Research Article
Anthracnose: An Economic Disease of Cassava in Africa
Not Available
A.G.O. Dixon
Not Available
T. Ikotun
Not Available
E.A. Tembe
Not Available
C.N. Akem
Not Available
R. Asiedu
Not Available
Cassava, Manihot esculenta Crantz (Euphorbiaceae) is a starchy root crop that is among the most important tropical food crops (Cock, 1985). It has the ability to produce economic yields under marginal production conditions, characteristic of Africa (Hahn and Keyser, 1985). However, in many cassava-growing areas its use as food helps to alleviate problems of hunger and carbohydrate intake deficiency, and thus its importance in terms of food security in these areas cannot be over-emphasized (IITA, 1990).
Total world production is estimated as 155.3 million tons of dry root weight (FAO, 1993). The recent FAO report also places Africa, as the leading producing region, with the production forecast reach to 71 million tons in 1994, which is 3% more than that reported in 1991. The leading producing countries in Africa includes, Zaire, Nigeria, Mozambique, Angola, Tanzania, Uganda, Central African Republic, Madagascar, Ghana and Ivory Coast (Table 1). This increase in production is due to the adoption by farmers of high yielding pest/disease resistant varieties under government support programmes, and the maintenance of restrictive cereal import policies (Nweke et al., 1994). Cassava constitutes the principal carbohydrate source for more than 800 million people in developing countries. About 80% of the cassava produced is consumed by humans, while the remaining 20% is used for animal feeds and agro-industrial purposes (Mc Cann, 1976., IITA, 1992).
Cassava is grown under very broad climatic and edaphic conditions. The plant is completely domesticated and shows a high degree of local adaptation. Cassava is a perennial crop and is mostly propagated by woody stem cuttings (CIAT, 1983; IITA, 1990). The large swollen true roots may be harvested 7 months after planting in warm areas. The multiplication of cassava through true seed is becoming important because of increasing breeding research for resistance to major diseases and other agronomic characters, which requires parent true seeds.
On dry matter basis, cassava roots contain 92.5% carbohydrate and 3.2% protein; starch and sugar predominate, comprising about 90% (Kawano, 1978 ; Hahn et al., 1989). The leaves contain 7% protein on a fresh weight basis and 20-35% on a dry weight basis, and are an important source of protein and essential nutrients for people who consume cassava products and leaves as a vegetable or in salads as soup (Table 2). Cassava leaves compare favourably with soybeans in protein quality and are considerably higher in lysine, although deficient in methionine and tryptophan (Jalloh and Dahniga, 1994).
| Table 1: | Annual area and yield yields of cassava in ten major producing countries of Africa. |
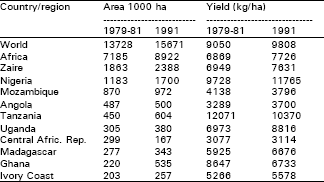 | |
| Source: FAO (1991). Production vol. 45 p. 94 Importance of country based on 1979-81 figures | |
| Table 2: | Dry matter, carbohydrate and protein content of roots and tubers. |
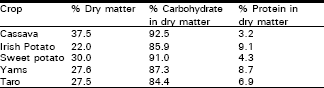 | |
| Source: IITA (1992). | |
Cassava products traditionally show variable composition in terms of calories, proteins, lipids, total carbohydrates and essential minerals (Table 3).
Economic difficulties in developing countries of Africa, have stimulated policy makers to re-evaluate native crops as substitute for foreign food imports. These policies have enhanced rapid expansion of cassava cultivation, with concurrent increase in international exchange of planting materials within National research programmes over the past 20 years. Large monocrops cassava plantation are on the increase as a source of revenue generation and with this change, pathological and entomological problems are on the increase, causing heavy loss in production.
Cassava anthracnose disease (CAD) is one of the major economic diseases of cassava in the tropics. The disease has attained epidemic levels in some high rainfall regions of Africa. This has led to the growing interest in the investigation of disease control strategies in many agricultural research programs.
