ABSTRACT
Twenty species from 13 genera were identified from leaf surfaces of onion plant (Allium cepa L.) on 1% glucose- and 2% cellulose-Czapek’s agar at 25°C. The microbial phyllosphere decreased through out the growth period on the two isolation media. While, the count of phylloplane fungi increased with the increase in plant age till February and decreased till the end of the experimental period. The most prevalent fungi were A. alternata, A. niger, A. sydowii, A. versicolor, C. herbarum, C. lunatus, P. herbarum, S. rostrata and U. botrytis on the two types of media. The prevalent fungi were screened for the antagonistic activity against the pathogenic fungus P. herbarum In vivo and In vitro. The culture filtrates of A. alternata, A. niger, C. lunatus and E. nidulans caused high inhibition of P. herbarum. Colony interactions in solid cultures indicated that maximum inhibition of P. herbarum was caused by A. alternata, A. niger, C. lunatus, S. rostrata and S. chartarum. Treatment of infected leaves by surface fungi significantly inhibited the lesion development caused by the pathogen, especially filtrates of A. alternata, A. niger, C. lunatus and U. botrytis. Volatile produced from the most fungal cultures have no significant inhibition on the growth of the fungal pathogen. Forty five fungal isolates were screened for their ability to produce chitinase enzyme. About 80% of the isolates could produce this enzyme with variable degrees of chitin hydrolysis.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2001.838.842
URL: https://scialert.net/abstract/?doi=pjbs.2001.838.842
INTRODUCTION
Aerial plant surfaces provide a suitable habitat for epiphytic micro-organisms which are influenced by the nutrients present on the leaf surfaces (Fokkema, 1976; Pandey et al., 1993). Chemical pesticides are generally effective in reducing the density of pathogens but have many undesirable attributes (Lima et al., 1997). The possibility of controlling the pathogenic fungi by antagonistic microorganisms has been explored by various workers (Blakeman, 1985; Mercier and Reelender, 1987; Pandey et al., 1993). Several modes of action have been associated with the ability of saprophytic fungi, to control plant pathogens and promote environmental safety, including substrate competition, antagonism by antibiotics or parasitism by cell wall degrading enzymes or by a combination of these modes of action (Perez-Leblic et al., 1985; Chet, 1987; Whipps, 1992; Roulston and Lane, 1988; Widden and Scuttolin, 1988; Boland, 1990; Be Langer et al., 1995).
Onion (Allium cepa L.) is extensively grown in Egypt as well as in many parts of the world. It is subjected to several destructive diseases; such as neck rot, onion smut, pink rot and downy mildews. Pleospora herbarum was the second fungus, infecting onion leaves (spot disease) causing heavy loss in onion crop. For controlling this disease, growers depend mainly on chemical treatment, but resistant varieties of this pathogen could greatly reduce the effect of such chemical control. So, biological control has been attempted to control the spread of the mould diseases (Ouf et al., 1997; Paul et al., 1997).
In Egypt, little work has been done on the antagonism between the pathogens and leaf surface fungi. The present work was designed to study the distribution of saprophytic fungi associated with the leaf surfaces of onion plants and the antagonistic activities of these fungi against the pathogens of onion in vitro and in vivo.
MATERIALS AND METHODS
Monthly samples of onion (Allium cepa L.) leaves (about 200 g) were collected randomly during January to April 1999 from onion plant fields in the Botanical Garden of Faculty of Science, Assiut University. The samples were placed in sterile plastic bags and transferred immediately to the laboratory for fungal analysis.
Isolation of phyllosphere fungi: Small pieces of the leaves were cut out using sterile scissor. Ten g of leaf segments were used for fungal isolation, employing the dilution plate-method (Johnson and Curl, 1972). One ml of the dilution was transferred into sterile Petri-dishes, which were then poured with melted but cooled agar medium. 1% glucose- and 2% cellulose-Czapek’s agar media were used. Six plates were used for each sample (3 for each medium) and incubated at 25°C for 7-10 days. The growing fungi were identified ( based on macro and microscopic characteristics ), counted and calculated per fresh gram leaves.
Isolation of phylloplane fungi: After shaking several times in sterile distilled water the cut pieces of leaves were removed and thoroughly dried between sterilized filter papers. Four pieces were placed on the surface of the medium in each plate. Plates were incubated at 25°C for 7-10 days. The developing fungi were counted, identified and calculated per 12 segments in each sample.
