Research Article
Survival of Paramecium caudatum at Various pH Values and Under Normoxic and Hypoxic Conditions
Laboratory of Protozoology, Department of Biology, Shahrekord University, Shahrekord-88186, P.O. Box 115, Iran
The objective of this study was to determine the survival of Paramecium caudatum, a protozoan ciliate, with different pH under normoxic and hypoxic conditions. P. caudatum was exposed to 18 treatments (in triplicate) of varying pH (4, 4.7, 5.7, 6.7, 7.7, 8.7, 9.7, 10.7 and 11.7) with two different conditions of normoxic and hypoxic. Survival was examined every second day for three weeks. P. caudatum mortality was 100% immediately when exposed to pH 4, 10.7 and 11.7 either in normoxic or hypoxic conditions, whereas at other pH values tested this ciliate could survive mostly under hypoxic conditions. The results suggest that under hypoxic condition, 4.7-6.7 is the best pH range for survival of this species.
One of the most influential factors in determining composition of freshwater invertebrates` communities including ciliates is pH. An increase in the rate of photosynthesis by the phytoplankton and other aquatic plants or the presence of high amount of lime may lead to pH over 9.0 (Wood, 2001). Conversely, pH below 6.0 is due to the high production of carbon dioxide by aquatic organisms or from the sulfate in the soil, which yields sulfuric acid (Boyd, 1998).
Freshwater protozoans are dominated at low pH (Packroff, 2000). The richness and abundance of ciliates in natural habitats is also greater in low pH (4.5) (Mieczan, 2007), even though there is a species-specific pH tolerance for planktonic freshwater ciliate species (Weisse and Stadler, 2006). Present knowledge of the survival effects of pH on P. caudatum is generally limited. Most studies that have been reported on Paramecium and pH largely concern the effect of pH on the forward swimming speed of the organism (Otter et al., 2005). While the toxicity effect of extreme values of pH on some species of Paramecium including species caudatum has been reported (e.g., Rao et al., 2006), few studies under laboratory conditions performed to simulate those hydrogen ions that usually occur in the natural habitat. The pH in natural, inland waters varies from 4.0 to 9.0 (Wetzel, 2001).
When oxygen supply is limited (hypoxia), all organisms ranging from protozoans to mammals show a protective response known as an apyrexia (Wood and Malvin, 1991; Malvin et al., 1994; Branco and Gargaglioni, 2006). In fact, hypoxia results in a decrease in body temperature and an apyrexia is a behavioural adaptation to hypoxia. An apyrexia facilitates survival because it lowers metabolic rate of the organism and increases binding affinity between O2 and its carrier (Branco and Malvin, 1996). Hypoxia-induced an apyrexia has also been observed in P. caudatum which is mediated by inhibition of oxidative phosphorylation (Malvin et al., 1994). Hypoxia can occur diurnally or seasonally in natural habitats where Paramecium lives (e.g., pond).
This study was therefore undertaken to examine the laboratory survival effect of pH on P. caudatum and tolerance of this ciliate under normoxic and hypoxic conditions. In this study normoxic condition was defined as air-aerated and a hypoxic condition as a low air-aerated for the media.
Paramecium is a small microscopic unicellular organism that is plentiful in freshwater habitats. Many physiological mechanisms hard to understand in higher vertebrates can be inferred using Paramecium. The cell surface of Paramecium has been used as GABA receptors in modulating inhibitory synaptic transmission in neurons (Ramoino et al., 2006). The thermoregulatory system of the unicellular Paramecium is much simpler than that of vertebrates. This is largely due to the extreme complexity of vertebrate thermoregulatory systems. For example, in Paramecium caudatum, hypoxia appears to exert its thermoregulatory effects by inhibiting oxidative phosphorylation and decreases in intracellular (ATP) and pH may be important intermediate signals (Malvin, 1998).
The experiment was carried in May 2007 at the Biology department, University of Shahrekord, Shahrekord, Iran. Stock culture of P. caudatum was grown in a wheat medium bacterised with Klebsiella pneumoniae under a photoperiod of 12:12 h light:dark (12L:12D) at room temperature (22-24°C).pH of the stock solution was set at 7.7. The ciliates were kept in exponential growth. The experimental tests to ascertain the effect of pH on Paramecium were performed in the following. P. caudatum was subjected to various pH values. Therefore, nine treatments of different pH solutions i.e., 4, 4.7, 5.7, 6.7, 7.7, 8.7, 9.7, 10.7 and 11.7 were made by adding acetic acid or sodium carbonate to the stock solution. Six 50 mL test tubes were selected for each treatment and filled (15 mL) from the stock culture. The caps of the three test tubes of any treatment were left open (normoxic or aerated) and the others were covered by means of parafilm to induce a hypoxia condition. Following Pedersen and Hansen (2003) to avoid shock effect of pH, the pH was raised or lowered stepwise (0.5-1 unit at 12 h intervals) until the desired level was reached. pH was measured using a pH meter, sensitivity 0.01 and a 2 points calibration. Due to shift in pH with time, the pH of each treatment was adjusted by addition of small amounts of 0.1 mol-1 acetic acid or sodium bicarbonate. The experiment lasted for three weeks and sampling was made every 24 h following Salvado and Gracia (1993). Thus sub-samples (20 μL each) of each tube were taken with a micropipette and the numbers of live P. caudatum were counted under an optical microscope with a magnification of 1000x. A highly viscous medium containing 0.5% methylcellulose (Iwadate et al., 1997) was used to slow P. caudatum swimming and therefore facilitate their counting. A paired t-test was used to compare the survival effect of varying pH under normoxic and hypoxic conditions. Significant differences were accepted at p<0.05.
