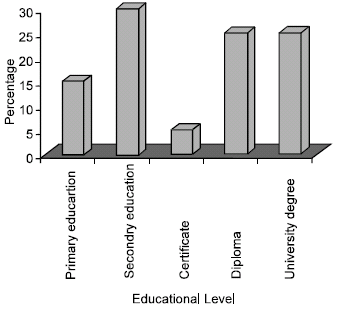
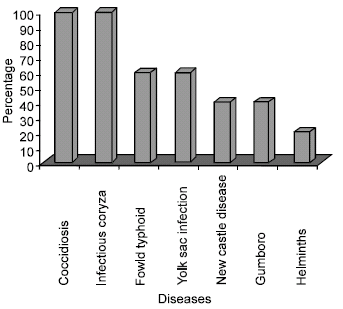
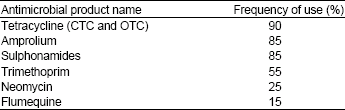
Research Article
Assessment of Antimicrobial Usage and Antimicrobial Residues in Broiler Chickens in Morogoro Municipality, Tanzania
Department of Veterinary Medicine and Public Health, Sokoine University of Agriculture, Box 3021, Morogoro, Tanzania
M. Mariki
Department of Veterinary Medicine and Public Health, Sokoine University of Agriculture, Box 3021, Morogoro, Tanzania
E.D. Karimuribo
Department of Veterinary Medicine and Public Health, Sokoine University of Agriculture, Box 3021, Morogoro, Tanzania
R.H. Mdegela
Department of Veterinary Medicine and Public Health, Sokoine University of Agriculture, Box 3021, Morogoro, Tanzania