| Table 3: | Composition of cassava products prepared traditionally in Cameroon*. |
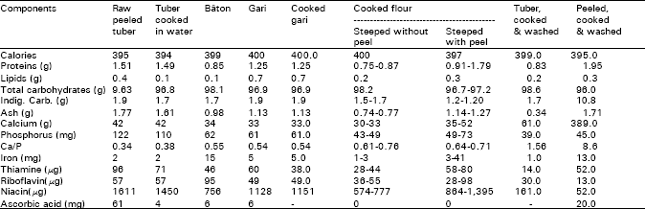 | |
| * Per 100g dry matter Source: Favier et al. (1971). | |
 | |
| Fig. 1: | Cassava field plot showing close canopy effect of leaves creating humid conditions, that favours Cassava anthracnose disease development |
Symptoms: CAD is characterized by particular symptoms (cankers on stems, branches and fruits, leaf spots and tip die back), on aerial parts of the diseased plants (Muimba, 1982). The appearance of the disease depends on the cassava varieties and the infected plant. Cassava production on a commercial scale is grown on large fields or plantation. The leaves develop and spread forming a close canopy (Fig. 1), which creates very humid condition favorable for CAD development and spread in the field.
On cassava leaves CAD is characterized by numerous small circular sunken spots on the leaf lamina measuring 10-30mm in diameter (Makambila, 1987). When the disease is fully developed, the center of the spot is white and studded with pinkish fruit bodies of the fungus (Theberge, 1985; Hahn et al., 1989b). Petiole infection is initiated by hyphae growing from the infected lamina through conducting tissues. In some susceptible cassava cultivars, leaf blight, dramatic wilt and defoliation may occur (Fig. 2). The disease could spread towards the top and then induces tip die back. CAD incidence on young cassava stems is characterized by oval pale-brown shallow depressions bearing a spot of normal green tissue in the center. The necrotic spots usually starts on the nodal zones, with a diameter of about 10-20cm from the top part of the plant. In older cassava stems, CAD infection usually occurs as round and stringy lesions, which develop into large canker (Fig. 3). Stem deformation occurs in some cultivars, causing the stem to be brittle and easy to break by wind action. The deeper cankers sometimes affect the pith of the plant thus blocking transportation of vital materials to part of the plant (Van der Bruggen and Maraite, 1987). Some resistant cassava genotypes usually recover from CAD infection by sprouting new twigs from the axillary buds at the lower edge of necrosis, producing a bayonet-like appearance.
Etiology: The causal agent of CAD is a weak pathogen Colletotrichum gloeosporioides f.sp. manihotis Henn (Penz).
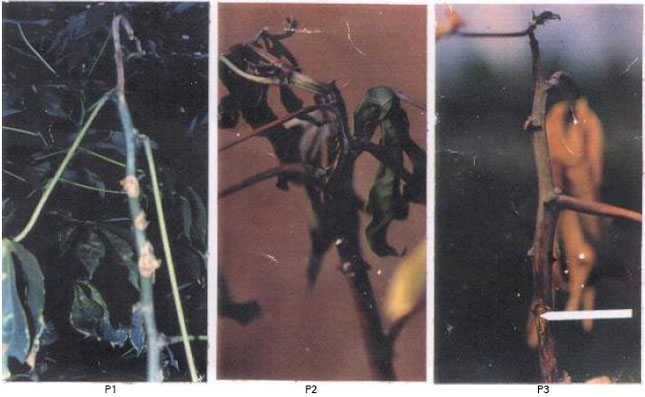 | |
| Fig. 2: | Cassava anthracnose disease shoot symptoms: P1= Young cassava plant with shoot die-back symptoms., P2= Cassava seedling with shoot die-back and wilt symptoms., P3 =Cassava seedlings showing defoliation, shoot die back and wilt symptoms. Arrow shows point of infected leaf defoliation. |
The fungus is best characterized by its relatively short straight cylindrical to broadly ellipsoidal conidia, with obtuse ends and truncated attachment points (Jeffries et al., 1990). Under natural conditions C. gloeosporioides f.sp. manihotis penetrates the host through mechanical wounds or sucking insect vectors (Pseudotheraptus devastans, Dist (Het. Coriedae) punctures (Boher et al., 1983). The insect P. devastans, is a cored bug associated with CAD. The life cycle consists principally of egg, five instar nymph stages and adult. They are common in young cassava fields found in most parts of the tropics where intensive cassava farming is practiced. These insects usually appear in fields during the cassava planting seasons, between May and August, when wet and high relative humidity conditions favour their growth and reproduction (Fokunang et al., 2000a). In the case of CAD pathogen penetration after insect puncture of young cassava stems, the cracks extend through the cork layer or the epidermis up to the underside of the sclerenchyma, then immediately the lesion extends tangentially under lignified fibers (Makambila and Bakala-Koumonou, 1982). The host reacts to the invasion through the generative zone, the layer of cork more precociously formed at the level of the initial necrotic lesion than in the primary healthy stem (Van der Bruggen and Maraite, 1987).
