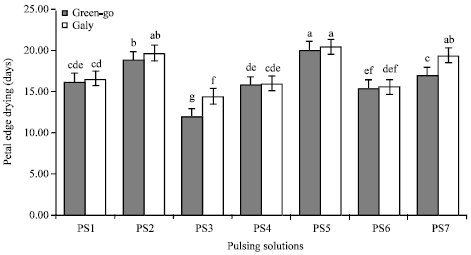
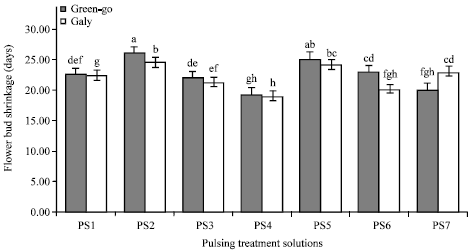
Research Article
Response of Carnation (Dianthus caryophyllus) Cultivars to Different Postharvest Preservatives
Jimma University College of Agriculture and Veterinary Medicine, P.O. Box 307, Jimma, Ethiopia
Derbew Belew
Jimma University College of Agriculture and Veterinary Medicine, P.O. Box 307, Jimma, Ethiopia
Negussie Kassa
Jimma University College of Agriculture and Veterinary Medicine, P.O. Box 307, Jimma, Ethiopia