Effect of culture filtrates of antagonists on the pathogen: The predominant fungi isolated from onion leaves were tested for their antagonistic activities towards the pathogen Pleospora herbarum (Anamorph= Stemphylium botryosum). Tested fungi were maintained individually on Czapek Dox yeast extract agar (CDYA) at 25°C. One agar block (1 cm2 diameter) of each fungus was cut from the active growing margins of 10 days old cultures, were separately inoculated into 250 ml Erlenmeyer flasks containing 100 ml sterile CDY liquid medium. Flasks were incubated in a shaker at 25°C for 10 days, the cultures were filtered through filter paper No. 1. One ml of culture filtrate of each fungus transferred to sterile Petri-dishes in triplicate and the plates were poured by 20 ml of autoclaved and cooled (40°C) CDYA medium were poured in each dish . Medium devoid of culture filtrates served as control. Petri-dishes were inoculated with a 1 cm2 agar block of pathogenic fungus (3 discs each plate) and incubated at 25°C. The radial growth of the pathogen colony was measured after 7 days.
Colony interactions In vitro: In this experiment, petri-dishes (duplicates) containing sterile and solidified CDYA medium were inoculated with two agar blocks (one antagonist and the other is the pathogen) adjacent to each other 3-5 cm apart. The plates were incubated at 25°C for 7 days. Interactions were scored by a key based on the methods and classification of Skidmore and Dickinson (1976). The inhibition (%) in radial growth was calculated by the formula:
100% (r1-r2) r1
where, r1 denotes the diameter of the pathogen on the opposite site, and r2 represents diameter of the pathogen from the point of inoculation towards the interacting site of the saprophytic fungi.
Interactions on the leaf surfaces: Interaction between the pathogen and the saprophytic fungi were studied on the leaf surfaces by measuring the lesion development produced by P. herbarum. Healthy leaves of onion plant ( aged 26 days ) were scratched on the upper surface with a sterile needle and a standard size drop (0.1 ml) of conidial suspension ( about 104 spores /ml) of each of the pathogen and the saprophytic fungus tested were placed onto the scratches (3 replicates of each tested fungus). Leaves were separately inoculated with an equal size drop of spore suspension of the pathogen without the spores of antagonists (as control). Inoculated leaves were covered with sterile plastic bags containing wet sterile cotton swab to maintain high humidity inside the bags. After 20 days, the lesion size of the treated and control leaves were measured. Percent inhibition in lesion development was calculated by the formula:
100% (I1 – I2) I1
where: I1 is the lesion size of leaves inoculated with the pathogen, and I2 denotes the same inoculated with the pathogen plus saprophytes.
Effect of volatile substances: Plates, each containing 20 ml CDYA medium were inoculated with agar block of each saprophytic fungus in triplicates. After incubation for 7 days, the lid of each petri-dish was replaced by the bottom of another petri dish containing 20 ml CDYA medium, pre-inoculated with agar block of P. herbarum, and incubated at 25°C for 10 days. For a control the lids of the pathogen were maintained. Radial growth and inhibition of the pathogen was calculated.
Chitinase production: Forty-five isolates of fungal species isolated in the current study were grown on a basal glucose-Czapek’s agar medium of which glucose was replaced with 1% chitin of practical grade from crab shells. Fungal inoculum were inoculated in the centers of plates (3 inoculum each plate) containing 20 ml of the used media. After the growing periods the clear zones around the fungal colonies were measured.
RESULTS
Counts of phyllosphere fungi of onion plants regularly fluctuated between 2.5-13.4 and 1.8-7.5 colonies/g fresh weight leaves on glucose- and cellulose-Czapek’s agar at 25°C, respectively. The highest count was occurred in January and then decreased gradually showing the lowest count in April on the two media used (Table 1). Twenty species representing 13 genera were identified from the leaf surfaces of onion plants on plates of glucose- (18 species and 11 genera) and cellulose-Czapek’s agar (15 and 11) at 25°C. Alternaria alternata, Aspergillus niger, A. versicolor, Cladopsorium herbarum, Cochliobolus lunatus, P. herbarum and Setosphaeria rostrata were recovered during all months. Also, the counts of these species regularly displayed giving the best counts during January or February (Table 1). Some species were isolated only on plates of glucose-agar but of rare occurrence or non on cellulose agar, such as: Aspergillus flavus, A. fumigatus, A. sydowii, C. oryzae, E. nidulans, G. fujikuroi, R. stolonifer and U. botrytis. Some others were of opposite pattern such as: C. cladosporioides, M. echinata and S. chartarum (Table 1).
