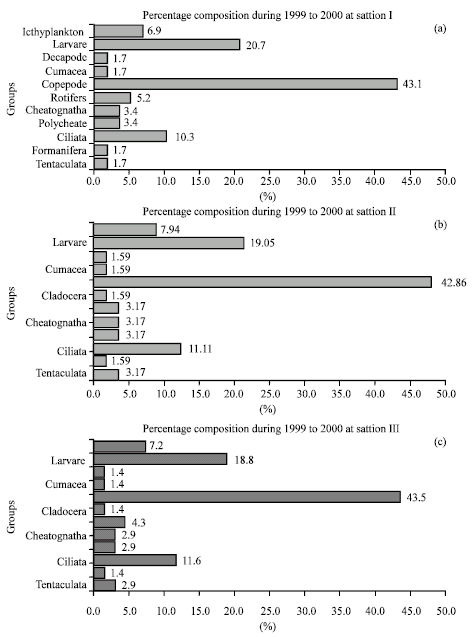
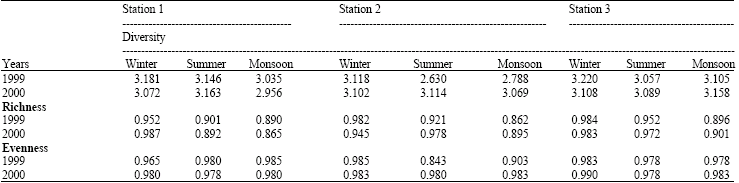
Research Article
Abundance and Seasonal Variations of Zooplankton in the Arid Zone Mangroves of Gulf of Kachchh-Gujarat, Westcoast of India
Center of Advanced Study in Marine Biology, Annamalai University, Parangipettai-608 502, Tamil Nadu, India
M. Rajkumar
Center of Advanced Study in Marine Biology, Annamalai University, Parangipettai-608 502, Tamil Nadu, India
J. Sesh Serebiah
Gujarat Institute of Desert Ecology, Bhuj-370 001, Gujarat, India
G. A. Thivakaran
Gujarat Institute of Desert Ecology, Bhuj-370 001, Gujarat, India