Research Article
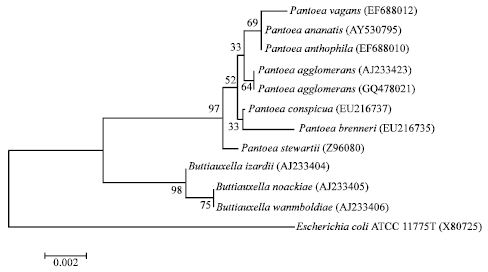
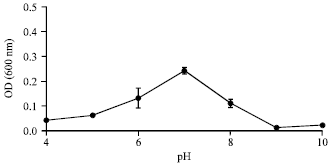
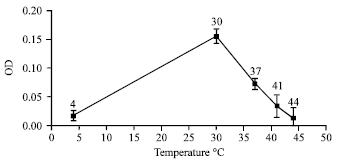
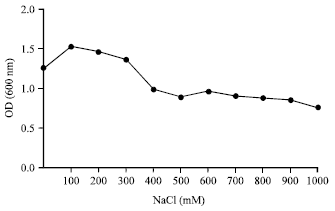
Isolation and Characterization of Plant Growth Promoting Traits of a Rhizobacteria: Pantoea agglomerans lma2
Department of Microbiology, Laboratory of Applied Microbiology, Faculty of Natural and Life Sciences, University of Ferhat-Abbas, Setif, Algeria
A. Silini
Department of Microbiology, Laboratory of Applied Microbiology, Faculty of Natural and Life Sciences, University of Ferhat-Abbas, Setif, Algeria
M. Ghoul
Department of Microbiology, Laboratory of Applied Microbiology, Faculty of Natural and Life Sciences, University of Ferhat-Abbas, Setif, Algeria
S. Yadav
Research Scholar School of Life Sciences, Hyderabad Central University, Hyderabad, India