temperature on growth and extracellular production of GABA of (a) L. brevis, (b) L. plantarum and (c) Co-cultured of both L. brevis and L. plantarum, results are means of three independent determinations and error bars represent the standard deviation |
In addition to an effect on biocatalyst activity and stability, temperature has an effect on the thermodynamic equilibrium of a reaction (Kim et al., 2009; Li et al., 2010). Figure 2a, b shows that the considerable variation in the growth of L. brevis and L. plantarum and GABA production under different fermentation temperatures. The bacterial growth increased with the increase of temperature and peaked at 35°C, then decreased with the increase of temperature. For GABA production, a trend similar to the bacterial growth was observed. The effective synthesis of GABA reached 100 mM and 90.6 mM by L. brevis and L. plantarum, respectively was recorded at 35°C. Results illustrated in Fig. 2c show that co-culturing of both L. brevis and L. plantarum resulted in enhancement of GABA production reached 110.66 mM. These data indicated that appropriate temperature was beneficial to produce GABA and excessively high temperature was unfavorable to the GABA production (Tung et al., 2011). Results indicated that high efficient conversion glutamate to GABA needed not only high cell density but also appropriate temperature.
Time course study of extracellular GABA content: The time factor plays an important role in the fermentation and the production of GABA as temperature and pH do. L. brevis and L. plantarum required 72 h of fermentation to reach the highest production of GABA at 100 and 90.68 mM, respectively (Fig. 3a, b). In the same trend, Di Cagno et al. (2010) reported that L. plantarum DSM19463 required 72 h of fermentation to reach the highest production of GABA at 4.83 mM.
From the results depicts in Fig. 3c, it is worth mentioning that co-culturing of both L. brevis and L. plantarum was not only able to enhance GABA production reached 125.56 mM but also it produced it in a short time (48 h).
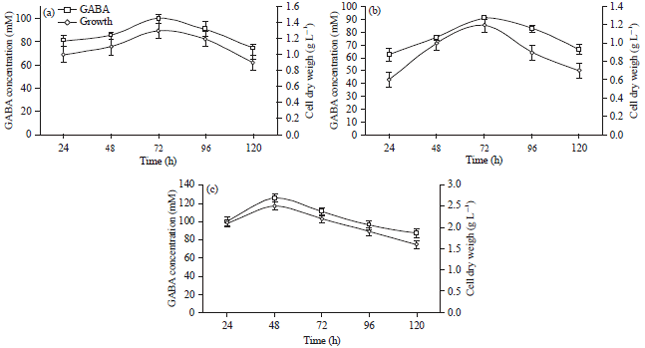 | |
| Fig. 3(a-c): | Time profiles of growth and extracellular production of GABA of (a) L. brevis, (b) L. plantarum and (c) Co-cultured of both L. brevis and L. plantarum, results are means of three independent determinations and error bars represent the standard deviation |
Effect of initial monosodium glutamate concentration: Sodium glutamate is known flavour enhancer in foods but its higher intake is associated with human body disorders like stroke, autism and Alzheimer’s disease because of its excitatory neurotransmitter effect in mammalian nervous system. Hence, it is very crucial to minimize the usage of glutamate in food industry for human consumption (Ali et al., 2009).
The content of produced GABA depends on various conditions such as the amount of precursor, an addition of salt, enzyme and the effect of pH (Mazur et al., 2011). As shown in Fig. 4a and b, the highest production of GABA by L. brevis and L. plantarum reached 110.89 and 99.12 mM, respectively were recorded in the presence of 750 M of monosodium glutamate (the precursor), however, when the concentration of monosodium glutamate exceeded 750 M, GABA production decreased. Co-culturing of both L. brevis and L. plantarum resulted in enhancement of GABA production reached 160.57 mM (Fig. 4c). So it can be deduced that the GABA production may be improved with increased concentration of MSG added to the culture medium and the MSG concentration above a certain level would inhibit cell growth.
