Research Article
Renal Damage Mediated by Oxidative Stress in Mice Treated with Aluminium Chloride: Protective Effects of Taurine
Department of Biological Sciences, Faculty of Science, King Faisal University, Al Hassa, Saudi Arabia
The impacts of aluminum intoxication on human health have been increasingly alarming in recent years. Nevertheless, human are frequently exposed to aluminium from various food additives, therapeutic treatments and the environment (Scancar and Milacic, 2006). Aluminium has no demonstrated essential function in mammals (Zhou et al., 2008). Aluminium accumulates in all tissues of mammals such as the kidneys, liver, heart, blood, bones and brain (Sanchez-Iglesias et al., 2007; Turner et al., 2008; Gonzalez et al., 2009). Accumulation of aluminium in biological systems has been linked to disease conditions such as dialysis encephalopathy, renal osteodystrophy, hypochromic microcytic anemia (Meshitsuka et al., 2001; Malluche, 2002; Zatta et al., 2002). A relationship between aluminium and Alzheimer’s disease has been suggested (Roberts et al., 1998; Abd El-Rahman, 2003; Matsuzaki et al., 2004; Exley, 2006; Aremu and Meshitsuka, 2005; Meshitsuka and Aremu, 2008). Aluminium intoxication has been reported to cause oxidative stress and a decrease in the intracellular levels of reduced glutathione (Gonzalez et al., 2007). The main route of aluminium excretion is urine (Bertholf et al., 1989; Sutherland and Greger, 1998; Taylor et al., 1998; Shirley et al., 2004; Stoehr et al., 2006). Different mechanisms of renal excretion of aluminium such as glomerular filtration (Yokel and McNamara, 1985), tubular reabsorption of filtered aluminium and secretion in distal nephron (Shirley et al., 2004; Shirley and Lote, 2005) and excretion in the distal tubules (Monteagudo et al., 1988) have been suggested. The kidneys play a major role in preventing the harmful accumulation of aluminium, excreting aluminium from the body. Diverse studies have been conducted on metabolism, excretion and the nephrotoxic effect of aluminium (Mahieu et al., 2000; Mahieu et al., 2003; Stoehr et al., 2006).
Taurine (2-aminoethanesulfonic acid) is the major intracellular free β-amino acid, which is normally present in most mammalian tissues. It has several functions in cell metabolism including osmoregulation, membrane stabilization, detoxification and regulation of cellular calcium homeostasis (Huxtable, 1992). Taurine has been demonstrated to function as a direct antioxidant by scavenging reactive oxygen radicals, inhibition of lipid peroxidation and as an indirect antioxidant by preventing changes in membrane permeability resulting from oxidant injury in many tissues including liver (Timbrell et al., 1995; Waters et al., 2001; Hagar, 2004). A number of investigators reported that taurine protects several organs in the body against toxicity and oxidative stress due to heavy metals and other toxins as well as drugs (Hwang et al., 1998; Dogru-Abbasoglu et al., 2001; Gurer et al., 2001; Hagar et al., 2006; Tabassum et al., 2006; Jagadeesan and Pillai, 2007; Manna et al., 2008, 2009; Parildar-Karpuzoglu et al., 2008). Therefore, the present study aimed to investigate whether taurine could offer any protection against aluminium chloride (AlCl3)-induced nephrotoxicity using mice as the animal model. AlCl3 was administered at a single dose of 25 mg Al3+ kg-1 b.w. intraperitoneally to male adult albino mice of Swiss strain. Renal damage was assessed by measuring serum creatinine and blood urea, in addition to lipid peroxidation and antioxidant status in renal tissues of mice exposed to AlCl3 alone or in presence of taurine. Finally, histological and ultrastructural studies were conducted in AlCl3-induced renal pathophysiology and evaluated the protection by taurine.
Animals: In this study male mice weighed 15-20 g were obtained from the animal house of Faculty of Veterinary Medicine, King Faisal University (Saudi Arabia). The animals were housed throughout the experiment in polypropylene cages (with each cage housing 5 animals) and allowed to acclimatize to the laboratory environment for 10 days. Animals were maintained under controlled conditions of temperature at 25±2°C, relative humidity of 50±15% and normal photoperiod (12-12 h light-dark cycle). The animals were allowed free access to standard dry pellet diet and water ad libitum. All mice were handles in accordance with the standard guide for care and use of laboratory animals.
