ABSTRACT
Removal of trivalent chromium from tannery effluent by microbial biomass generated from fermentation industry was carried out. Adsorption studies were conducted in batch mode at different adsorbent dosage, pH, initial concentration and contact time. The 94.6 and 64.48% of Cr (III) removal from aqueous solution and tannery effluent was observed at pH 4 by 1% adsorbent dosage, respectively. A comparison of kinetic models and correlation coefficient (R2 = 0.998) indicated that Ho and Mckay kinetic model correlates well with the experimental kinetic data. The surface properties of the adsorbent before and after chromium sorption were characterized by X-ray diffraction and infrared technique. It was concluded that Cr (III) is significantly removed by Aspergillus biomass and can be used economically for the removal of Cr (III) from aqueous solution and tannery effluent.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/rjes.2010.1.12
URL: https://scialert.net/abstract/?doi=rjes.2010.1.12
INTRODUCTION
Trivalent chromium is an important pollutant introduced into natural waters by a variety of industrial waste water including those from the textile, leather tanning, electroplating and metal finishing industries (Gaballah and Kilbertus, 1998). Tannery industry is one of the oldest and fastest growing industries in India and there are about 2,161 tanneries excluding cottage industries, which discharge annual of 9,420,000 m3 wastewater into the environment.
Tannery industries use a great number of chemical agents and produce an enormous volume of residual waters and solid residues (Mohan et al., 2006). The effluent from the chromium stream consists primary of a saline solution of spent chromium and residual fungicide at a pH between 3 and 5 (Tobin and Roux, 1998). In the world, the most usual technology for the treatment of the liquid residues generated by the leather industries constitutes of physico-chemical conventional treatment methods such as ion exchange (Tiravanti et al., 1997; Rengaraj et al., 2003; Petruzzelli et al., 1995), reduction (Seaman et al., 1999), chemical precipitation (Zhou et al., 1993). Membrane (Shaalan et al., 2001; Kozlowski and Walkowiak, 2002), electrochemical precipitation (Kongsricharoern and Polprasert, 1996), photocatalytic reduction (Testa et al., 2004), adsorption (Srivastava et al., 1997; Gupta et al., 1999) and biosorption (Gupta et al., 2001; Arvindhan et al., 2004).
The precipitation, oxidation/reduction, lime neutralization have traditionally been the most commonly used. Although, these technologies are quite satisfactory in terms of purging chromium from water, they produce solid sludge containing toxic compounds. Therefore, the need exists for a simple and robust treatment strategy that addresses local resources and constraints. Biosorption may be an effective and versatile method for removing chromium, particularly when combined with appropriate regeneration steps. This solves the problems of sludge disposal and renders the system more economically viable, especially because low cost adsorbents are abundant in nature (Ahluwalia and Goyal, 2007). In this study, microbial waste biomass of Aspergillus sp. (MB1) obtained from fermentation industry was used for removal of Cr (III) from aqueous solution and tannery effluent. Adsorption kinetics was studied at varying adsorbent dosage, pH and initial Cr (III) concentrations.
MATERIALS AND METHODS
The chromium removal study was carried out in the Department of Biotechnology and Environmental studies, Thapar University, Patiala (Punjab) from January to July 2008.
Collection and Characterization of Microbial Biomass and Tannery Effluent
Microbial waste biomass of Aspergillus sp. (MB1), which is generated as a byproduct of pharmaceutical fermentation industry for the production of certain antibiotics was collected from Ranbaxy (fermentation industry) Paonta Sahib, Himachal Pradesh, India. This biomass was characterized for physical and chemical parameter such as CHN analysis, pH, moisture, ash content, bulk density and calorific value as per the procedure (MacDonald et al., 1996).
Tannery effluent was collected from A.V. Tanneries, Kapurthala, Punjab, India. Characterization of effluent was performed as per procedure described elsewhere (Clesceri et al., 1998; APHA, 1998).
