Research Article
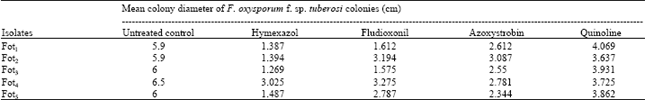
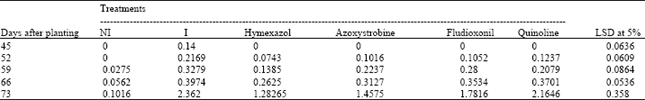
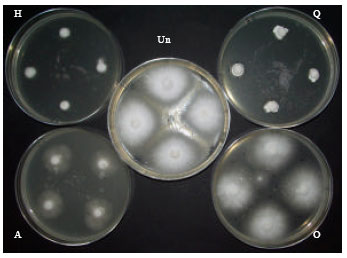
Evaluation of Fungicides for Control of Fusarium Wilt of Potato
Horticultural High School of Chott-Mariem, 4042 Sousse, Tunisia
Mejda Daami-Remadi
National Institute of Agronomic Research, Tunisia (PRRDA-CE Chott-Mariem, 4042 Sousse- Tunisia)
Hayfa Jabnoun-Khiareddine
Horticultural High School of Chott-Mariem, 4042 Sousse, Tunisia
Khaled Hibar
Horticultural High School of Chott-Mariem, 4042 Sousse, Tunisia
Mohamed El Mahjoub
Horticultural High School of Chott-Mariem, 4042 Sousse, Tunisia