Experimental design: The animals were randomly divided into four experimental groups:
| • | Group 1 (Control): Control mice received an injection of 0.5 mL of physiological solution per animal (vehicle) |
| • | Group 2 (AlCl3): Each animal received single intraperitoneal injection of 0.5 LD50 AlCl3 solution in physiological saline (25 mg Al3+ kg-1 b.wt.) |
| • | Group 3 (AlCl3+Taurine): Shortly after AlCl3 treatment, single dose of taurine (1 g kg-1 b.wt.) was intraperitoneally administrated to mice |
| • | Group 4 (Taurine): Single dose of taurine (1 g kg-1 b.wt.) was intraperitoneally applied to mice |
Dose selection of AlCl3 and taurine was based on published studies (Viezeliene et al., 2006; Bosgelmez et al., 2008). The substances were administered in the morning (between 09:00 and 10:00 h) to non-fasted animals. Mice from each group were sacrificed 24 h after administration of the compounds and blood samples were collected. Serum was separated into metal-free plastic tubes and the kidney tissues were quickly excised and washed immediately with ice-cold physiological saline (0.9% NaCl) and one part immediately stored at -80 °C until analysis. The other part of the tissue was taken from the animals for histological and electron microscopic analysis.
Tissue extraction: One gram of tissue was washed in cold isotonic saline containing 1 mM EDTA and then homogenized in 8 mL of cold buffer (50 mM potassium phosphate buffer, pH 7.5, containing 1 mM EDTA). After centrifugation at 10,000 rpm for 20 min at 4°C, the supernatant was removed and frozen at -80°C until assayed. For determination of malondialdehyde (MDA), a 500 mg sample of tissue was washed with cold isotonic saline and homogenized in 2.5 mL buffer (20 mM tris buffer, pH 7.4). Fifty microliters of 0.5 M butylated hydroxytoluene (BHT) in acetonitrile was added to prevent sample oxidation. After centrifugation at 6000 rpm at 4°C for 10 min, the supernatant was removed and frozen immediately at -80°C until assayed.
Biochemical assays: The reduced glutathione (GSH) level was measured from the reduction of 5,5'-dithiobis (2-nitrobenzoic acid) (DTNB). The chromogen formed is directly proportional to the GSH concentration (Beutler et al., 1963). Lipid peroxidation (LPO) was measured by thiobarbituric acid (TBA) method (Esterbauer et al., 1991) tha t determines aldehyde formed by degradation of hydroperoxide, including malondialdehyde (MDA). Cytosolic glutathione peroxidase (selenium-dependent) (GPx) activity was determined from the azide insensitive rate of oxidation of NADPH in the presence of hydrogen peroxide, glutathione and glutathione reductase (Flohe and Gunzler, 1984). Glutathione reductase (GR) activity was measured from the rate of reduction of oxidized glutathione by NADPH (Smith et al., 1988). Catalase (CAT) activity was measured from the rate of dismutation of hydrogen peroxide (H2O2) to water and molecular oxygen in a two-step coupling reaction (Aebi, 1984). Superoxide dismutase (SOD) activity was measured in a coupling reaction through formation of formazan dye from tetrazolium salt by superoxide radicals generated by xanthine oxidase and hypoxanthine (Malstrom et al., 1975). The serum creatinine level was determined from the red-complex formed through reaction with basic picrate solution (Murray, 1984). The urea was hydrolysed into ammonia and CO2 and the indophenol formed from reaction of ammonia with hypochlorite and salicylate was measured (Kaplan, 1984). Total protein content was determined according to Lowry et al. (1951).
Histological study: After fixation in Bouin’s fluid for 48 h, the kidneys were dehydrated in graded ethanol and blocks were made in paraffin. Sections of 4 μm thickness were stained with haematoxylin and eosin (H and E) and examined under Olympus CX41 light microscope.