Surface Characterization of Microbial Biomass
X-Ray Diffraction (XRD) pattern and Fourier Transform Infra-Red (FTIR) spectroscopy studies were carried on microbial biomass to determine the adsorption mechanism. A structural change of biomass before and after reacting with Cr (III) was determined by using an X-ray diffractometer (Philips PW 1830). The FTIR spectrum of native and chromium treated biomass was obtained after drying the biomass at 70°C. The finely powdered samples were encapsulated with potassium bromide to prepare translucent sample disks and spectra were recorded by fourier transform infrared spectroscope (Hartmann and Braun, BOMEM Canada).
Adsorbent Preparation
Microbial biomass was oven dried at 80°C for two days and ground in a blender and sieved to pass through a 2.0 mm sieve in order to obtain uniform particle size (2 mm) and washed several times with double distilled water and dried before being used as a sorbent for removal of Cr (III) from aqueous solution and tannery effluent.
Preparation of Aqueous Chromium Solution
Aqueous solution of chromium was prepared by dissolving 0.38 g of Cr2O12S3 in 100 mL distilled water to obtain a concentration of 1000 mg L-1. Stock solution and tannery effluent were diluted with distilled water to obtained working solution in the concentration range of 5 to 50 mg L-1 and pH 2 to 5 of the solution was adjusted with 1N NaOH and 1N HCl.
Batch Adsorption Studies
Batch experiments were conducted with microbial waste biomass (MB1) in Erlenmeyer flask (250 mL) using different biomass dosage (0.25-2% of biomass) and aqueous solution (100 mL) of Cr (III) (25 mg L-1). The suspension was shaken at 120 rpm, 28°C for 24 h. Similarly, experiments were conducted at different pH (2 to 5) and chromium concentration (5-50 mg L-1). The residual concentration of chromium in aqueous solution was measured by (AAS) atomic absorption spectrophotometer (GBC 932AA, Australia) with an air-acetylene flame using Cr-hallow cathode lamp having sensitivity limit of 0.05 μg mL-1. Working standard solution of chromium was prepared from stock (1000 mg L-1) procured from Acros Organic Ltd., New Jersey, USA. Triplicates of each sample were analyzed. The mean value and relative standard deviation as given by AAS were recorded.
Chromium uptake (qe) was calculated as follows:
| (1) |
where, Ci and Cf are the initial and final Cr (III) concentrations in mg L-1, respectively, V is the volume of the Cr (III) solution in ml, m is weight of the biomass in g.
The percentage of removal of Cr (III) ions (R %) in aqueous solution was calculated using Eq. 2:
| (2) |
RESULTS
Characterization of Microbial Biomass and Tannery Effluent
Different physico-chemical parameters of Aspergillus biomass (MB1) are as follows: pH, 5.0±0.52; bulk density (g cm-3), 0.58±0.35; ash (%), 5.58±0.56; moisture (%), 7.50±0.52; carbon (%), 46.24±0.20; hydrogen (%), 6.8±0.42; nitrogen (%), 3.22±0.54; calorific value (MJ kg-1), 16.42±1.51, Tannery effluent was acidic (pH, 3.5), greenish in colour with Cr (III) (1700.9 mg L-1), total solids (96,000 mg L-1), total dissolved solids (80,000 mg L-1) had Chemical Oxygen Demand (COD) of 332.8 mg L-1 and Biochemical Oxygen Demand (BOD) of 290 mg L-1. Traces of other heavy metals such as Fe, Pb Co, Cu, Cd and secondary elements like Ca, Mg and Na were also present.
Surface Characterization of Microbial Biomass
To determine the behaviour of microbial biomass towards adsorption of Cr (III), X-Ray Diffraction (XRD) and Fourier infrared spectroscopy (FTIR) of native and tannery effluent loaded biomass were carried out. X-Ray diffraction of both biomass before and after saturation with Cr (III) was carried out to identify the phase present on adsorbent (Fig. 1, 2). The XRD analysis of Aspergillus sp. biomass did not show any peak after tannery effluent treatment indicating amorphous nature of the biomass. The FTIR spectrum of biomass displayed a number of adsorption peaks at 1655, 1546, 1319 and 1034 cm-1 indicating the presence of CN, CC, NH, CS-NH, OH, C-Cl, C-O groups on the biomass surface Fig. 3.
Batch Sorption Studies
Batch sorption studies on Cr (III) removal from aqueous solution by Aspergillus sp. (MB1) was carried out to optimize various parameters such as adsorbent dosage, pH, chromium concentration and contact time, removal of Cr (III) from tannery effluent.
