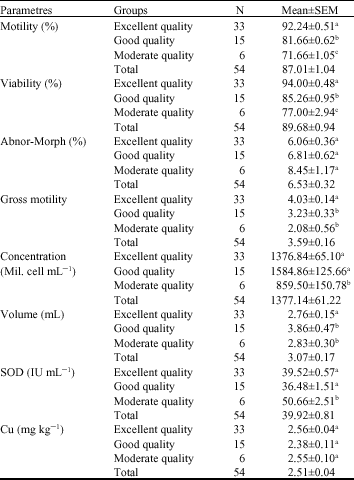
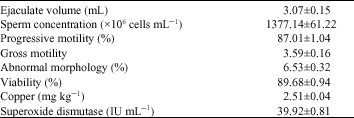Research Article
Effects of Copper and Superoxide Dismutase Content of Seminal Plasma on Buffalo Semen Characteristics
Division of Theriogenology, Department of Veterinary Clinical Science, Faculty of Veterinary Medicine, University of Urmia, Urmia, 57153, Iran
S.M. Alavi-Shoushtari
Division of Theriogenology, Department of Veterinary Clinical Science, Faculty of Veterinary Medicine, University of Urmia, Urmia, 57153, Iran
S. Asri Rezaii
Department of Veterinary Clinical Pathology, Faculty of Veterinary Medicine, University of Urmia, Urmia 57153, Iran