Ultrastructural study: For electron microscopic examinations of kidney tissues, primary fixation was done in 3% glutaraldehyde in sodium phosphate buffer (200 mM, pH 7.2) for 3 h at 4°C. Kidney tissues were washed with the same buffer and postfixed in 1% osmium tetroxide (Agar Sci. Ltd.) in sodium phosphate buffer, pH 7.2, for 1 h at 4°C. Tissue samples were washed with the same buffer for 3 h at 4°C and then embedded in Araldite (Agar Sci. Ltd.). Thin sections were cut with LKB ultramicrotome. Samples were stained with 2% uranyl acetate and lead citrate. The sections were viewed and photographed on a Jeol 100 CX transmission electron microscope (Jeol Ltd., Japan) at 80 kV.
Statistical analyses: Data were analyzed using SPSS 17.0 for windows. Significance was calculated using one-way analyses of variance (ANOVA) followed by Least Significance Difference (LSD) for multiple comparisons. p<0.05 was considered statistically significant.
Biochemical results: Data relating to lipid peroxidation and antioxidant status in the kidney are stated in Table 1. MDA content, an end product of lipid peroxidation, in the mice treated with AlCl3 was significantly increased 7.5-fold in kidney (p<0.05) when compared to the vehicle control group. Post-treatment with taurine resulted in a significant decrease in the content of renal (p<0.05) MDA when compared to the AlCl3 group. Conversely, the level of GSH in the mice treated with AlCl3 was significantly decreased in kidney (1.4-fold, p<0.05) when compared to the vehicle control group. Results showed that post-treatment with taurine was able to prevent the AlCl3 induced decreased renal GSH content. Treatment of mice with taurine alone did not affect the levels of MDA and GSH when compared to the vehicle control group.
As presented in Table 1, the activity of GPx and GR in the AlCl3 group was significantly decreased in kidney (2.6-, 1.7- fold, respectively, p<0.05). When compared to the vehicle control group. However, post-treatment with taurine resulted in a significant increase in the activity of GPx in kidney (p<0.05) in comparison with that of the AlCl3 group, indicating that taurine might play protective effect on aluminum toxicity in mice. Furthermore, post-treatment with taurine was more effective in restoring the activity of GR in kidney to control values. On the other hand, the activities of CAT and SOD in the AlCl3 group were non-significantly changed in kidney (p>0.05) when compared to the vehicle control group. It appeared that treatment of taurine after AlCl3 showed no change in the activity of CAT and SOD in kidney (p>0.05) when compared to the AlCl3 group. In groups treated only with taurine, antioxidant enzymes were notably elevated when compared to the vehicle control group.
On the other hand, serum urea and creatinine were increased in mice receiving single dose of AlCl3 (4.5- and 1.9-fold, respectively, p<0.05) as compared to the vehicle control group (Table 2). Taurine administration shortly after AlCl3 further attenuated changes in the level of serum markers of renal damage.
Histopathological findings: Photomicrographs of kidney sections from various treatment groups are shown in Fig. 1A-E. In histopathological examination, normal architecture was observed in control animals whereas the administration of AlCl3 caused significant morphological damage to the kidneys, especially to the renal cortex. In AlCl3-treated kidneys, the affected glomeruli were observed exhibiting different forms of degeneration. Some glomeruli showed prominent dilatation of Bowman’s space with glomerular atrophy (Fig. 1B), whereas a small number of others exhibited congestion in the capillary loops with an adhesion between visceral and parietal layers of Bowman’s capsule (Fig. 1C).