Table 2 shows that the counts of phylloplane fungi of onion plants on glucose- and cellulose-Czapek’s agar ranged, respectively from 6-12 and 7-13 colonies/cm2 of leaf surface and the highest counts were achieved at February and the lowest at the end of the experiment. Nine species appertaining to 7 genera were isolated from the phylloplane of onion plants on the two isolation media (Table 2). Fungi of the dark mycelia represented the highest incidence such as: C. herbarum, C. lunatus, P. herbarum and S. rostrata. They were recovered in all months and showed their peaks during different months. The remaining species were less common (Table 2). The results showed that the numbers of genera and species recovered from the phylloplane (7 genera & 9 species) were lower than their respectives in the phyllosphere (13 & 20). This means that there are several fungi which are not realy inhabitants of the leaf surface but are deposited from the air.
Ten species were screened for their ability to suppress Pleospora herbarum In vitro and In vivo (Table 3). The results indicated that the efficiency of the culture filtrates of the different species tested was variable. The majority of the species (6 species) showed high effect in reducing the growth colony of the pathogenic i.e., filtrates of A. alternata, A. niger, C. lunatus and E. nidulans with radial growth inhibition from 49.2-55.4% while, A. sydowii, S. rostrata and S. chartarum (27.7-30.8%) showed moderate effect, and the remaining were of the least effects (Table 3).
Colony interactions in solid cultures were tested between the pathogen (P. herbarum) and each of the prevalent leaf surface fungi. The radial growth of the pathogen was significantly reduced by most of the species tested (Table 3). The most pronounced inhibition was observed by A. alternata, A. niger, A. sydowii, C. lunatus, and U. botrytis ( 27.6- 48.4% ) . E.nidulans, S. rostrata and S. chartarum were less effective on the growth of the pathogen ( 17.4- 22.6 % ), while A. versicolor and C. herbarum were the least effective (Table 3). Treatment of infected leaves with propagules of leaf surface fungi significantly inhibited the lesion development caused by the pathogen. The culture filtrates of A. alternata, A. niger, C. lunatus, E. nidulans and S. rostrata were the most effective in reducing lesion size ( inhibited lesion development from 48.3-62.4 %), Whereas moderate inhibitory effect was obtained with the spores of A. sydowii, S. chartarum and U. botrytis, but A. versicolor and C. herbarum showed less inhibitory effects.
Volatile metabolites produced by the common saprobic leaf surface fungi have visually non significant inhibition on the colony growth of fungal pathogen. But, most pronounced inhibition in radial growth of the pathogen was observed by volatiles of A. alternata, A. niger, S. rostrata, U. botrytis and E. nidulans. The remaining tested fungi showed no effect on the radial growth.
Lytic enzyme was examined by screening fungal isolates on agar medium containing 1% colloidal chitin and 80% of the isolates produced substantial chitinolytic enzyme in chitin-containing medium within 10 days (Table 4).
| Table 1: | Counts (per g fresh leaves) of phyllosphere fungi of Allium cepa during the periods January–April 1999 on 1% glucose and 2% cellulose-Czapek’s agar at 25°C. |
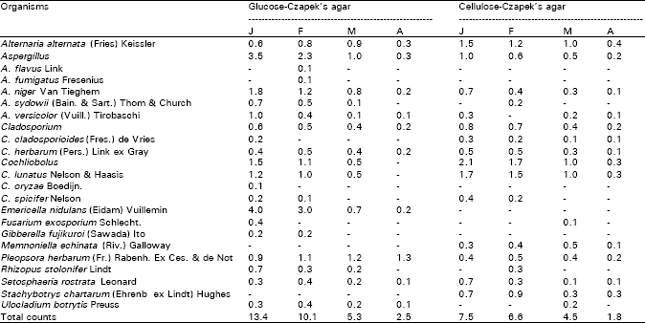 | |
| Table 2: | Counts (per square cm. of leaf surface) of phylloplane fungi of Allium cepa during the periods January–April 1999 on 1% glucose and 2% cellulose-Czapek’s agar at 25°C. |
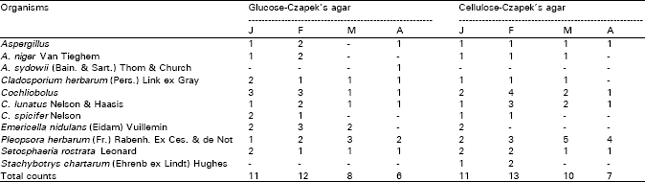 | |
| Table 3: | Interactions between Pleospora herbarum and common phylloplane fungi of onion in vitro and in vivo, on 1% glucose-Czapek’s agar at 25°C. |
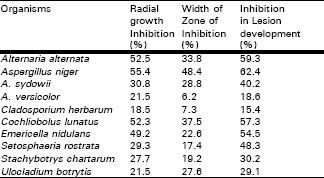 | |
Most isolates (16 isolates, 44.4% of total) of A. alternata, A. niger, A. sydowii, A. versicolor and S. rostrata produced higher amounts of the enzymes, while 14 isolates (38.9%) of C. herbarum, E. nidulans and U. botrytis exhibited moderate enzyme production.
