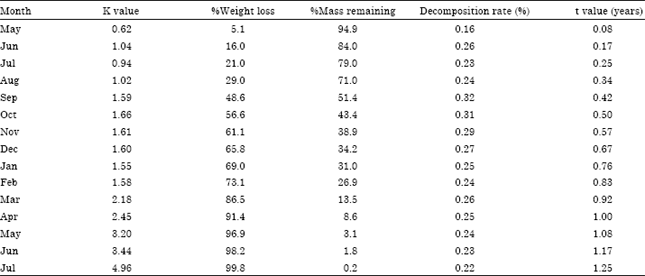
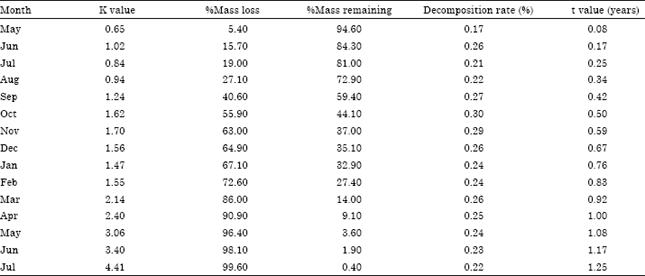
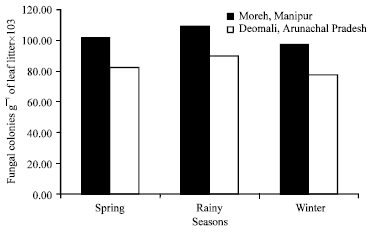
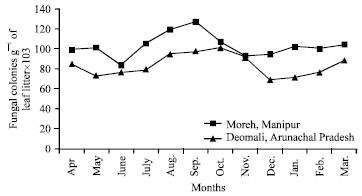
Research Article
Leaf Litter Decomposition Pattern in Dipterocarpus tuberculatus and Dipterocarpus retusus Forests of North East India
Rain Forest Research Institute, P.B.-136, Jorhat, Assam, India
Ashwani Tapwal
Rain Forest Research Institute, P.B.-136, Jorhat, Assam, India
Debu Moni Baruah
Rain Forest Research Institute, P.B.-136, Jorhat, Assam, India