Effect of Adsorbent Dosage
Effect of adsorbent dosage (0.25-2%) on sorption of Cr (III) from aqueous solution (pH4) containing (25 mg L-1) with agitation rate of 120 rpm at ambient temperature was studied. Increasing adsorbent dosage (0.25-2%), Cr (III) removal increased rapidly from 24.3 to 94.6% by (Aspergillus sp. (MB1)).
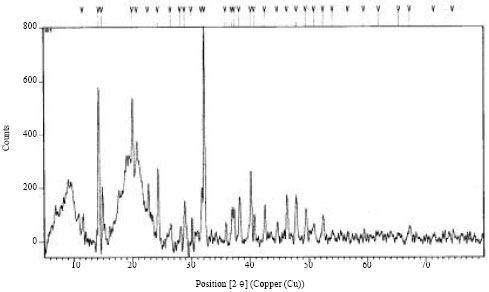 | |
| Fig. 1: | XRD patterns of Aspergillus sp. (MB1) biomass before treatment |
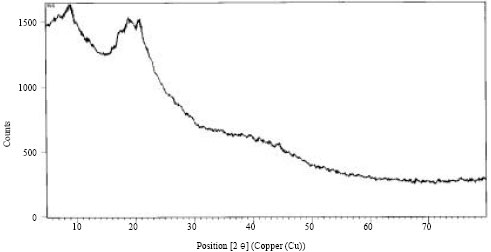 | |
| Fig. 2: | XRD patterns of Aspergillus sp. (MB1) biomass after tannery effluent treatment |
Effect of pH
The amount of Cr (III) adsorbed increased from 1.17 to 2.24 mg g-1 in Aspergillus sp. MB1 as the pH increased from 2 to 5. Maximum Cr (III) removal of 94.6% was observed with Aspergillus sp. (MB1) at pH 4.0.
Effect of Chromium Concentration
Kinetic studies of Cr (III) removal by Aspergillus sp. biomass (MB1) was conducted at varying initial concentrations (5-50 mg L-1) of Cr (III). Maximum removal of Cr (III) from aqueous solution and tannery effluent by Aspergillus sp. biomass was 94.6 and 64.48%, respectively. Removal of Cr (III) was dependent on concentration of chromium because increase in initial chromium concentration.
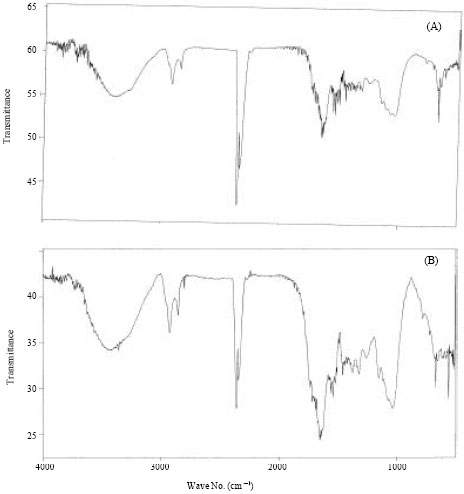 | |
| Fig. 3: | FTIR spectra of Aspergillus sp. (MB1) biomass, (A) untreated biomass and (B) tannery effluent treated biomass |
Effect of Contact Time
Biosorption of chromium onto Aspergillus sp. biomass increased with contact time from 0.08 to 4 h led to an increase in Cr (III) removal from 40-70% and 20-50% of Cr (III) from aqueous solution tannery effluent, respectively within first 2 h, which represents the time at which equilibrium of chromium biosorption is presumed to have been attained. This suggests that the adsorption is rapid and typically 40-50% of the ultimate adsorption occurs within the first hour of contact and saturation is reached with in next 48 h.
Adsorption Kinetics
Adsorption kinetics of Cr (III) study describes solute uptake rate controls which residence time of adsorbate uptake at solid and solution interface. Conformity between experimental data and the models predicted values was expressed by correlation coefficients (R2 values near or equal to 1). A relatively high R2 value indicates that model successfully described kinetics of Cr (III) adsorption.