| Table 4: | Chitinase production by common leaf surface fungi. |
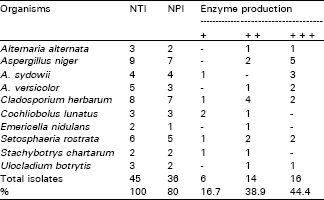 | |
| NTI = number of tested isolates; NPI = number of positive isolates. + = weak producers; ++ = moderate producers; +++ = high producers. | |
On the other hand, weak amounts of the enzyme was achieved by 16 isolates of C. lunatus and S. chartarum (Table 4).
DISCUSSION
At early stages of plant development, leaf surfaces phyllosphere fungi colonization was higher than at late stages. This could be due to the increase of the superficial mycelial growth of the pathogen which was probably due to nutrient competition, consequently reducing the number of saprobic sites. A. alternata, A. niger, A. versicolor, C. herbarum, C. lunatus, P. herbarum and S. rostrata. In this respect, Nicoletti and Rinaldi (1993) made a survey of the mycoflora on the leaf surface of olive and could isolate 23 fungal species. Among these fungi: A. alternata, C. sphaerospermum, Phoma spp., Aureobasidium pullulans were the predominant. They noticed that there was no important difference in the composition of the mycoflora of the leaf surface in relation to the level of infection by Spilocaea oleagina, although reports in the literatures have indicated that some of the species recorded can play an antagonistic role against certain fungal phytopathogens. Studies on the incidence of fungi on aerial parts of some plants appeared that A. flavus, A. niger, F. moniliforme (G. fujikuroi), and F. solani (N. haematococca) were the predominant. Whereas, other fungi were found rarely, such as: A. ochraceus, C. cladopsorioides, Penicillium spp., T. viride, R. stolonifer and Pestalotia spp. (Bagy and Hemida, 1992; Kurakov and Kostina, 1997).
The results showed that numbers of genera and species recovered from the phylloplane were lower than their respectivas in the phyllosphere, which indicated that there were several fungi which were not real inhabitants of the leaf surface but were deposited from the air. The present observations revealed that fungi of the dark mycelia were of highest incidence among the fungi collected. In this respect, Adhikari (1990) examined the mycoflora associated with three grasses and noticed that Cladosporium, Fusarium and Rhizopus were successful colonizers of the leaf surface of all the three grasses. Also, Kurakove and Kostina ( 1997) showed that among the microorganisms populating the root surfaces of all plant species tested ( including onion) were representatives of Mucor, Fusarium, Trichoderma, Aspergillus and mycelia sterilia and the frequent instances of microscopic fungi belonging to Gliocladium, Acremonium, Cladosporium, Alternaria and Penicillia of the section Biverticillata.
The present data revealed that, there were various degrees of inhibition in the growth of the test pathogen. Dennis and Webster ( 1971 ) reported that these differences were due to the volume of the culture filtrates, metabolic products, antibiotic secretion, nutrient improvement and pH alteration in the medium. Also, successful experiments have been carried out with the antagonistic fungi which were used as biological control such as: Trichoderma (Gullino and Garibaldi, 1988); Gliocladium roseum and Penicillium spp. (Sutton and Peng, 1993); Ulocladium atrum (Kohl et al., 1995). Present results are nearly similar to those obtained by Pandey et al. (1993). They noticed that filtrates of A. niger, F. exosporium, P. citrinum and C. roseo-griseum caused more than 50% growth inhibition of C. glycosporioides and P. psidii.
Colony interactions between the pathogen and the most common leaf surface fungi revealed that the radial growth of the pathogen was significantly reduced by most of the species tested . Similar observations were reported by Pandey et al. (1993). They noticed that, different grades of colony interactions in dual cultures were recognized between the two pathogens and the phylloplane fungi examined.
