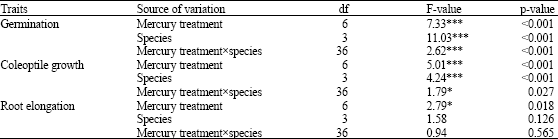
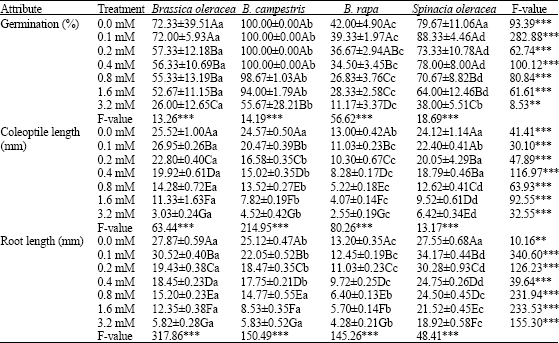
Research Article
Effect of Mercury to Seed Germination, Coleoptile Growth and Root Elongation of Four Vegetables
School of Environmental and Municipal Engineering, Institute of Environmental Ecology, Lanzhou Jiaotong University, Lanzhou 730070, Peoples� Republic of China
Y. Fangke
School of Environmental and Municipal Engineering, Institute of Environmental Ecology, Lanzhou Jiaotong University, Lanzhou 730070, Peoples� Republic of China
R. Jun
Demonstration Center for Experimental, Teaching of Environmental Science and Technology in Colleges of Gansu Province, Lanzhou 730070, Peoples� Republic of China