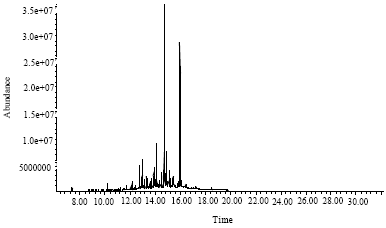
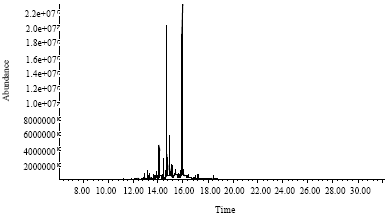
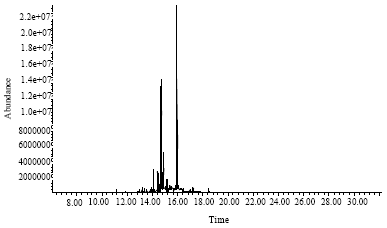
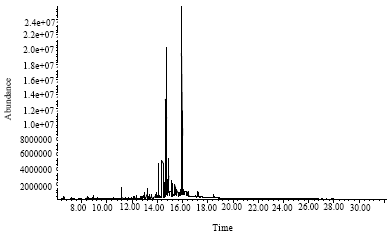
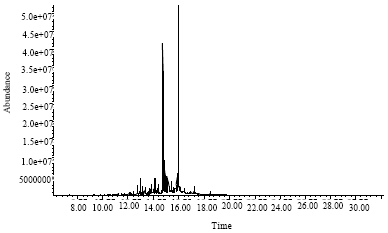
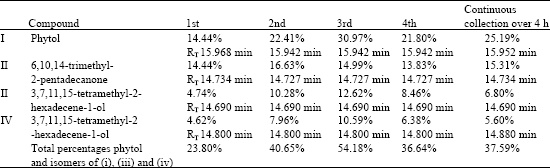
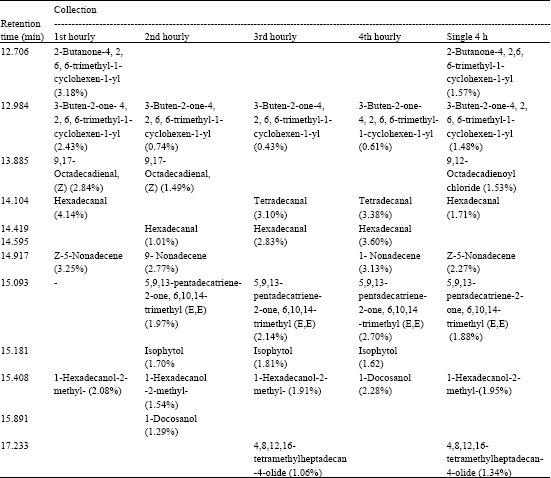
Research Article
Analysis of Essential Oil Constituents in Hydro-Distillates of Calotropis procera (Ait.) R.Br
Department of Chemistry, University of Lagos, Akoka, Lagos, Nigeria
M. Ogunlesi
Department of Chemistry, University of Lagos, Akoka, Lagos, Nigeria
E. Ofor
Department of Chemistry, University of Lagos, Akoka, Lagos, Nigeria
E.A.S. Osibote
Department of Chemistry, State University of New York (SUNY), Binghamton, USA