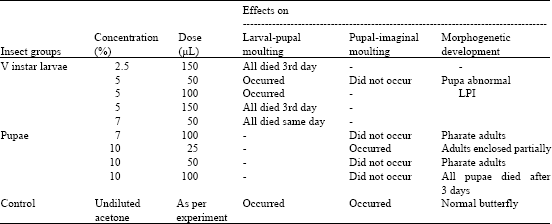
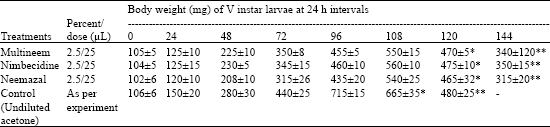
Research Article
Reduction in Haemocyte Mediated Immune Response in Danais chrysippus Following Treatment with Neem-Based Insecticides
Department of Animal Sciences, School of Life Sciences, University of Hyderabad, Hyderabad-5000 46, India
R.K. Tiwari
Department of Zoology, R.N. Govt. Postgraduate College, Gyanpur-221 304, S.R.N. Bhadohi, India
Dinesh Kumar
Department of Zoology, Banaras Hindu University, Varanasi-221, India