Research Article
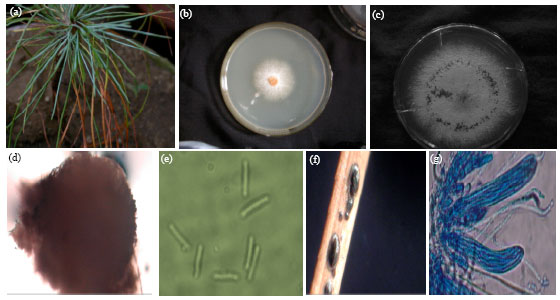
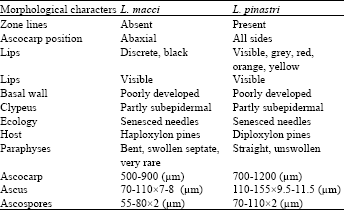
First Report of Needle Blight on Blue Pine (Pinus wallichiana) and Aleppo Pine (P. halepensis), Caused by Lophodermium macci, from Asia
Division of Plant Pathology, S.K. University of Agricultural Sciences and Technology of Kashmir, Shalimar, Srinagar 191 121, Jammu and Kashmir, India
Gh. Hassan Dar
Division of Plant Pathology, S.K. University of Agricultural Sciences and Technology of Kashmir, Shalimar, Srinagar 191 121, Jammu and Kashmir, India
Muzafar A. Beig
Division of Plant Pathology, S.K. University of Agricultural Sciences and Technology of Kashmir, Shalimar, Srinagar 191 121, Jammu and Kashmir, India
T.A. Sofi
Division of Plant Pathology, S.K. University of Agricultural Sciences and Technology of Kashmir, Shalimar, Srinagar 191 121, Jammu and Kashmir, India