ABSTRACT
The first peanut crosses ever tried in Greece were performed over two growing seasons (1985 and 1986). The 13 successful crosses in 1985 belong to the following crossing schemes: VirginiaxSpanish, VirginiaxValencia, ValenciaxVirginia, VirginiaxVirginia, ValenciaxValencia and ValenciaxSpanish. The 7 successful crosses in 1986 belong to 2 crossing schemes: VirginiaxValencia and VirginiaxVirginia. It resulted that in the climatic conditions of Greece a higher percentage of success can be achieved (16%), when the crosses are realized as early as possible so that the high summer temperatures can be avoided. Also, the transgressive segregation for yield characteristics of 100-pod weight and 100-seed weight was studied during the years 1985-1990 in the Cotton Research Institute. Pedigree selection was applied from the F3 to F5 generation and segregated materials together with the parental varieties were evaluated. Most of the selections that showed transgressive segregation belonged to the cross type VirginiaxSpanish. Data indicated that yield and quality of peanuts could be improved by exploiting the phenomenon of transgressive variation occurring principally in crosses between varieties that belong to peanut types Virginia and Spanish.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2006.968.973
URL: https://scialert.net/abstract/?doi=pjbs.2006.968.973
INTRODUCTION
Peanut (Arachis hypogaea L.) is an annual subtropical plant cultivated in some areas of Greece since 1915. The genus Arachis includes over 50 species (Gregory and Gregory, 1976). The various species differ in regard to plant habit, stem, leaf, root, fruit and seed characteristics (Banks, 1976). According to Krapovickas (1973) and Krapovickas and Gregory (1994), the cultivated species have been taxonomically classified into two subspecies (hypogaea and fastigiata) with two and four botanical varieties respectively. Krapovickas (1973) also described the differences in the type of flowering between the subspecies hypogaea (market types Virginia and Runner) and fastigiata (market types Valencia and Spanish). Arachis hypogaea has as main distinctive characteristic, the formation of a peg that penetrates into the soil to form an underground fruit (Ramanatha and Murty, 1994). Peanut is normally a self-pollinating species because very few flowers are cross-pollinated and selfing before the flowers are open (cleistogamy) is the rule. Van der Stok (1910) reported natural hybrids in the cultivated peanuts. Hybridization of the cultivated peanut is performed in greenhouses, however in some countries (India, China) hybridization is performed in field nurseries. Hybridization followed by pedigree selection (Norden, 1980) is considered the main breeding method for peanut (Wynne and Gregory, 1981). Pollination among species usually results in fertilization, but with high rate of embryo abortion during early reproductive development, or later in the developmental cycle (Tallury et al., 1995).
The final target of plant crossing is the development of new genotypes that combine useful characteristics from the parents. Progeny superiority was found on both cross-pollinated species after hybridization (Allard, 1960) and self-pollinated species after transgressive segregation in homozygous condition (Fasoulas, 1981). Transgressive segregation enhances response through selection and establishes the superiority of inbred over hybrid vigor and also the semidominant allelic action over codominant and overdominant (Fasoula and Fasoula, 1997). The intervarietal transgression could be explained by complementary action of additive genes dispersed among the original parents (Xu et al., 1998). The transgressive segregation phenomenon was depicted, evaluating earliness and plant height in cereals (Heyne and Smith, 1967), or in case of resistance in diseases in wheat (Wallwork and Johnson, 1984; Broers and Jakobs, 1989), in cotton (Shepherd, 1974) and in barley (Cherif and Harrabi, 1993). Also found in rice, where new inbred lines overyielded the original parents and their hybrid (Xiao et al., 1995) and in red beet (Beta vulgaris L.) populations (Wang and Goldman, 1997). In peanut, there are reported a few data but this phenomenon has not been widely investigated (Coffelt, 1989).
Crossing experiments have never been reported to be undertaken in Greece. The aim of this work was to make the first attempts to cross peanuts in Greece and to study the transgressive segregation phenomenon in pedigree selection schemes for yield characteristics of 100-pod weight and 100-seed weight, in order to determine whether they are useful for peanut breeding.
MATERIALS AND METHODS
The first peanut crosses in Greece took place in the greenhouse of Cotton and Industrial Plants Institute from 15-30 June and 16-30 July 1985 and 2-12 July and also 5-22 August 1986. The method used is described in detail by Norden (1980). The plants used in the crosses as female parents, were grown in pots of 30 cm diameter and 35 cm height on greenhouse benches. Pollen parents were grown outdoors to provide fresh pollen, so that the breeder could make pollinations immediately after emasculating the flowers of the female parent. Since fresh soil was used to fill the pots, this soil was inoculated with Rhizobium bacteria to improve its fertility.