The development of leaf pathogen is affected by the common saprophytes if both are present at the same infection site. Similarly, Parashar et al. (1992) studied the biological control of bacterial blight of clusterbean by epiphytes present on leaf surface and noticed that all the antagonists reduced the incidence of the disease when used as seed dressings or spray treatments. Culture of filtrate sprays of P. citrinum, A. niger and the white sterile fungus were also effective in reducing the disease incidence and P. citrinum provided the greatest reduction. Also, Pandey et al. (1993) reported that application of a spore suspension of A. niger, F. exosporium and P. citrinum inhibited lesion development of guava leaves caused by the test pathogens in vitro. Also, when the suspension from F. graminearum was sprayed directly to some plants, it effectively controlled rice blast, rice brown spot and barley powdery mildew with >80% disease control (Kim et al., 1995). Thus, the possibility of controlling the pathogenic fungi on aerial plant surfaces by antagonistic microorganisms has been explored.
Volatiles produced from the cultures of A. niger, F. exosporium, P. citrinum and P. oxalicum inhibited the growth of C. gloeosporioides, whereas, volatiles from C. roseo-griseum, F. exosporium and T. harzianum inhibited the growth of P. psidii ( Pandey et al., 1993). Also, Kim et al. (1995) studied the antifungal substances of F. graminearum isolated from the phylloplane of tomato plant, against 22 pathogenic fungi and observed that the substance completely inhibited the growth of Pyricularia oryzae, Cochliobolus miyabeanus and Alternaria solani. Differences in the antagonistic behaviour of phylloplane fungi in vitro and in vivo have been observed by several workers (Bhatt and Vaughan, 1968; Pace and Campbell, 1974; Rai and Singh, 1980).
Eighty percent of the isolates produced substantial chitinolytic enzyme in chitin-Containing medium within 10 days. A number of fungal isolates are well known to be the producers of hydrolytic enzymes ( chitinase and protease) such as Trichoderma spp. (De la Cruz et al., 1992; Lima et al., 1997) and some other fungi (Rodriguez et al., 1993; Nilsson and Bjurman, 1998). Also, chitinase played an important role in the autolysis of the cell walls of different fungi (Perez-Leblic et al., 1982, 1985; Zarain-Herzberg and Arroyo-Begovich, 1983; Humphreys and Gooday, 1984). It could be concluded that in spite of the increasing number of reports describing potential biocontrol agents for plant diseases, cases of commercial success remain relatively rare. We know a great deal about pathogens, but we have a reasonable knowledge of the saprophytic floras which has often been acquired as a side effect of the pathogens. If pesticides are to continue to be used on crops we need to know more about their effects on leaf surface microbes. Alternatively if the use of pesticides is to be reduced for ecological reasons then we need to turn to biological and integrated control measures for which a great deal of understanding of microbial interactions on the leaf surface is needed. So that, there is a continuing need to study leaf microbiology.
REFERENCES
- Belanger, R.R., N. Dufour, J. Caron and N. Benhamou, 1995. Chronological events associated with the antagonistic properties of Trichoderma harzianum against Botrytis cinerea-indirect evidence for sequential role of antibiosis and parasitism. Biocontrol Sci. Technol., 5: 41-54.
Direct Link - Boland, G.J., 1990. Biological control of plant diseases with fungal antagonists: Challenges and opportunities. Can. J. Plant Pathol., 12: 295-299.
CrossRefDirect Link - Dennis, C. and J. Webster, 1971. Antagonistic properties of species-groups of Trichoderma: I. Production of non-volatile antibiotics. Trans. Br. Mycol. Soc., 57: 25-39.
CrossRefDirect Link - Gullino, M.L. and A. Garibaldi, 1988. Biological and integrated control of grey mould of grapevine. EPPO Bull., 18: 9-12.
Direct Link - Lima, L.H.C., C.J. Ulhoa, A.P. Fernandes and C.R. Felix, 1997. Purification of a chitinase from Trichoderma sp. and its action on Sclerotium rofsii and Rhzioctonia solani cell walls. J. Gen. Applied Microbiol., 43: 31-37.
PubMed - Pandey, R.R., D.K. Arora and R.C. Dubey, 1993. Antagonistic interaction between fungal pathogens and phyloplane fungi of guava. Mycopathology, 124: 31-39.
Direct Link - Skidmore, A.M. and C.H. Dickinson, 1976. Colony interactions and hyphal interference between Septoria nodorum and phylloplane fungi. Trans. Br. Mycol. Soc., 66: 57-64.
CrossRefDirect Link - Whipps, M., 1992. Status of biological disease control in horticulture. Biocontrol Sci. Technol., 2: 3-24.
CrossRef








