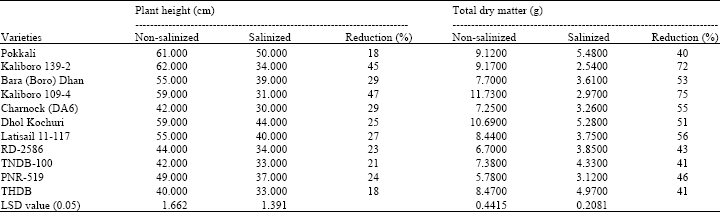
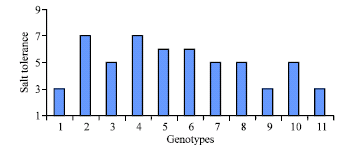
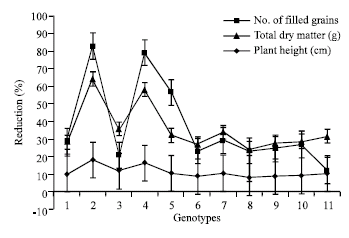
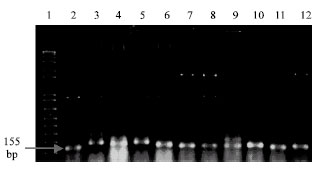
Research Article
Identification of Salt Tolerant Rice Cultivars Via Phenotypic and Marker-Assisted Procedures
Department of Biotechnology, Bangladesh Agricultural University, Mymensingh 2202, Bangladesh
Mirza Mofazzal Islam
Bangladesh Institute of Nuclear Agriculture, P.O. Box 4, Mymensingh 2200, Bangladesh
Reza Mohammad Emon
Bangladesh Institute of Nuclear Agriculture, P.O. Box 4, Mymensingh 2200, Bangladesh
Shamsun Nahar Begum
Bangladesh Institute of Nuclear Agriculture, P.O. Box 4, Mymensingh 2200, Bangladesh
Aysha Siddika
Department of Biotechnology, Bangladesh Agricultural University, Mymensingh 2202, Bangladesh
Sharmin Sultana
Department of Biotechnology, Bangladesh Agricultural University, Mymensingh 2202, Bangladesh