Individual genotypes of the fungal pathogen can differ from each other in many inherited characteristics, such as the morphology, physiology and pathogenicity (Tjamos et al., 1993). New races of the pathogen can arise by mutation in somatic cells, recombination of nuclear genes during sexual reproduction, by reassortment or exchange of genetic material in somatic cells and by mutation of extrachromosomal or cytoplasmic genes (Hahn et al., 1989b; Suresh et al., 1990). C. gloeosporioides f.sp. manihotis is a specialized fungus on cassava host, and sporulation and germtube development of the physiologic races of the fungus is highly correlated with pathogenicity and virulence on the cassava host plant (Fokunang et al., 2000b). The growth potential of the fungus is highly influenced by the growth media, pH and temperature (Fokunang et al., 1995).
Epidemiology: CAD is one of the important cassava disease frequent in cassava growing regions of Africa (Hahn and Keyser, 1985), South America (Lozano et al., 1981) and Asia (Chadrasekharan-Nair et al., 1979). The importance of the joint action of the puncture by an insect Pseudotheraptus devastans. Dist, and the infection by the fungus in the development and spread of the disease has been established (Muimba, 1982; Boher et al., 1983; Fokunang et al., 2000a). The fungus attacks tissues, weakened by the insect puncture and lytic action of the saliva, thus inducing dark-brown lesions. These lesions can either remain limited, or becomes deep light brown cankers with lacerated epidermis (Theberge, 1985).
The pathogen survives environmental conditions in its perfect state Gloeosporium cingulata that appears to increase during the dry season. Dissemination of CAD and the geographical distribution may also depend upon climatic factors such as rainfall, relative humidity, temperature and wind action. Planting time, temperature, relative humidity rainfall, has a significant influence on the level of CAD inoculum pressure, vector developmental stages under field conditions (Fokunang et al., 1999a). CAD survival and transmission is also possible through breeder seeds and post harvest debris in the field. (Fokunang et al., 1997). Locally, water splash, air current, insect or other forms of contact can disperse it.
C. gloeosporioides f.sp. manihotis generally attacks its host more frequently during the prolonged periods of rainfall.
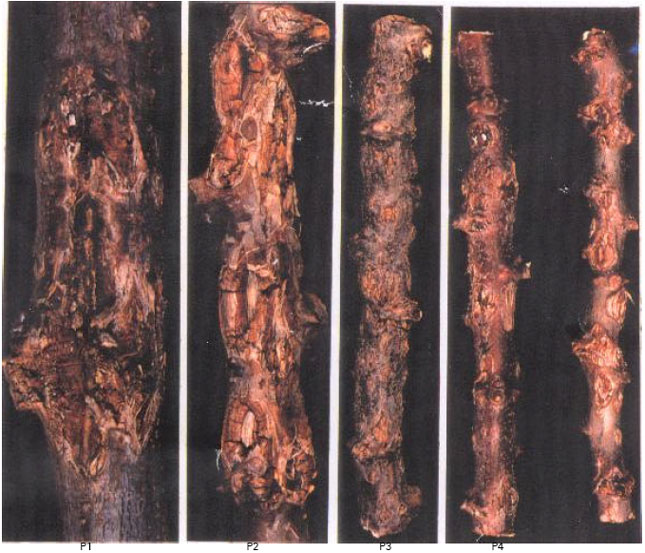 | |
| Fig. 3: | Cassava stem pieces showing different forms of CAD cankers: P1= Large deep expanding cankers., P2 Crocodile-like deep anthracnose cankers., P3= superficial invading cankers., P4= Non expanding superficial cankers |
New leaves appearing in the small rains are mostly affected but the disease tends to be less severe on the approach of the dry season (Muyolo, 1984). The passage from one small wet season to a prolonged dry season initiates modification in physiological activities of the cassava plants, thus making them most susceptible to CAD.
Relative humidity plays an important role in the developmental stages of P. devastans and tissue damage, especially in the morning when the insect is very active in feeding (Boher et al., 1983). Rainfall regime plays an important role in the development and dispersal of CAD pathogen. The rain splash dislodges spores from the acervuli and spread them along the young cassava stems. Temperature range of 25"2EC is suggested as optimum range for the fungal development (Fokunang et al., 1995).