In 1985 crosses of the following varieties took place: a) of Virginia type: Vrasna, Vergina, Lakonia, Karolina b) of Spanish type: Ispaniki, Sakania c) of Valencia type: Serraiki, Taskent (Kotzamanidis, 1994) (Fig. 1). Crossings were performed from 15 to 30 of June, involving 170 flowers and during 16-30 July on only twelve flowers.In 1986 crosses of the Virginia type varieties took place: Lakonia, Pyrgos, Kineziki, NC-7, Vrasna and of Valencia type: B-2510, Serraiki, B-1011 (Kotzamanidis, 1994) (Fig. 2). The crosses were performed in two periods (seasons), 23 of them during 2-10 of July and 179 of them during 5-22 of August.
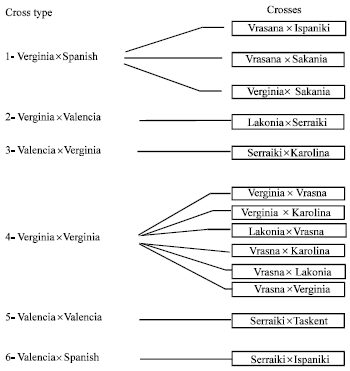 | |
| Fig. 1: | Cross types and varieties used in crosses made in 1985 on peanuts |
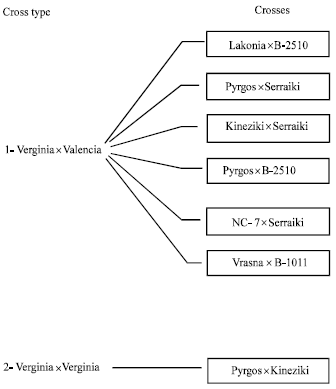 | |
| Fig. 2: | Cross types and varieties used in crosses made in 1986 on peanuts |
The main instruments required for emasculation are a forceps and a sharp penknife. Helpful is a 2xto 3xmagnifier attached to a head band. In the preparation of the female, one flower bud per inflorescence was emasculated, which should be as near to the main stem as possible. Flower buds appear above the leaf axils in the afternoons of warm days and emasculations were done as early as 17.00 h. For the identification of the emasculated flowers, small different coloured threads were used to attach the hypanthium of the emasculated flower and drape the other end over the edge of the pod. On the morning of the day following the emasculation the pollination began which was accomplished between 07.00 and 10.00 h, because peanut pollen is most viable when collected in the early morning (DeBeer, 1963). Two healthy flowers removed from the male parent and transferred to the stigma of the emasculated flowers, were shaken and their pollen was applied onto the stigma. For the protection of the flower from the sun and the high temperatures a paper towel was required, approximately 12x12 cm in size, which was dampened with water and placed carefully over the flower immediately after pollination. The operator was dipping the forceps and rinsing fingers in an alcohol solution before changing from one pollen source to another to reduce the possibility of pollen contamination.
After pollinations were completed, all unpollinated flowers on the plant were removed with forceps. When fertilization was successful, an aerial peg was usually becoming visible 10 to 13 days after pollination. This peg grew down into the soil where the peanut will develop. The developing peg, with the withered flower and string still attached was marked with a colour-coded wire, which was looped around the peg before its penetration into the soil. Developing pegs without attached strings or wires were removed daily for a period of 10 days after the last cross. Approximately 2 months later, the mature peanut fruit with the peg still attached can be harvested. Statistical analysis on the successful crosses was not performed due to unequal size of treatments across seasons, as a result of experimental conditions in field or greenhouse (lack of standard experimental design). Correlations were based on standard procedure described by Snedecor and Cochran (1980), after coding for the four periods of crossing into arithmetic values. This transformation was properly fitted because the four periods were almost equal and successive time intervals.
From the crosses performed during 1985, after pedigree selection was applied, the following crosses were evaluated for yield characteristics of 100-pod weight and 100-seed weight: VrasnaxIspaniki, VrasnaxSakania, VerginaxSakania, LakoniaxSerraiki, SerraikixKarolina and VerginaxVrasna. From the above mentioned crosses 19 accessions were studied together with their parents. The Randomized Complete Block Design was used with 5 replications. The plots were consisted of double rows, 8 m long and distance between plants 0.5 m and between rows 1m. Equal (by weight) amounts of pods and seeds were formed and 5 samples were randomly taken in order to evaluate 100-pod weight and 100-seed weight. ANOVA was based on the procedure described by Snedecor and Cochran (1980).
