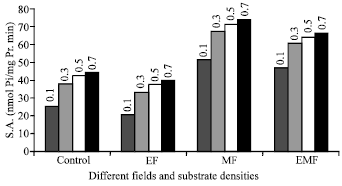
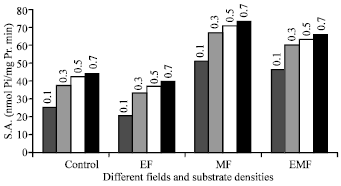
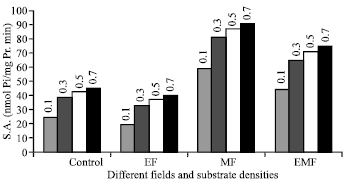
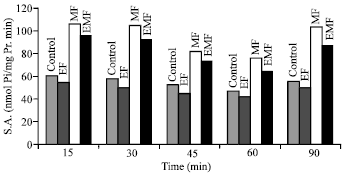
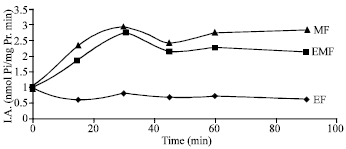
Research Article
Effect of Video Display Terminal Radiations on Ion-transporting Adenosine Triphosphatase in Brain
School of Physics, University College of Science, Tehran, Iran
Nazly Mahdavi
Institute of Biophysics and Biochemistry of University of Tehran, Tehran, Iran