The Pseudo First-Order Equation
Lagergren (1989), the Pseudo first-order equation is generally expressed as follows:
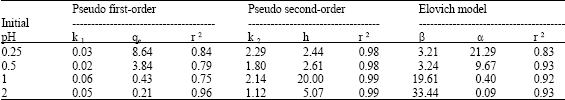
| Table 1: | The adsorption kinetic model rate constants for the MB1 (Aspergillus sp.) at different adsorbent dosage (Ci = 50 mg L-1, adsorbent dosage 1%, pH 4, 120 rpm) |
 | |
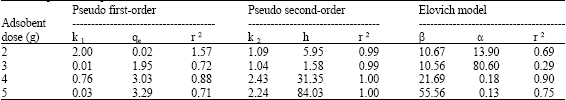
| Table 2: | The adsorption kinetic model rate constants for the MB1 (Aspergillus sp.) at different pH (Ci =50 mg L-1, pH 4, 120 rpm) |
 | |
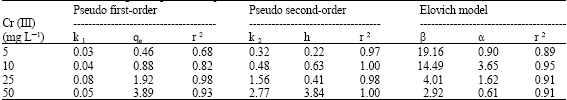
| Table 3: | The adsorption kinetic model rate constants for the MB1 (Aspergillus sp.) at different concentration of chromium (Adsorbent dosage 1%, pH 4, 120 rpm) |
 | |
| (3) |
where, qe and qt are the adsorption capacity at equilibrium and at time t, respectively (mg g-1), kl rate constant of pseudo first-order adsorption process (L min-1). After integration and applying initial conditions t = 0 to t = t and qt = 0 to qt = qt, the integrated form of Eq. 3 becomes:
| (4) |
Plot of log (qe–qt) vs. t should give a linear relation from which kl and qe can be determined from slope and intercept of the plot, respectively. R2 of Cr (III) under different conditions were calculated from plots at different pH (Table 1), biomass dosage (Table 2) and Cr (III) concentration (Table 3). A plot of log (qe–qt) versus agitation time, gave a straight line with R2 of 0.88 for MB1 at pH 4 (Table 1-3). Results showed that adsorption reaction can be approximated with pseudo first-order kinetic model. It was observed that k1 (0.03-0.05) and qe (8.64-0.21) at different adsorbent dosage (0.25-2%) and values of k1 (2.00-0.03) and qe (0.02-3.29) at different pH. In most of the cases, first-order equation of Lagergren did not apply throughout complete range of contact time and is generally applicable over initial (20-30 min) sorption process. The pseudo second-order equation (Ho and Mckay, 1999; Ho et al., 1996).
The pseudo second-order adsorption kinetics model based on the sorption capacity of solid phase can be used in this case assuming that measured concentrations are equal to the cell surface concentrations. If the rate of sorption is a second order mechanism, the pseudo second-order chemisorption kinetic equation for Cr (III) both singly and in the mixture is expressed as:
| (5) |
where, k2 is rate constant of pseudo second-order adsorption (g/mg/min). For initial condition t = 0 to t = t and qt = 0 to qt = qt, integrated form of Eq. 5 becomes:
| (6) |
This is integrated rate law for a pseudo second-order reaction. Equation 6 can be rearranged as, which has a linear from:
| (7) |
If the initial adsorption rate, h (mg/g/min) is:
| (8) |
| (9) |
Plot of (t/qt) and t of Eq. 7 should give a linear relationship, from which qe and k2 can be determined from slop and intercept of plots, respectively. R2 for linear plots are found superior (in most cases = 0.99) (Table 1-3). It was observed that k2 (2.29-1.12) and h (2.44-5.07) increase with increase in biomass dosage and k2 Values of k2 (1.09-2.24) and h (5.95-84.03) increased at different pH. Values of k2 (0.32-2.77) and h (0.22-3.84) increased with increase in initial Cr (III) concentration. Values of h and k2 were found higher for MB1 at different parameters (adsorbent dosage, pH and Cr(III) concentration). Experimental points shown together with theoretically generated values reflect extremely high correlation coefficients (Table 1-3).
