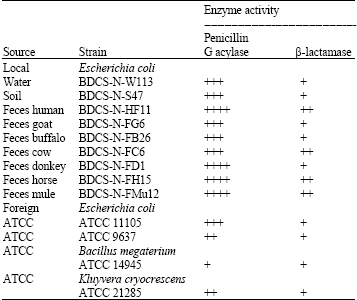
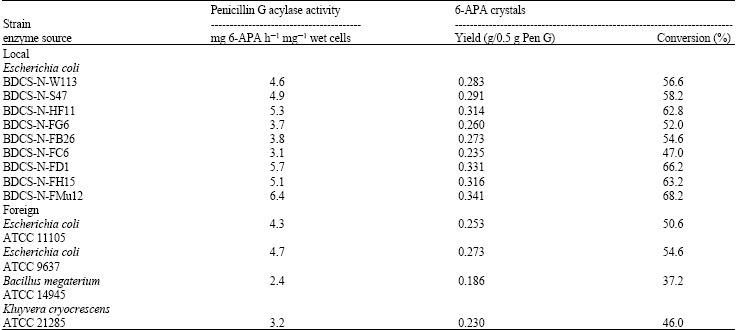
Research Article
6-Aminopenicillanic Acid Production by Intact Cells of E. coli Containing Penicillin G Acylase (PGA)
Nuclear Institute for Agriculture and Biology (NIAB), P.O. Box 128, Jhang Road, Faisalabad, Pakistan
Shafqat Farooq
Nuclear Institute for Agriculture and Biology (NIAB), P.O. Box 128, Jhang Road, Faisalabad, Pakistan
Syed Shahid Ali
Department of Zoology, University of the Punjab, Quaid-e-Azam Campus, Canal Bank Road, Lahore, Pakistan