RESULTS
First peanut crosses in Greece were conducted in 1985 and 1986 and most of them during the first year involving 6 different crossing types (Fig. 1). Crossing type VirginiaxVirginia was the most successful followed by VirginiaxSpanish.
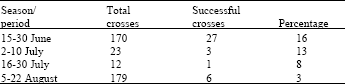
| Table 1: | Total crosses, successful crosses and percentage of success in peanut, during different seasons |
 | |
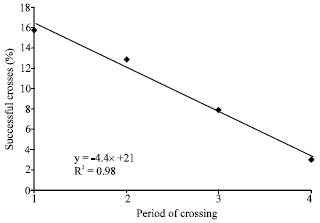 | |
| Fig. 3: | The linear relationship between percentage (%) of successful crosses and the period of practicing the crossings (1= June, 2= early July, 3= late July, 4= August). Correlation significant at p<0.05 |
In total, 13 crosses between varieties were successful. Crossing type VirginiaxValencia was widely examined during the second year and 7 crosses between varieties were successful (Fig. 2). The best period for conducting crossing was found to be late June (15-30) followed by early July, since the percentage of successful crosses showed the highest values at this period (13-16%, Table 1). For late July and August, the percentage of successful crosses declined rapidly. Figure 3, presents the negative correlation between the time of crossing and the successful percentage of crosses (R2 = 0.98, significant at p<0.05), indicating an almost linear relationship.
Regarding transgressive segregation for the traits 100-pod weight and 100-seed weight, there were found significant differences between all parents and accessions (Tables 2 and 3). In cross type VirginiaxSpanish and occasionally in types VirginiaxValencia and ValenciaxVirginia there was found the highest number of accessions exhibiting transgressive segregation for both traits examined (Table 2 and 3). Concerning transgressive segregation of 100-pod weight, in cross VrasnaxIspaniki, the accessions IB-19-89 and IB-21-89 overyielded midparent values by 42-43 and 35-40% respectively in the F4 and F5 generations. The same was found for accession IB-38-89 in cross VrasnaxSakania that overyielded midparent values by 43-78% in all generations. Concerning transgressive segregation of 100-seed weight, in cross VrasnaxIspaniki, the accessions IB-19-89 and IB-21-89 overyielded midparent values by 13-52% in all generations. The same was found for accession IB-38-89 in cross VrasnaxSakania that overyielded midparent values by 39-70% in all generations.
| Table 2: | Comparison between parents and recombinant lines for transgressive segregation of 100-pod weight in peanuts |
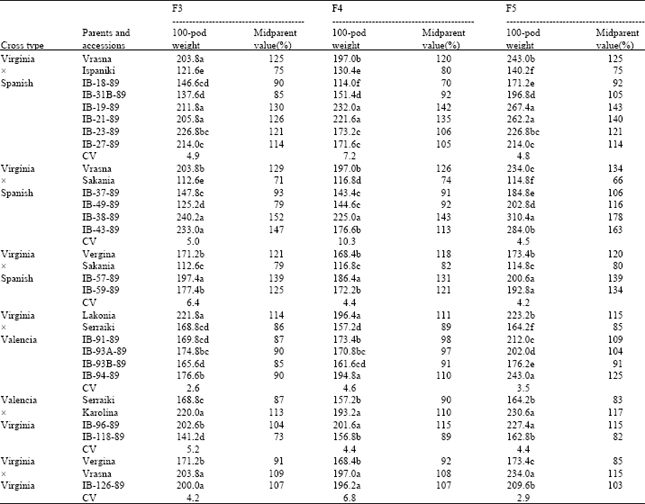 | |
| Means followed by the same letter within columns do not differ significantly at p = 0.05 (Duncan’s test) | |
| Table 3: | Comparison between parents and recombinant lines for transgressive segregation of 100-seed weight in peanuts |
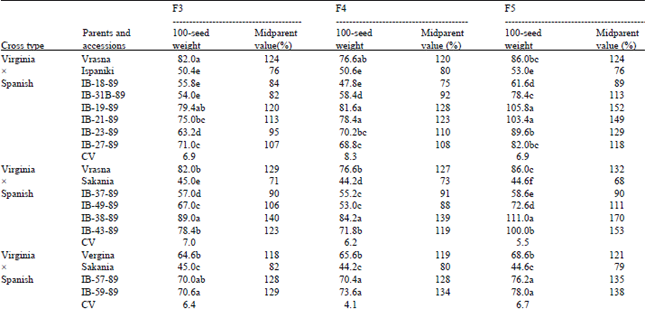 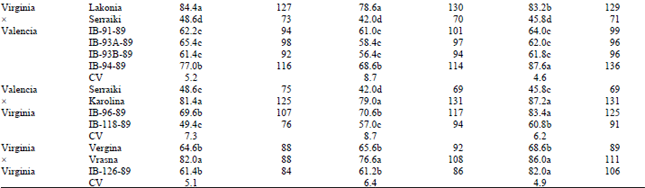 | |
| ContinuedMeans followed by the same letter within columns do not differ significantly at p=0.05 (Duncan’s test) | |
DISCUSSION
In both crossing years, varieties well adapted to the Greek conditions, early-maturing and high-yielding with variable characteristics and of different origin were used as parents in the crosses (Kotzamanidis, 1994). The selection of these parents is in accordance with the statements made by Briggs and Knowles (1971) and Simmonds (1979) who depicted that, excellent crosses more easily arise from high-yielding varieties of different origin, because their characteristics are controlled by various groups of genes complementing one another. We also took care that parents of different botanical type (between the two subspecies hypogaea and fastigiata, Krapovickas and Gregory, 1994) are crossed giving high heterosis to the progenies (Wynne et al., 1970; Wynne and Gregory, 1981; Qujada and Layrisse, 1985). Branching pattern of F1s is usually similar to the varieties from subspecies hypogaea and this trait is better described by a quantitative model than by a qualitative one (Qujada and Layrisse, 1985).