Losses: Accurate assessment of yield loss due to CAD has not been quantified especially due to the problem of occurrence of multiple disease complex in most cases with cassava bacterial blight during severe conditions in the wet season. However, in 1975, 90% of local cassava cultivars in Zaire were rated as severely affected (PRONAM, 1977). Muyolo (1984) also reported more than 80% cassava plants in Congo severely infected by CAD in Mbe and Odziba regions of Congo. There are also many unreported cases of local farmers= fields and experimental plots severely invaded by CAD in rainfall cassava growing zones of Eastern Nigeria. Although there is no quantification in yield loss, a strong negative correlation between CAD incidence and severity with yield parameters (storage root weight, tuber root number and percentage root dry matter) has been established ( Mordue, 1971; Fokunang et al., 2000c).
CAD severity could lead to a significant loss in planting materials and low biomass production (Hahn et al., 1989a). Severely infected cassava stem in some cases results in 40-60% germination rate, and increase in the period of stem sprouting of 7-8 days as compared to disease-free planting materials. Infected seed materials can lead to 50-75% loss in seed viability and low seedling germination of 20-40%. Total crop failure has been reported in cases, where infected stem cuttings have been used as planting material (Ikotun and Hahn, 1992).
Control Strategies: Little attention has been made in the development of control measures for CAD because the fungus was considered a weak pathogen, and its importance was overwhelmed by sudden outbreak in two other major diseases, -African cassava mosaic virus and cassava bacterial blight. However, devastating incidence and severity of CAD due to high inoculum pressure in both farmers and research fields has led to investigation of some integrated disease management systems (IDMS), such as: cultural control, chemical control, varietal resistance and quarantine/sanitation measures.
Cultural control: These measures are implemented to reduce or eradicate the spread of the pathogen. Crop rotation, deep burial or burning of infected plant debris, fallow has been shown to reduce inoculum build up, especially in fields where disease-free planting materials are used for the new planting season (Onyeagba et al., 1978; Lozano et al., 1981). The pathogen cannot survive in the field for more than one farming season; thus fallowing can reduce carry-over effect of CAD in the field, and also enhance the pathogen elimination.
Planting time could be manipulated to reduce CAD incidence and severity in intensive cassava growing regions. This is possible since cassava is ideally planted at the beginning of the rainy season. After a few rains, the plants establish, but the conditions are very favorable for infection and disease spread. By planting towards the end of the wet season, there is reduction in inoculum propagules of the disease. By the onset of dry season, plants usually have accumulated pectin, lignin and cellulose on the cell walls, which confers resistance of the plant to pathogen penetration. Due to rain and high temperature, there is also an increase in plant growth and inoculum potential, but the inoculum is not very effective to the older plants, which are due harvesting in only a few months. Clean planting material is essential and effective in producing anthracnose disease-free cuttings for propagation (Ikotun and Hahn, 1992).
Chemical control measures: This control measure for CAD has not yet been well established. However, Lozano et al., (1981); Theberge, (1985) and Roger (1990) suggested spraying plants with copper fungicides in cases where resistant stocks were not available. Further investigation in the area of chemical control has been slow due to the rising cost of production and sales of limited fungicides available in the market, which the poor subsistent farmers are unable to purchase.
Alternative control measures with anti-microbial plant products such as neem plants Azadirachta indica (A. Juss), for pre-treatment of planting material has been a promising preventive control practice (Fokunang et al., 1999b). These natural plant products are not only readily available to the farmers, but are biodegradable, environment friendly and very promising within the framework of integrated pest and disease management systems (IPDMS).
Varietal resistance: The breeding for resistant varieties appears to be the most efficient means of controlling CAD. Research work on the resistance of cassava genotypes to CAD, did not make much progress until Muimba (1982) who demonstrated a relationship between CAD and a sap-sucking insect P. devastans. A major part of the breeding and selection work at the Tuber and Root Crop Improvement Programme (TRIP), International Institute of Tropical Agriculture (IITA), Nigeria is on the development of resistant lines to major economic diseases: anthracnose, bacterial blight and mosaic virus. The project also aims at improving cultivar resistance in a wide range of environmental conditions, and for a long period in view of achieving stable productivity.