The Elovich Equation
The Elovich model equation is generally expressed as (Chien and Clayton, 1980):
| (10) |
where, α is the initial adsorption rate (mg/g/min), β is the desorption constant (g mg-1) during any one experiment. To simplify the Elovich equation, assumed αβt >> t and by applying the initial conditions (qt = 0 at t = 0 and qt =qt at t = t) Eq. 10 becomes:
| (11) |
If, Cr (III) adsorption fits Elovich model, a plot of qt vs. ln (t) should yield a liner relationship with a slope of (l/β) and an intercept of (1/β) ln (α β). R2 were determined from plot between qt and ln (t), respectively at different biomass dosage, pH and chromium concentration (Table 1-3). Computed value of α and β were calculated at biomass dosage [α (21.29-0.09), β (3.21-33.44)], pH [α (13.90-0.13) β (10.67-55.56)] and Cr (III) concentration [α (0.90-0.61) β (19.16-2.92)]. Sorption kinetics of Cr (III) examined using Elovich model gave R2 of 0.90 for MB1, which indicates that dynamics data fitted for biomass at pH 4.
DISCUSSION
Physicochemical treatment of such effluent is not only expensive but not eco-friendly. Use of low cost natural resource as the adsorbents for the removal of chromium is constantly encouraging in preference to other conventional methods. Biosorption offers excellent potential for the removal of metals from effluent. A multitude of biomass types comprising fungal, bacterial, algae, peat biomass and so on have been studied for metal biosorption (Kapoor and Viraraghavan, 1998; Chandrasekhar et al., 1998; Tobin and Roux, 1998; Gupta et al., 2001; Cossich et al., 2004). Different non conventional biosorbents of plant and microbial origin were tested for removal of heavy metals from synthetic effluents in single metal and multi-metallic state of industrial effluent to develop an inexpensive adsorbent. The process can be made economical by procuring natural bulk biomass or spent biomass from various fermentation industries (Ahluwalia and Goyal, 2007). Waste biomass generated by different industrial processes was developed as a biosorbent for its large scale application for removal of chromium from mixed metal effluent (Gupta et al., 1999; Dias et al., 2000; Li et al., 2004; Fahim et al., 2006).
In this study, greenish tannery effluent (pH, 3.5; high Cr (III), 1700.9 mg L-1) was treated with microbial waste biomass from fermentation industry. Physicochemical parameters of Aspergillus sp. biomass and calorific value were found to coincide with the relative calorific value (17.5 MJ kg-1) of solid fuels such as biomass, municipal waste, industrial waste, peat and brown coal (MacDonald et al., 1996). The XRD data of treated adsorbent provided evidence of decrease in the peak intensity which shows the adsorption of Cr(III) on the surface of the adsorbent changing its structure from crystalline to amorphous. This is in agreement with the previous studies on adsorption (Gopal and Elango, 2007; Karthikeyan and Ilango, 2008).
Comparison of the FTIR spectra of native and tannery effluent treated biomass revealed that new adsorption peaks at 2360, 1451 and 1376 cm-1 were formed indicating the involvement of amine N-N=O, CS-NH, OH group, respectively. The assignment of a specific wave number to a given functional group was not possible because the adsorption bands of various functional groups overlap and shift, depending on their molecular structure and environment. Shifts in absorption positions may be caused by many factors. These include: (1) the physical state (2) electronic and mass effects of neighbouring substitutes (3) conjugation (4) intra-molecular and intermolecular hydrogen bonding and (5) ring strain (Mohan et al., 2006).
In batch sorption studies chromium removal affinity of the microbial biomass was determined by aqueous solution and tannery effluent at different adsorbent dosage (0.25-2 g), pH (2-5), initial chromium (III) concentration (5-50 mg L-1) and contact time. With increasing adsorbent dosage (0.25-2%), Cr (III) removal increased rapidly from 24.3 to 94.6% by (Aspergillus sp. Similar observation were made with Aspergillus sp. (Chandrasekhar et al., 1998) which showed 70-75% removal of Cr (III) from aqueous solution and exhibited an adsorption capacity of 1.8-2 mg g-1 (Mattuschka and Straube, 1993) and comparable with commercial activated carbon (2.7 mg g-1). Chromium exhibits different types of pH dependent adsorption (Mohan et al., 2006) maximum (94.6%) removal of Cr (III) was observed with Aspergillus sp. (MB1) at pH 4.0. Similar observations were made for biowaste generated as a by-product of large-scale industrial fermentation (Dias et al., 2000; Cossich et al., 2004; Fahim et al., 2006; Chandrasekhar et al., 1998). It is clear that sorption of trivalent chromium species increased with increase in solution pH from 2 to 5.0. Studies were not performed beyond pH 5.0 due to the possibility of Cr (III) precipitation (Mohan et al., 2006). Adsorption of Cr (III) is therefore an electrostatic interaction between biomass and Cr (III) cations. Initially, the immediate solute uptake achieved within a 2 to 4 h, subsequently the second stage of solute uptake continued for longer time (24 h) period (Demirbas et al., 2004).