Regarding the proper crossing period, from the above results one can draw the following conclusions: The crosses in the peanut plants in our country have to be carried out as early as possible and preferably during the month of June when the temperatures are not so high. Indeed, as it is reported by Norden (1980), temperatures between 22 and 33°C are most ideal for flowering and fruiting of cultivated peanuts. Ono et al. (1974) noted that optimum soil temperature was 31 to 33°C (minimum were 15 to 17°C and maximum 37 to 39°C). DeBeer (1963) noted that plants grown at temperatures of 33°C produced pollen of low viability and that the temperature at 36-96 h preceding flower opening also influenced viability. During the month of July but also in August in our country the temperature can often reach even 40°C in the region (N.E. part of Greece) where the Institute is located and where the crosses were carried out (Fasoulas and Fotiadis, 1984). Another reason was, as reported by Norden (1980), that the percentage of successful hybridization is usually higher when the plants are young and in the early stage of flowering. Spanomitsios and Kotzamanidis (1997) reported that, higher temperatures (2°C on the average) from sowing to flowering caused a mean flowering earliness of about 4 days. Consequently high summer temperatures mature the fruits earlier. That was the reason why only a few crosses were successful, especially in August (percentage 3%). Even if we take into consideration that the crosses were carried out in the greenhouse where the temperatures are even higher, we can conclude that we must avoid carrying out crosses during the months of July and August.
The size of seed is described by the characteristic 100-seed weight and is a character of quality and commercial value of varieties (Knauft et al., 1991). Also, the size of pods is described by the characteristic 100-pod weight and is correlated to the yielding performance of peanut (Mohammed et al., 1978). Thus, these two characteristics are considered important parameters for peanut breeding. According to above mentioned results, transgressive segregation was found significant for both yield characteristics 100-pod weight and 100-seed weight and especially for cross type VirginiaxSpanish and occasionally in types VirginiaxValencia and ValenciaxVirginia. Consequently, as both yield characteristics showed transgressive segregation and are correlated to yield and quality, it means that peanut breeders may found accessions with useful characteristics by crossing varieties belonging to different cross types and especially in cross type VirginiaxSpanish.
ACKNOWLEDGEMENT
Author is grateful to Dr. C.G. Ipsilandis for his valuable suggestions and support on presentation of current study.
REFERENCES
- Fasoula, D.A. and V.A. Fasoula, 1997. Gene action and plant breeding. Plant Breed. Rev., 15: 315-374.
Direct Link - Wynne, J.C., D.A. Emery and P.W. Rice, 1970. Combining ability estimates in Arachis hypogaea L. II. Field performance of F1 hybrids. Crop Sci., 10: 713-715.
CrossRefDirect Link - Xiao, J., J. Li, I. Yuan and S.D. Tankley, 1995. Dominance is the major genetic basis of heterosis in rice as revealed by QTL analysis using molecular markers. Genetics, 140: 745-754.
Direct Link - Snedecor, G.W. and W.G. Cochran, 1980. Statistical Methods. 7th Edn., Iowa State University Press, Iowa, USA., ISBN-10: 0813815606, Pages: 507.
Direct Link