Screening methodologies have been developed using stem puncture techniques, which induce necrotic lesions similar to those observed in the field, thus making it possible for evaluation of resistance of cassava genotypes in expanding field conditions (Muimba, 1982; Ikotun and Hahn, 1992). The screening test for CAD resistance has contributed in the recommendation of some resistant improved cassava genotypes possessing some desirable agronomic traits for use as resistant parent materials for breeding trials. The field screening methods for CAD resistance is generally based on phenotypic expression of the disease symptoms by the plants that are affected by the fungus. The economic value of resistant cultivars is roughly equated with the saved cost of fungicides, time, labour and any other risks involved. However, the main problem in using resistant varieties is the possibility of the pathogen overcoming the resistance, and the duration involved in breeding resistant lines. This suggests the necessity for a continuous breeding and evaluation study of disease resistance, and looking at the long-term genotype by environment interaction in broader agro-ecological zones.
Cassava resistance to CAD has been associated to phenol metabolic changes, which is a common reaction with many plants (Friend, 1981). Increased resistance to CAD is also associated to increase peroxidase activity around the dark brown lesions formed. The peroxidase participates in the last stage of lignin synthesis and plays a role in the development of brown margin, which is a defense mechanism of the plant against the pathogens (Van der Bruggen and Maraite, 1987). Lignin and peroxidase production in casssava resists an extension of CAD infection by confinement of germ tube or appresorial development of the cell initially penetrated (Jeffries et al., 1990). Lignifications in cassava at the onset of tuberization, plays a role in cassava resistance, a period very favorable for CAD pathogen growth and development (Fokunang et al., 1999b). Other sources of CAD resistance in cassava are their ability to release toxic cyanide from non-toxic complexes. This cyanide is toxic to most of the microorganisms by interfering with their mitochondria respiratory process, thus imposing resistance to pathogen invasion (Asiedu et al., 1992).
Quarantine/ Sanitation measures: Major means of dissemination of cassava diseases and pests have been from the exchange of cassava stem cuttings for propagation. The origin of CAD has not been reported but its ubiquitous nature in the cassava growing regions is an indication of exchange through distribution of infected materials. Recent report shows that anthracnose is seed-borne and can be transmitted by infected cassava seeds (Fokunang et al., 1997). Several sanitation measures, in addition to those legally established by quarantine regulations could reduce the risk of disseminating the disease by means of propagating material (Lozano et al., 1981 ). The recipient country should be cautious in accepting stock planting materials from countries or regions with reports of high inoculum pressure of the pathogen.
Vegetative or sexually propagative materials should be collected only from healthy plants in the fields apparently free from CAD symptoms. Before such material is collected, a field's overall sanitary condition should be determined by more than one inspection during the peak of cassava cultivation to the end of the wet seasons. Any abnormal seeds or cuttings should be discarded. Particular care should be taken when introducing cassava seed that serve as an important breeding tool for cultivar resistance and inheritance studies.
Pre-treatments with hot water at 60 EC, for 15-20 min or fungicides seed dressing with benlate, captan has also been recommended for CAD control. Recently there has been some progress in the use of antimicrobial plant products such as neem seed extracts (Azadirachta indica A. Juss), Bitter leaf (Vernonia amygdalina. Del) and Ocimum gratissimum. L in reducing CAD pathogens in cassava seeds (Fokunang et al., 1999b).
Cassava Multiple Disease Resistance Research at the International Institute of Tropical Research IITA, Nigeria: Through collaborative studies involving cassava breeders, pathologist, entomologist and biochemist, some cassava cultivars have been selected through intensive breeding and screening of the cultivars at preliminary yield trial (PYT) and advance yield trial (AYT) stages for disease and pest resistance. Some cultivars such as TME 30001, 30211, 91/00684, 91/00313, have been established as resistant to CAD, and further screening for CAD resistance is on-going, at different agro-ecological regions, through exchange of planting materials. Molecular approach in disease diagnosis, characterization of physiological races of the pathogen using Polymerase Chain Reaction (PCR) techniques, is in progress at IITA. This is aimed at improving the diagnostic approach of CAD, especially in latent infection conditions of the disease, and possible advancement in the implementation of new control options through an environment friendly disease and pest control strategies.
CAD pathogen control on cassava genotypes is of prime importance in the sustainability of food crop production for the poor subsistent population in developing countries, where cassava is a staple crop. With progress in integrated management control strategies being introduced in most National research programs, there is still the need for extension workers to transfer these control strategies to be implemented in areas where CAD is endemic. The goal of all cassava research is geared towards the preservation of stable productivity system since yield stability is crucial for cassava growers. This can be achieved by defining the diverse approaches to disease control, then introducing these approaches into production realities, conditioned by the environment, the cultivars, the socio-economic status of the producers and consumers in the target zones.