| Table 1: | Effect of aluminum chloride and taurine on parameters indicative of oxidative stress in kidneys |
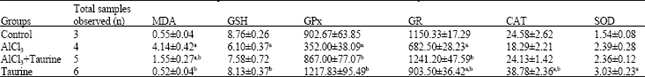 | |
| Treated mice received single dose of AlCl3 (25 mg Al3+ kg-1 b.wt, i.p.) and/or taurine (1 g kg-1 b.wt, i.p.), MDA: Malondialdehyde (μmol mg-1 protein), GPx: Glutathione peroxidase (nmol min-1 mg-1 protein), GR: Glutathione reductase (nmol min-1 mg-1 protein), GSH: Reduced glutathione (μmol mg-1 protein), SOD: Superoxide dismutase (mU mg-1 protein), CAT: Catalase (U mg-1 protein), each value represents the mean±SE, ap<0.05 compared with control group, bp<0.05 compared with AlCl3 group (LSD-multiple comparison test) | |
| Table 2: | Effect of aluminum chloride and taurine on parameters indicative of kidney function in serum of mice |
 | |
| Treated mice received single dose of AlCl3 (25 mg Al3+ kg-1 b.wt., i.p.) and/or taurine (1 g kg-1 b.wt., i.p.), each value represents the mean±SE, ap<0.05 compared with control group, bp<0.05 compared with Al group (LSD-multiple comparison test) | |
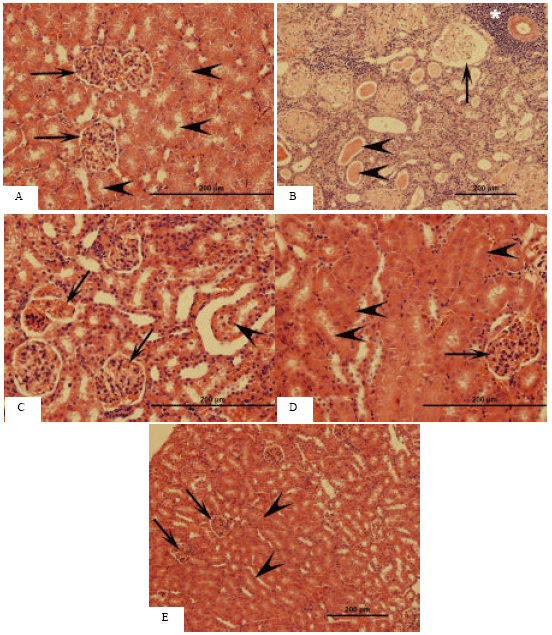 | |
| Fig. 1: | Light microscopic images of mice kidney. HE stain. (A) Kidney sections of control mice. Bowman’s space (arrows), tubular lumen (arrowheads). (B) Kidney sections of mice treated with AlCl3. Expanded Bowman’s space (arrow), intratubular hyaline casts (arrowheads) and perivascular inflammation (*) can be noted. (C) Kidney sections of mice treated with AlCl3. Congestion of glomerular capillaries (arrows) and sloughing of tubular epithelium (arrowhead) are clearly discernable. (D) Kidney sections of mice treated with AlCl3 plus taurine. Glomeruli (arrow) and renal tubules (arrowheads) appear to be restored as compared to B and C. (E) Kidney sections of mice treated with taurine alone. No histopathological changes can be observed in glomeruli (arrows) and tubules (arrowheads) |
The renal tubules showed cytoplasmic vacuolization, intraluminal exfoliation with granular cast formation as well as pyknosis of the nuclei (Fig. 1B, C). Tubular damage was commonly accompanied by mild peritubular and perivascular infiltration of inflammatory cells (Fig. 1B). These findings in the renal cortices of AlCl3-treated mice were spread throughout the subcortical areas as well. In the AlCl3+ taurine group, these pathological changes were alleviated (Fig. 1D). In taurine administration group, histological appearances of the glomeruli and tubules were normal (Fig. 1E).
Ultrastructural observations: In the examination of samples obtained from the kidneys of mice belonging to the control group, endothelial cells in the glomerular capillaries, basement membrane, slit membrane formed by podocytes on it and a mesangial cellular structure having indented nucleus were observed (Fig. 2A, B). The glomeruli following AlCl3 injection showed a morphological impairment characterized by focal nodular thickenings of glomerular capillary basal lamina, increase of the content of mesangial matrix and narrowing of the capillary lumen (Fig. 2C).