Maximum removal of Cr (III) from aqueous solution and tannery effluent by Aspergillus sp. biomass was 94.6 and 64.48%, respectively. Earlier reports also indicate that removal of Cr (III) is dependent on concentration of chromium because increase in initial chromium concentration (50-300 mg L-1) increases the amount of Cr (III) adsorbed (Demirbas et al., 2004).
Biosorption of chromium onto Aspergillus sp. biomass increased with contact time from 40-70% and 20-50% of Cr (III) from aqueous solution tannery effluent, respectively. Diminishing removal with increasing time may be due to intraparticle diffusion process dominating over adsorption (Chandrasekhar et al., 1998; Kapoor and Viraraghavan, 1998).
The kinetics of Cr (III) adsorption on the Aspergillus sp. were analyzed using pseudo first-order (Lagergren (1989)), pseudo second-order Elovich (Chien and Clayton, 1980) and intraparticle diffusion (Srivastava et al., 1997) kinetic models. A relatively high R2 value indicates that the model successfully describes the kinetics of Cr (III) adsorption. Adsorption kinetic of Cr (III) removal has been further studied at different adsorbent dosage, pH, chromium concentration and contact time. In most of the cases, first-order equation of Lagergren did not apply throughout the contact time and is generally applicable over initial (20-30 min) sorption (Mohan et al., 2006; Lagergren (1989)). Slope and intercepts as calculated from plots were used to determine k1 (first-order rate constant) and equilibrium capacity (qe). Values of qe are found lower than experimental one. Therefore, chromium-adsorbent systems do not follow a first-order rate equation (Mohan et al., 2006; Ho, 2004; Demirbas et al., 2004). Data showed good compliance with pseudo-second order kinetic model (R2>0.99) (Mohan et al., 2006; Fahim et al., 2006; Li et al., 2004) . However, these two models do not provide a definite mechanism. Therefore, another simplified model (Ho et al., 1996; Ho and Mckay, 1999) was tested, in which kinetic data is better fitted by second-order rate equation (Mohan et al., 2006; Ho, 2004; Demirbas et al., 2004). Experimental qe values were compared with qe value determined by pseudo-first and second-order rate kinetic models, which suggests that sorption system is not a pseudo first-order reaction and that a pseudo-second-order model can be considered (Demirbas et al., 2004).
Novelty and significant contribution of the present study is that the capability of industrial waste microbial biomass (Aspergillus sp.) for the removal of trivalent chromium from aqueous solution and tannery effluent has been studied. The waste biomass can be easily procured from industrial fermentation processes as byproduct and no cost is involved in its production for using as sorbent which serve as highly economical with constant supply in an eco-friendly manner. Present investigation revealed that the industrial waste biomass at lower concentration can efficiently remove Cr (III) under various laboratory conditions. In addition, kinetic models were in agreement with experimental data, which clearly indicates its significant role in remediation of chromium (III) and leading to a product as well as process development which will have industrial application and commercial value and significance in pollution abatement. The strategy will lead to value addition to the waste biomass of one industry and the product can be used for treatment of effluent from another industry which will be one time treatment of metal containing effluent leading to its detoxification in cost effective and eco-friendly manner. Present study provides a significant contribution towards the trivalent chromium removal by waste biomass, which can be subsequently extrapolated for treatment of industrial wastewater.
