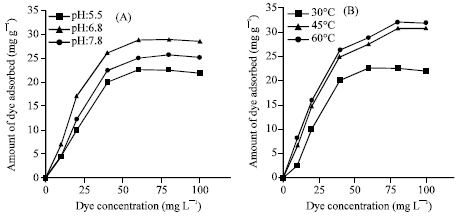
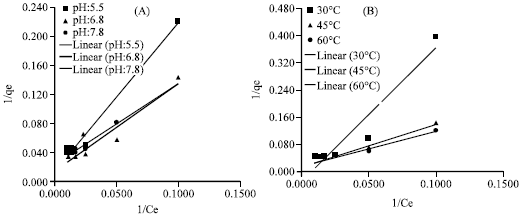
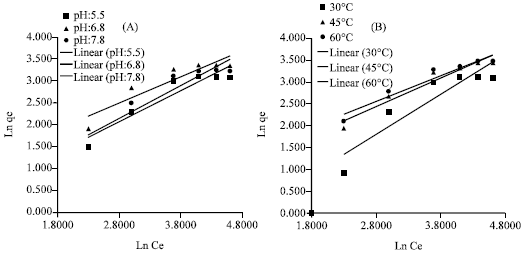
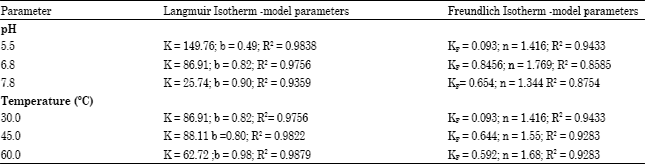
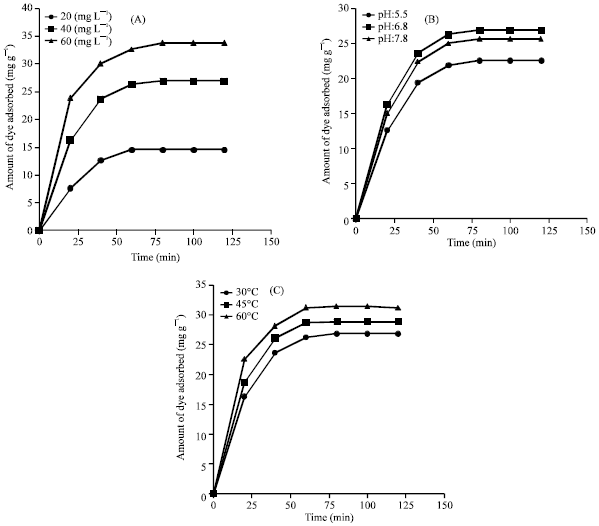
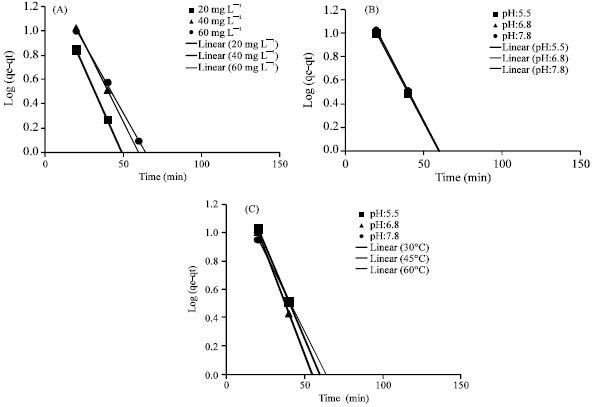
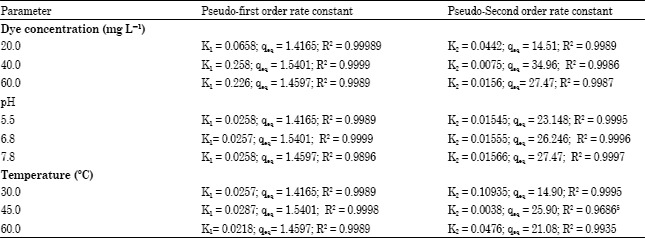
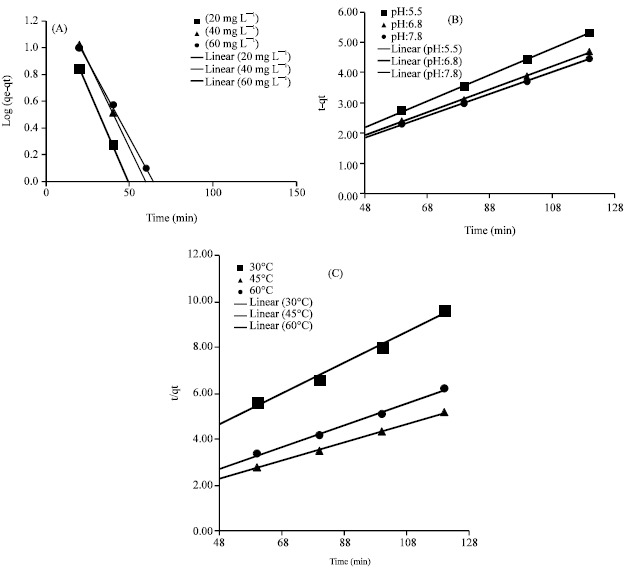
Research Article
Nano-porous Adsorbent from Fruit Peel Waste for Decolorization Studies
Environmental Nanotechnology Division, Sri Paramakalyani Centre for Environmental Sciences, Manonmaniam Sundaranar University, Alwarkurichi - 627 412, Tamil Nadu, India
G. Annadurai
Environmental Nanotechnology Division, Sri Paramakalyani Centre for Environmental Sciences, Manonmaniam Sundaranar University, Alwarkurichi - 627 412, Tamil Nadu, India