Research Article
Enzyme Digestive Activities During Ontogeny in Marosatherina ladigesi Larvae
Department of Aquaculture, Faculty of Fishery and Marine Science, Indonesia Muslim University, Makassar-90231, South Sulawesi, Indonesia
LiveDNA: 62.16564
Amrah Husma
Department of Aquaculture, Faculty of Fishery and Marine Science, Indonesia Muslim University, Makassar-90231, South Sulawesi, Indonesia
Ilmia
Department of Aquaculture, Faculty of Fishery and Marine Science, Indonesia Muslim University, Makassar-90231, South Sulawesi, Indonesia









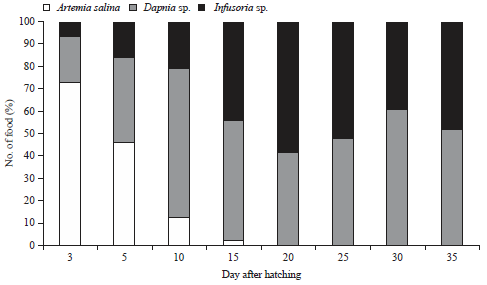
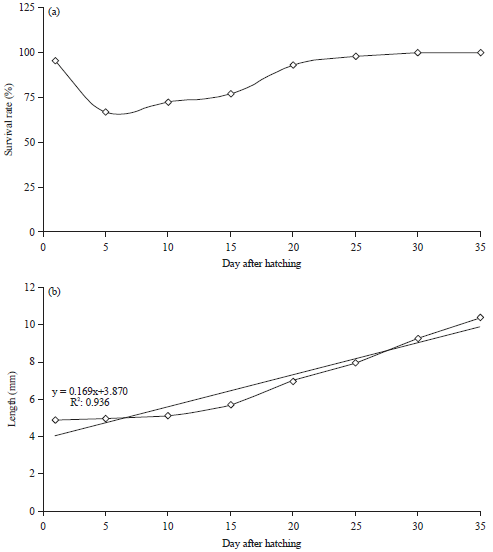
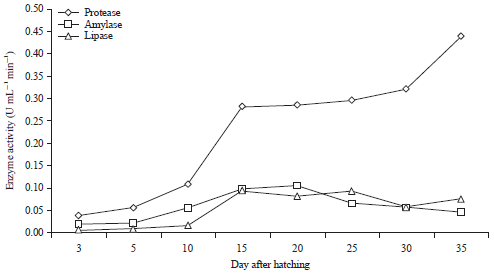
Jayadi Reply
I am very satisfied