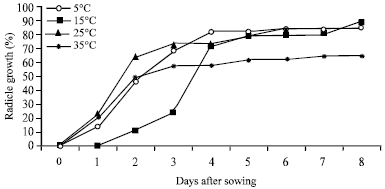
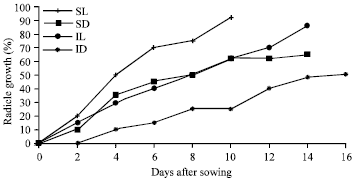
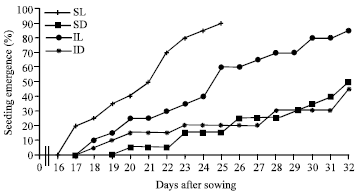
Research Article
Factors Affecting Regeneration Potential of Quercus semecarpifolia, Smith: A Poor Regenerated Oak of Himalayan Timberline
Department of Botany and Microbiology, School of Life Sciences, HNBG Central University, Srinagar (G), Uttarakhand, 246-174, India
Vinay Prakash
Department of Seed Science and Technology, School of Agriculture and Allied Sciences, Chauras Campus, HNBG Central University, Srinagar (G), Uttarakhand, 246-174, India
A.R. Nautiyal
High Altitude Plant Physiology Research Centre, School of Agriculture and Allied Sciences, HNBG Central University, Srinagar (G), Uttarakhand, 246-174, India