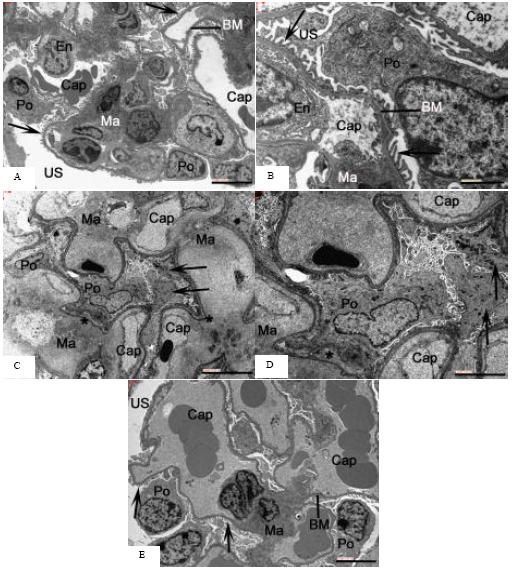 | |
| Fig. 2: | Electron microscopic images of mice kidney. (A-B) Kidney of control mice. Glomerular capillaries (cap) show porous endothelium (En). Regularly arranged podocyte processes (Po) with slit pores (arrows) are separated from the endothelium by a thick basement membrane (BM). The BM is continuous with the mesangial matrix (Ma). US: urinary space. Scale bars: (A) 5 μm and (B) 2 μm. (C) Kidney of mice treated with AlCl3; (D) Higher magnification. Note expansion of mesangial matrix (Ma) and constriction of glomerular capillaries (cap). Some podocyte processes appear broadened and contain electron dense deposits (arrows); Po: podocyte. Glomerular endothelium contains gaps instead of regular pores. The glomerular BM appears with a local thickening (*). Scale bars: (C)10 μm, (D) 5 μm. (E) Kidney of mice treated with AlCl3 plus taurine. The porous endothelium and podocytes appear better preserved than in C and D. Po: podocyte, arrows indicate foot processes, Cap: glomerular capillaries, BM: basement membrane, Ma: mesangial matrix, US: urinary space. Scale bar: 5 μm |
The endothelium showed local irregularities in profile and pore arrangement. Some podocyte processes appeared broadened or showed degenerative features such as accumulation of electron dense deposits. Foot processes were occasionally lost leaving fragmented parts in their vicinity and frequently exhibited focal adhesion forming blunted sheath of the cytoplasm (Fig. 2C, D). In contrast, this effect was appreciably less severe in glomeruli from the taurine group, which had a normal filtration barrier without accumulation of electron dense material (electron microscopy, Fig. 2E). Podocytes showed a normal
structure and an orderly arrangement and resembled those of control mice. Foot processes seemed to be thinner and longer. The capillary lumen was patent. There was no apparent endothelial cell injury in the glomerular structures. Moreover, there was marked reduction of mesangial area and the basement membrane thickening as compared to aluminium treated mice. In taurine alone group, the glomerular endothelium and podocytes showed normal structure.
As for the investigations made in the tubular area of the cortex of control mice, the Proximal Convoluted Tubules (PCT) lined with cells characterized with their microvilli and well-developed basal foldings were observed (Fig. 3A). Alterations in proximal tubular cells were observed in most kidney samples from AlCl3 treated mice (Fig. 3B, C).
