Research Article
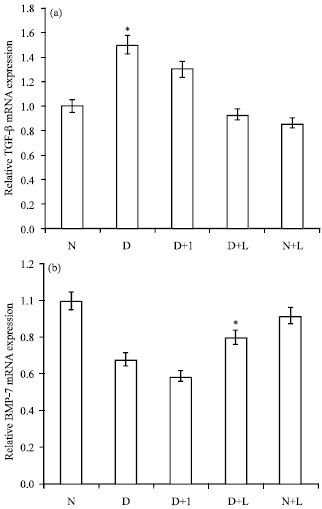
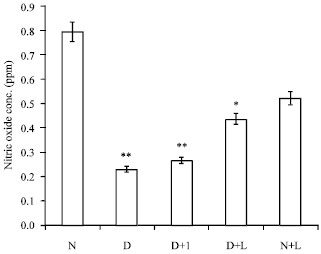
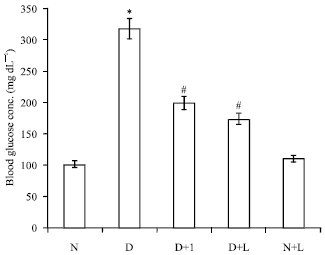
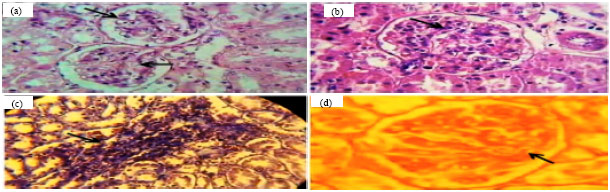
The Effect of Lactobacillus reuteri on Bone Morphogenetic Protein-7 and Beta Transforming Growth Factor Gene Expressions in Streptozotocin-induced Diabetic Rat’s Kidneys
Department of Clinical Biochemistry, Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran
G. Irajian
Department of Bacteriology, Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran
M. Najafi
Department of Clinical Biochemistry, Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran
M. Nourbakhsh
Department of Clinical Biochemistry, Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran
J. Maleki
Department of Clinical Biochemistry, Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran
M. Shabani
Department of Clinical Biochemistry, Faculty of Medicine, Tehran University of Medical Sciences, Tehran, Iran