P. caudatum can resist and survive under various acidic and alkaline situations even though its survival appears to be at more acidic and hypoxic environment. At pH values of less than 4.7 or greater than 9.7, whether in normoxic or hypoxic conditions, P. caudatum immediately died, whereas at pH 4.7-9.7 were able to survive. No significant differences were found in the cells at pH values of 4.7, while there were significant differences at pH values of 5.7-9.7 between normoxic and hypoxic conditions (p<0.05,Table 1,Appendix). During the
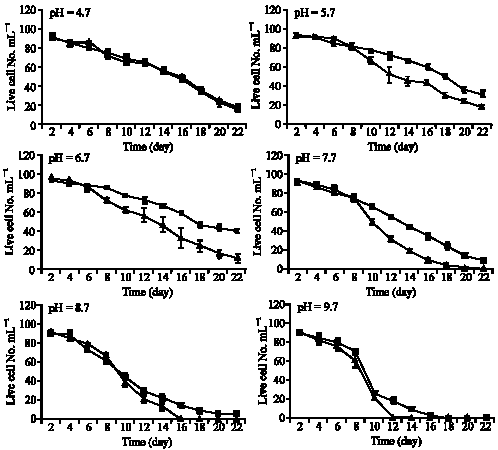 | |
| Fig. 1: | No. of live Paramecium caudatum at different pH under normoxic ( |
| Appendix: | Paired samples test |
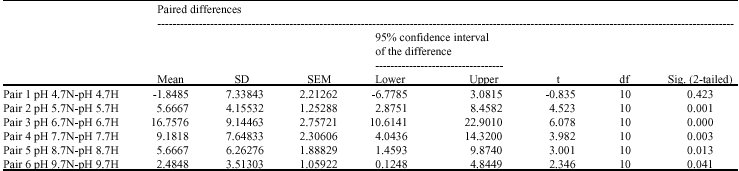 | |
| Table 1: | A paired t-test for comparing the survival effect of varying pH under normoxic (n) and hypoxic (h) conditions |
 | |
first week of the experiment, the growth rates were not significantly influenced under normoxic and/or hypoxic condition. The ciliates survived properly. However, the live number of cells mL-1 declined with time so that there was a significant reduction in the number of live cells at the end of the second week. The rate of this phenomenon (a rapid reduction in number of live cells or extinction) was promoted under normoxic rather than hypoxic condition in particular at pH more than 7.7, so that at the end of the third week nearly all the cells became extinct under normoxic condition.
The cell reduction at pH value of 4.7 did not significantly differ throughout the experiment under either normoxic and/or hypoxic condition so that a parallel reduction in the number of Paramecium caudatum was found. The process of divergence in the cell number reduction between normoxic and hypoxic condition appeared to have taken placed from pH 5.7 onward. This was evident in particular at pH 7.7 (on day 9 onward) until the end of the experiment. Except for some days (e.g., day 7 at pH 5.7 or day 8 at pH 8.7) the number of ciliates under normoxic condition almost always was less than those under hypoxic condition. In effect, P. caudatum died more quickly under normoxic than hypoxic condition particularly at higher pH values. Indeed, the higher the pH value, the shorter the life expectancy. For example at pH 9.7, the ciliates died at a higher rate than at pH 6.7 (Fig. 1).
This study is the first to report some data on the effect of two environmental factors i.e., pH and normoxic/hypoxic conditions on the survival of P. caudatum, showing that while this ciliate protozoan has a wide pH range of tolerance from 4.7 to 9.7, it prefers more acidic pH (4.7-6.7) and its survival is higher in hypoxic than normoxic condition. Freshwater protozoans are dominated at low pH (Packroff, 2000) and the better survival of P. caudatum under acidic condition of this study may be parallel with an intracellular pH of 6.80 in this species (Umbach, 1982).