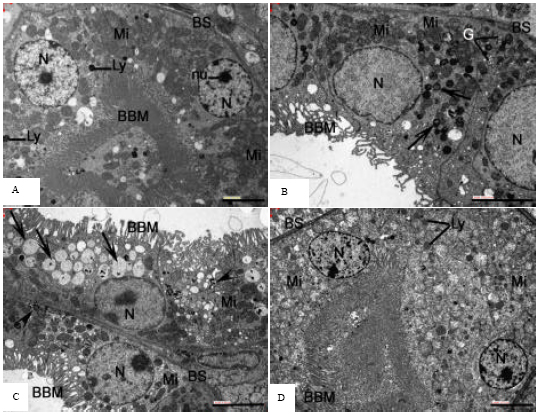 | |
| Fig. 3: | Representative electron microscopy images of proximal tubular epithelium of mice kidney. (A) Kidney of control mice. Note the normal-sized mitochondria (Mi) with intact cristae and homogeneous matrix. Nucleus (N) is of normal size and has distinct nucleolus (nu) and evenly distributed chromatin. Brush border microvilli (BBM) are present on the apical cytoplasmic surface. BS: basolateral membrane, Ly: lysosomes. (B) Kidney of mice treated with AlCl3. Note degenerated BBM and markedly swollen mitochondria (Mi) which have disoriented and partially deteriorated cristae. BS: basolateral membrane, N: nucleus, G: Golgi apparatus, arrows indicate myelin figures. (C) Kidney of mice treated with AlCl3. Markedly dilated endoplasmic reticulum (ER) in the cytoplasm of a convoluted tubular cell can be seen (arrows). The dilated ER contains fine granular material and it obviously compresses the cell nucleus (N). Brush border microvilli (BBM) are greatly distorted. BS: basolateral membrane, Mi: mitochondria, arrowheads indicate myelin figures. (D) Kidney of mice treated with AlCl3 plus taurine. The degenerative changes in epithelial cells of proximal convoluted tubule (PCT) are minimal as compared to B and C. BBM: brush border microvilli, BS: basolateral membrane, N: nucleus, Mi: mitochondria, Ly: lysosomal elements. Scale bars: 5 μm (A, B, C and D) |
Cytoplasmic vacuolation, apical blebbing and attenuation or loss of the microvillar brush border were general findings. The mitochondrial ultrastructural abnormalities included a marked swelling of the mitochondria in the majority of the convoluted tubular cells. In the swollen mitochondria, cristae were disoriented, marginated and short in comparison with the control well-preserved parallel linear ones. Noticeable reduction of the matrical density and indistinction of the inner membranes were obvious in the swollen mitochondria. In many of the affected mitochondria, there was a loss of cristae structures due to cristolysis and only matrical flocculent densities were detected. The affected renal tubular cells showed dilatation of the endoplasmic reticulum. The dilated rough endoplasmic reticulum (RER) lost the characteristic parallel arrangement of their cisternae and disclosed partial detachment of the bound polyribosomes and accordingly had degranulated appearance. Occasional marked dilatation of Endoplasmic Reticulum (ER) was discerned and the dilated ER was filled with fine granular material (Fig. 3C). Also, some tubular cells contained hypertrophied Golgi bodies and an increasing number of myelinated figures. Basolateral processes were dilated and in some instances were absent. These changes were less expressed in Al+taurine treated mice (Fig. 3D). Administration of Taurine to the AlCl3 treated animals prevented the ultrastructural alterations such as atrophied brush border microvilli, increased size of mitochondria, dilatation of ER and formation of myelin figures observed in PCT epithelial cells of AlCl3 treated mice. In kidney of mice treated with taurine alone, PCT epithelium was comparable to control.
Aluminium-induced damage to body organs has already been reported in several studies (Parkinson et al., 1981; Wills and Savory, 1983; Exley et al., 1997) and accumulation in the kidney has been related to worsening renal function (Nesse et al., 1997). Indeed the kidney may be exposed to high concentrations of aluminium during the normal process of excretion and is therefore a site for aluminium mediated toxicity (Bellia et al., 1994). However, the mechanism of this effect was unclear and the question was posed if ROS play any part in this process.
The antioxidant enzymes play important protective roles in the kidney (Ichikawa et al., 1994). Primarily because of its transport functions, the kidney has a very active oxidative metabolism that results in the production of ROS. The antioxidant enzymes GSH-Px and CAT are responsible for the protection against ROS. Oxidant injury is now recognized as playing a key role in the induction of experimental renal diseases (Ichikawa et al., 1994; Haugen and Nath, 1999).
The present study was carried out to investigate the protective effects of taurine on aluminium-induced oxidative stress and biochemical alterations in kidney of mice. In the AlCl3 treated mice, the levels of thiobarbituric acid reactive substances (TBARS) were found to be elevated but the activities of GPx and GR were significantly decreased in renal tissues. These observations are similar to the data reported by Yousef (2004), Yousef et al. (2005, 2007) and Nehru and Anand (2005) who indicated that aluminium intake produces oxidative stress. Although aluminium is not a transition metal and therefore cannot initiate peroxidation, many investigations have searched for a correlation between aluminium accumulation and oxidative damage in the body tissues (Cherroret et al., 1995; Wilhelm et al., 1996; Nehru and Anand, 2005). An in vitro study has indicated that aluminium greatly accelerates iron-mediated lipid peroxidation (Xie and Yokel, 1996). In fact, aluminium, a non-redox-active metal, is a pro-oxidant both in vivo and in vitro (Exley, 2004).
