Research Article
Chronic Effect of the Hydroalcholic Extract of Urtica dioica Leaves on Regeneration of ß-cells of Hyperglycemic Rats
Department of Embryology and Histology,
Vahid Khori
Department of Pharmacology,
Soraya Ghafari
Department of Embryology and Histology, Gargan University of Medical Sciences, Gargan, Iran
Anneh Mohammad Gharravi
Department of Embryology and Histology, Gargan University of Medical Sciences, Gargan, Iran









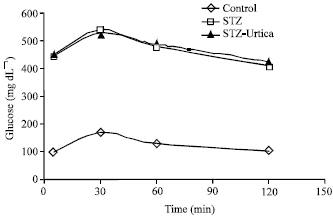
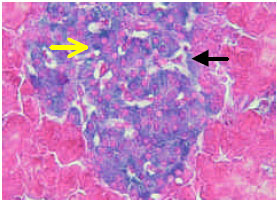
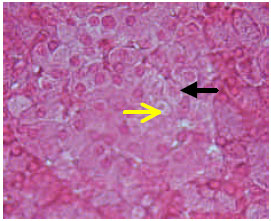
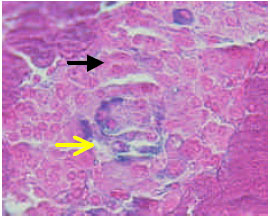
 ) beta cells (
) beta cells ( )]
)]