CONCLUSION
Microbial waste biomass comprising of Aspergillus sp. involved in fermentation process was effective in removal of Cr (III) from aqueous solution as well as tannery effluent. To our knowledge, this microbial biomass does not find any economic application and represents a solid pollutant to the environment. In this study, maximum chromium removal capacities of Aspergillus sp. biomass (MB1) were 94.6% from aqueous solution and 64.48% from tannery effluent at ambient temperature, rpm 120 and initial concentration (50 mg L-1) of Cr (III). A comparison of kinetic models (Lagergren, Ho and McKay, Elovich and Morris-Weber) applied to MB1 indicates that adsorption of Cr (III) on MB1 follows a best Ho and McKay pseudo second-order rate equation and correlation coefficient (R2) correlated with experimental data. XRD and FTIR studies reveals that Cr (III) adsorption takes place on the surface of the adsorbent. This can provide means for using solid waste of one industry to treat chromium containing effluents from another industry for development of a suitable cost effective biosorbent.
ACKNOWLEDGMENTS
The authors are thankful to Council of Scientific and Industrial Research (CSIR) New Delhi, vide Sanction No. 24 (0284)/05/EMR-II, for providing financial support, Ranbaxy Paonta Sahib, Himachal Pradesh, India for providing microbial biomass and director, Thapar University for providing infrastructural support.
REFERENCES
- Ahluwalia, S.S. and D. Goyal, 2007. Microbial and plant derived biomass for removal of heavy metals from wastewater. Bioresour. Technol., 98: 2243-2257.
CrossRefDirect Link - APHA, AWWA and WEF., 1998. Standard Methods for the Examination of Water and Wastewater. 20th Edn., American Public Health Association/American Water Works Association/Water Environment Federation, Washington, DC., USA., ISBN-13: 9780875532356, Pages: 1220.
Direct Link - Arvindhan, R., B. Madhan, J.R. Rao, B.U. Nair and T. Ramasami, 2004. Bioaccumulation of chromium from tannery wastewater: an approach for chrome recovery and reuse. Environ. Sci. Technol., 38: 300-306.
CrossRef - Sekhar, K.C., S. Subramaniam, J.M. Modak and K.A. Natarajan, 1998. Removal metal ions using industrial biomass with reference to environmental control. Int. J. Miner. Process., 53: 107-120.
CrossRef - Chien, S.H. and W.R. Clayton, 1980. Application of elovich equation to the kinetics of phosphate release and sorption in soils. Soil Sci. Soc. Am. J., 44: 265-268.
CrossRefDirect Link - Cossich, E.S., E.A. Da Silva, C.R.G. Tavares, L.C. Filho and T.M.K. Ravagnani, 2004. Bio sorption of chromium (III) by biomass of Seaweed Sargassum sp. in a fixed-bed column. Adsorption, 10: 129-138.
CrossRef - Demirbas, E., M. Kobya, E. Senturk and T. Ozkan, 2004. Adsorption kinetics for the removal of chromium (VI) from aqueous solutions on the activated carbons prepared from agricultural wastes. Water SA., 30: 533-539.
Direct Link - Dias, M.A., H.F. Castro, P.F. Pimentel, N.C.M. Gomes, C.A. Rosa and V.R. Linardi, 2000. Removal of heavy metals from stainless steel effluents by waste biomass from Brazilian alcoholic beverage production. World J. Microbiol. Biotechnol., 16: 107-108.
CrossRef - Fahim, N.F., B.N. Barsoum, A.E. Eid and M.S. Khalil, 2006. Removal of chromium (III) from tannery wastewater using activated carbon from sugar industrial waste. J. Hazard. Mater., 136: 303-309.
CrossRef - Gaballah, I. and G. Kilbertus, 1998. Recovery of heavy metal ions through decontamination of synthetics solutions and industrial effluents using modified barks. J. Geochem. Explor., 62: 241-286.
CrossRef - Gopal, V. and K.P. Elango, 2007. Equilibrium, kinetic and thermodynamic studies of adsorption of fluoride onto plaster of Paris. J. Hazard. Mat., 141: 98-105.
CrossRef - Gupta, V.K., A.K. Shrivastava and N. Jain, 2001. Biosorption of chromium (VI) from aqueous solutions by green algae Spirogyra species. Water Res., 35: 4079-4085.