The primary effects of aluminium on the brain, liver and kidney functions are thought to be mediated via damage to cell membranes. LPO of biological membranes results in the loss of membrane fluidity, changes in membrane potential, an increase in membrane permeability and alterations in receptor functions (Nehru and Anand, 2005). In the present experiment, there was a significant increase in LPO after AlCl3 exposure, measured in terms of TBARS levels in the kidney. Nehru and Anand (2005) also reported a significant increase in brain TBARS after stimulation by aluminium salts. The ionic radii of Al3+ most closely resemble those of Fe3+, therefore the appearance of Al3+ in Fe3+ sites is probable. Aluminium is known to be bound by the Fe3+ carrying protein transferrin thus reducing the binding of Fe2+. The increase in free intracellular Fe2+ causes the peroxidation of membrane lipids and thus causes membrane damage (Nehru and Anand, 2005).
Orihuela et al. (2005) reported that at high doses, aluminium was able to induce oxidative stress in the intestinal mucosa, as indicated by the significant increase in the concentration of both, oxidized glutathione/reduced glutathione (GSSG/GSH) ratio and TBARS levels. Aluminium might affect the GSH synthesis by decreasing the activity of Glutathione-Synthase (GS), a non-limiting step of whole reaction, thus leading to a reduced GSH content. On the other hand, it has been demonstrated that aluminium is able to inhibit NADPH-generating enzymes such as glucose 6-phosphate dehydrogenase and NADP-isocitrate dehydrogenase. Since, NADPH is shown to be a main factor for the GSH regeneration, the decreased GSH level could be also ascribed to insufficient supply of NADPH. Besides, aluminium was able to diminish the activity of enzymes related to cell antioxidant defense, such as glutathione reductase, catalase and GSH-peroxidase in the kidney. Therefore, aluminium could indirectly contribute to unbalance redox equilibrium in the renal tissue. On the other hand, taurine may ameliorate aluminum induced renal injury by enhancing the activities of endogenous antioxidants. Support for this concept comes from the present results, which show that taurine produced a remarkable significant increase in renal GPx and GR activities. This could be attributed to the role of taurine in maintaining a normal IGF-I level (Dawson et al., 1999) and its antioxidant action against lipid peroxidation, thus conserving the internal antioxidants system. The stimulatory effect of taurine on endogenous antioxidants was also reported by others (Erdem et al., 2000).
The elevation (p<0.05) in serum urea and creatinine levels reported in the present study in AlCl3-treated mice is considered as a significant marker of renal dysfunction and this is supported by the finding of Mahieu et al. (2005), who reported that alterations in serum urea may be related to metabolic disturbances (e.g., renal function and cation-anion balance). In addition, Katyal et al. (1997) reported that aluminium has been implicated in the pathogenesis of several clinical disorders, such as renal dysfunction. On the other hand, Newairy et al. (2009) showed that the increase in urea concentrations in plasma of animals treated with 34 mg AlCl3 kg-1 b.wt. for 70 days may be due to its effect on liver function, as urea is the end-product of protein catabolism and this is confirmed by the decrease in plasma proteins and/or referred to liver dysfunction as proven by the increase in activities of plasma transaminases and lactate dehydrogenase.
In the current study, acute exposure to AlCl3 induced pathological changes in mice glomeruli as indicated by failure to trace the fenestrate belonging to endothelial cells of the glomerlular capillaries, focal thickening of glomerular basement membrane and deterioration of the slit membrane structures formed by the podocytes. These morphological changes may be in part resulted in the interference with glomerular hemodynamic. The present findings also showed that Alcl3 induced clear cell alterations in the mice kidney proximal epithelia including degeneration of apical microvilli, mitochondrial swelling, dilatation of ER and loss of basal infoldings. Similar effects of aluminium treatment have been described in vivo (Kutlubay et al., 2007) and in vitro (Sargazi et al., 2001). In addition, similar changes have also been noted in kidneys of experimental animals upon exposure to other trace metals such as lead (Jarrar, 2001), cadmium (Abdel-Moneim and Said, 2007) and mercury (Abdel-Moneim, 2009).