In the same regard, GABA concentration reached 161 mM after cultivation in the medium containing 500 mM of glutamate by L. paracasei NFRI 7415 (Komatsuzaki et al., 2005). Lactobacillus brevis also increased GABA production by the addition of 6% glutamic acid (Binh et al., 2014). However, S. salivarius subsp. thermophilus Y2 did not increase GABA production significantly when glutamate was added 10-20 g L–1 of media, suggesting that these concentrations of glutamate are not appropriate for the synthesis of GABA in this species (Yang et al., 2008).
The addition time for the GABA substrate also affects the final GABA yield as well as the concentration of the substrate in the medium. A significant difference in GABA yield among various times of MSG addition was shown in Fig. 4a-c.
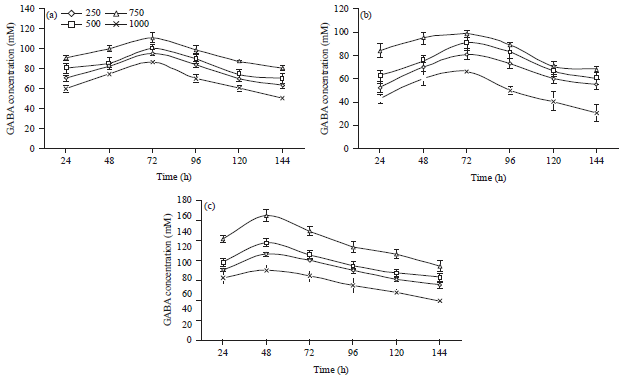 | |
| Fig. 4(a-c): | Effect of initial monosodium glutamate (MSG) concentration on extracellular production of GABA by (a) L. brevis, (b) L. plantarum and (c) Co-cultured of both L. brevis and L. plantarum. Results are means of three independent determinations and error bars represent the standard deviation |
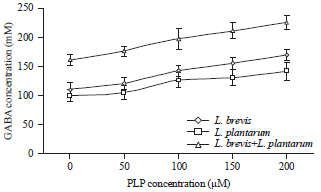 | |
| Fig. 5: | Effect of PLP addition on extracellular production of GABA by L. brevis, L. plantarum and co-cultured of both L. brevis and L. plantarum, Results are means of three independent determinations and error bars represent the standard deviation |
It is clear that individual strains produced the highest GABA at 72 h while co-culturing of both strains was at 48 h. These results indicate that the highest GABA production by microorganisms can depend on the addition of appropriate medium additives and optimum additional time for the additives.
Effect of PLP addition on extracellular GABA production: Glutamic acid decarboxylase (GAD, EC 4.1.1.15) catalyzes the irreversible α-decaboxylation of glutamate to produce GABA. The GAD uses PLP as coenzyme. So, it might be hypothesized that addition of PLP to the culture medium would improve GABA production. The effect of PLP addition was examined by analysis of extracellular GABA content in culture broth of L. brevis and L. plantarum with PLP at different concentrations. Figure 5 shows that the higher the concentration of PLP was the higher level of extracellular GABA the strain would produce. The highest concentration of GABA reached 168.58 and 140.69 mM produced by L. brevis and L. plantarum, respectively were recorded at 200 μM PLP. In the same line, Lin (2013) reported that L. rhamnosus YS9 in medium supplemented with 200 mM of monosodium glutamate and 200 μM of pyridoxal phosphate (PLP), produced 187 mM of GABA. These results differed from which reported by Li et al. (2010) that PLP did not affect the cell growth and gamma-aminobutyric acid production of L. brevis NCL912.
Co-culturing of both L. brevis and L. plantarum resulted in enhancement of GABA production reached 224.69 mM at 200 μM PLP. The results suggest that co-culturing of the two strains raises the each other’s properties of the acidification of the medium and production of GABA or acid resistance (Watanabe et al., 2011).
Lactobacillus brevis and L. plantarum can be potential starter cultures for GABA-enriched functional foods. Co-culturing of microbes is beneficial not only for food production but also for human health since a large number of microbes live together in the animal intestines, in other word they are co-culturing. The microbes may collaborate to carry out important biological functions; however, very little is understood. The mechanism of the co-culturing of different microbes needs to be explored and the findings can be extended to the industrial application as well as well as the improvement of human health.