Research Article
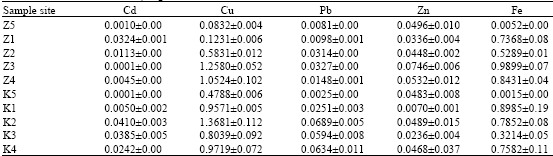
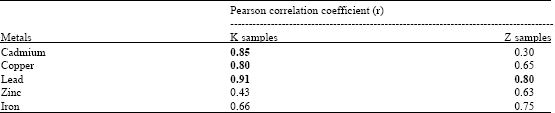
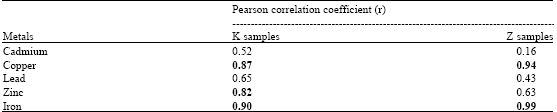
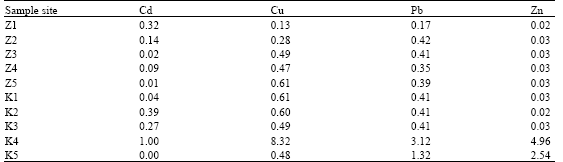
Earthworm (Eudrilus eugenia Kinberg) as Bio-Indicator of the Heavy Metal Pollution in Two Municipal Dump Sites of Two Cities in Northern Nigeria
Department of Chemistry, Ahmadu Bello University, Zaria 810005, Nigeria
V.O. Ajibola
Department of Chemistry, Ahmadu Bello University, Zaria 810005, Nigeria
F.M. Folaranmi
Department of Chemistry, Ahmadu Bello University, Zaria 810005, Nigeria