Nephroprotective action of taurine deserves special consideration. According to the present data, its administration to aluminium treated mice effectively improves renal function, as concluded from (1) decreased serum urea and creatinine concentrations (Table 2), (2) attenuated histological and ultrastructural changes characteristic of aluminium nephrotoxicity such as mesangial matrix expansion, compression of capillaries in glomeruli and foot process effacement (Fig. 2E). Other authors have also reported nephroprotective effects of this compound under diabetic conditions, attributing them mainly to attenuation of oxidative stress (Ha et al., 1999; Mozaffari et al., 2003). In agreement with the present data, other studies have shown that taurine exert morphological and functional protection against acute renal damage induced by gentamicin (Erdem et al., 2000) and acetaminophen (Das et al., 2010). Also, a renoprotective effect by the taurine was reported in a study examining methiocarb-induced oxidative damage in rat model (Ozden et al., 2009).
Taurine have been demonstrated to enhance renal kallikrein synthesis and consequently, increases the availability of the vasodilatory kinins (Nandhini et al., 2004). Bradykinin is shown to exert its effect on renal O2 consumption through dependence on nitric oxide synthetase (NOS) activity (Adler et al., 2004). Nitric Oxide (NO), a potent vasodilator is an important regulator of renal hemodynamics and that produced by endothelial isoenzyme (eNOS) is the prime regulator (Adler et al., 2004). In addition, taurine as an antioxidant is expected to increase the availability of NO, since oxidative stress depletes NO via scavenging via superoxides (Adler et al., 2004). This possibility leads to the increase in Renal Blood Flow (RBF) and Glomerular Filtration Rate (GFR), thus enhancing urinary excretion of urea and creatinine. Also, the activation of renal kallikrein-kinin system by taurine is reported to attenuate gentamycin-induced proximal tubular damage with the consequent reduction of brush border loss and protein cast formation (Nandhini et al., 2004; Bledsoe et al., 2006). Taurine also prevents the formation of myeloid bodies. Again, this could be related to the activation of renal kallikrein-kinin system, since bradykinin is shown to enhance the activity of phospholipase A2 in endothelial cells, leading to increase the metabolism of phospholipids (Higaki et al., 1999). Taurine by interaction of the neutral phospholipids of biological membranes (Abeywardena et al., 1984) can protect the membrane organization (e.g., membrane bound enzymes) against free radical attack. Furthermore, the renoprotective effect of taurine against the degenerative effects of aluminium could be achieved via other mechanisms, including calcium modulation, osmoregulation and preservation of membrane integrity (Erdem et al., 2000).
Accumulation of aluminum is the net consequence of uptake, biotransformation and elimination processes within an individual. Once aluminum is absorbed, it may be transformed into aluminum-thionein or metallothionein. Although the half-life of aluminumthionein in the liver and kidney is not exactly known, it is many years (Goyer, 1996). With continued retention, there is progressive accumulation in these tissues. The accumulated amount of aluminum in the tissue has been reported to be effectively reduced by taurine (Yeh et al., 2009). Taurine is a special amino acid, which possesses an amino group and a sulfonate group. These functional groups might bind with heavy metals and then stimulated the excretion of such compounds. Yeh et al. (2009) found that the amount of aluminum in the feces of rats fed with taurine added was slightly increased. There is no evidence that taurine directly reduces the production of free radicals but it may well operate by binding aluminum which is then not absorbed or is more rapidly excreted. In other words, it may act by reducing the overall bioavailability of aluminum or the intracellular availability of absorbed aluminum. Hence, administration of taurine may play a role to reduce the toxic effect of aluminum in the kidney.
Biochemical evaluations, histopathological and ultrastructural changes in this study showed that AlCl3 affects kidney in mice even if it is under the LD50 dose level. Data collected lead to the suggestions that the renoprotective effects of taurine could be partly attributed to its property causing an enhancement of the antioxidant defense system and to an increased release of NO by endothelial cells.