One of the major environmental factors of freshwater ecosystem is pH. It is impacted by biological processes such as photosynthesis or respiration (Weisse and Stadler, 2006). The rapid death of P. caudatum at pH 4, 10.7 and/or 11.7 of this study may be consistent with Doughty (1986) study in which the P. caudatum were almost immediately immobilized at pH values of <4.0 or >10.0. In addition, as with the natural, inland waters of ciliate habitats in which the pH is rarely outside the range of 4.0-9.0 (Reid, 1961; Nyberg, 1974; Wetzel, 2001), this study also shows that P. caudatum have maximum (9.7) and minimum tolerances (4.7) and optimal ranges (4.7-6.7) of pH for survival. Nevertheless, the wide pH range of tolerance for P. caudatum which is promoted under hypoxia has ecological consequences. As P. caudatum is amongst the dominant species of protozoan in natural habitats of ponds (Tharavathi and Hosetti, 2003), with diurnal or seasonal hypoxia, their ability to survive in different ranges of pH under hypoxia condition could be an adaptation to the extreme environment where these species are found. Indeed, the relative survival abilities of protozoa will be determined by the rate and timing of declines in their population numbers and by the periodicity of resource fluctuation that can be tolerated (Jackson and Berger, 1984).
It has been shown that survival in ciliates is related to ability to form food vacuoles (FVs), but usually in mixed cultures and due to the competition. For example, survival or extinction of Paramecium multimicronucleatum cultured with Paramecium tetraurelia is associated with its ability or inability to form food vacuoles (Maruyama and Takagi, 1997). This could be ruled out in the current study as just one type of the Paramecium was originally used. In addition, the better survival of Paramecium under low pH and/or hypoxic condition may be related to the secretion of substances which promoted survival because it appears that ciliates including Paramecium are able to secrete growth-factor-like substances into the medium (Tanabe et al., 1990; Christensen and Rasmussen, 1992; Takagi et al., 1993; Christensen et al., 1995; Vallesi et al., 1995; Tokusumi et al., 1996) and they function as survival factors (Christensen and Rasmussen, 1992; Wheatley et al., 1993; Christensen et al., 1995).
Regardless of normoxic/hypoxic condition, a reduction in number of the cells took place within the second week when P. caudatum was maintained at high pH value particularly at 9.7. This finding is in agreement with Pedersen and Hansen (2003) study who found that exposure to high pH (>9 or higher) for several days to weeks will result in significance decrease in number of protozoan. That maintaining P. caudatum only at pH 4.7 rather the other pH values showed no significance difference (p = 0.423) suggests that at acidic extremes of pH, the effect of pH is much far than normoxic/hyopix effect. From pH 4.7 to 9.7 a significance difference was found under normoxic/hypoxic condition (p<0.05). This shows that at pH values higher pH than 4.7, condition of maintenance the cells regarding normoxic/hypoxic situations became important in survival of this ciliate.
The survival of living systems depends on their ability to respond appropriately to new situations (Gutierrez et al., 2001). P. caudatum of this study was able to survive at pH 4.7-9.7 while it was promoted under hypoxic condition. This could be a compensatory response in P. caudatum in the current study like an apyrexia which is protective response and facilitates survival (Malvin et al., 1994; Branco and Malvin, 1996). As an apyrexia has already been reported in P. caudatum (Malvin et al., 1994), therefore the observed hypoxia-induced an apyrexia in P. caudatum may have been led to survival of this species. As it appears that P. caudatum rarely encyst under deteriorating conditions including pH change, their ability to survive for long periods of time under hypoxic condition suggests that these protozoa utilize the other survival strategies (Jackson and Berger, 1984). In fact, the improved survival of P. caudatum under hypoxic condition may be simply due to a decrease in respiratory and growth rates.
This study tested the effect of constant external pH on survival of P. caudatum. Due to shift in pH with time, particularly for the hypoxic test tubes which were driven upward, I had to add small amounts of acetic acid or sodium bicarbonate into the solution in order to maintain a desired pH. pH variation in medium affects not only the rate of uptake of CO2 but also membrane transport processes and metabolic functions involved in the biochemical composition, the rate of metabolic processes and the extracellular production (Raven, 1980; Nimer et al., 1994; Taraldsvik and Myklestad, 2000). It is obvious that the enzymes responsible for metabolic pathways in the cells have different pH optima, implying that a shift in internal pH may affect the rate metabolic processes (Ouellet and Benson, 1951). Since these effects are equilibrium-controlled ion fluctuations (Pedersen and Hansen, 2003) this could explain the significance difference between survival of the cells particularly at high pH.
In fishes, tolerance to low pH is related to their ability in maintaining ion balance (Gonzalez, 1998) while in aquatic protist tolerance to pH has been suggested to heat-shock proteins (Gerloff-Elias et al., 2005). Therefore, further direction of this research would be to determine the function and nature of these proteins. In addition, in this study the desired pH values were reached and maintained by means of chemicals. However, pH and oxygen condition of natural freshwater habitats rarely remain constant and may fluctuate diurnally. Thus some other studies need to be conducted in situ to elucidate the various effects of these environmental factors on Paramecium survival.
The author wishes to express his sincere gratitude to Mr. Seydaee who helped me in making and maintaining stock solution.