Direct Link - Ho, Y.S., D.A.J. Wase and C.F. Forster, 1996. Kinetic studies of competitive heavy metal adsorption by sphagnum moss peat. Environ. Technol., 17: 71-77.
CrossRefDirect Link - Gupta, V.K., D. Mohan, S. Sharma and K.T. Park, 1999. Removal of chromium (VI) from electroplating industry waste using bagasse fly ash a sugar industry waste material. Environmentalist, 19: 129-136.
CrossRefDirect Link - Ho, Y.S., 2004. Citation review of Lagergren kinetic rate equation on adsorption reactions. Scientometrics, 59: 171-177.
CrossRefDirect Link - Kapoor, A. and T. Viraraghavan, 1998. Biosorption of heavy metals on Aspergillus niger: Effect of pretreatment. Bioresour. Technol., 63: 109-113.
CrossRefDirect Link - Karthikeyan, G. and S.S. Ilango, 2008. Adsorption of Cr(VI) onto activated carbons prepared from indigenous materials. E-Journals Chemistry, 5: 666-678.
Direct Link - Kongsricharoern, N. and C. Polprasert, 1996. Chromium removal by a bipolar electro-chemical precipitation process. Water Sci. Technol., 34: 109-116.
Direct Link - Kozlowski, C.A. and W. Walkowiak, 2002. Removal of chromium (VI) from aqueous solutions by polymer inclusion membranes. Water. Res., 36: 4870-4876.
CrossRef - Li, Y.S., C.C. Liu and C.C. Chiou, 2004. Adsorption of Cr(III) from wastewater by wine processing waste sludge. J. Colloid. Interface. Sci., 273: 95-101.
CrossRef - Mohan, D., K.P. Singh and V.K. Singh, 2006. Trivalent chromium removal from wastewater using low cost activated carbon derived from agricultural waste material and activated carbon fabric cloth. J. Hazard. Mater., 135: 280-295.
CrossRef - Petruzzelli, D., R. Passino and G. Tiravanti, 1995. Ion exchange process for chromium removal and recovery from tannery wastes. Ind. Eng. Chem. Res., 34: 2612-2617.
CrossRef - Rengaraj, S., C.K. Joo, Y. Kim and J. Yi, 2003. Kinetics of removal of chromium from water and electronic process wastewater by ion exchange resins: 1200H, 1500H and IRN97H. J. Hazard. Mater., 102: 257-275.
CrossRefDirect Link - Seaman, J.C., P.M. Bertsch and L. Schwallie, 1999. In situ Cr(VI) reduction within coarse-textured, oxide-coated soil and aquifer systems using Fe(II) solutions. Environ. Sci. Technol., 33: 938-944.
CrossRef - Shaalan, H.F., M.H. Sorour and S.R. Tewfik, 2001. Simulation and optimization of a membrane system for chromium recovery from tanning wastes. Desalination, 141: 315-324.
Direct Link - Srivastava, S.K., V.K. Gupta and D. Mohan, 1997. Removal of lead and chromium by activated slag-A blast-furnace waste. J. Environ. Eng., 123: 461-468.
CrossRefDirect Link - Testa, J.J., M.A. Grela and M.I. Litter, 2004. Heterogeneous photocatalytic reduction of chromium (III) over TiO2 particles in the presence of oxalate: Involvement of Cr(VI) species. Environ. Sci. Technol., 38: 1589-1594.
CrossRef - Tiravanti, G., D. Petruzzelli and R. Passino, 1997. Pretreatment of tannery wastewaters by an ion exchange process for Cr(III) removal and recovery. Water. Sci. Technol., 36: 197-207.
Direct Link - Tobin, J.M. and J.C. Roux, 1998. Mucor biosorbent for chromium removal from tanning effluent. Water Res., 32: 1407-1416.
Direct Link - Zhou, X., T. Korenaga, T. Takahashi, T. Moriwake and S. Shinoda, 1993. A process monitoring/controlling system for the treatment of wastewater containing chromium (VI). Water Res., 27: 1049-1054.
CrossRef - Ho, Y.S. and G. McKay, 1999. Pseudo-second order model for sorption processes. Proces. Biochem., 34: 451-465.
CrossRefDirect Link








