ABSTRACT
The hyphal growth of Botrytis cinerea, was approximately the same on kiwifruit stored at 0, 5 or 10°C. Maximum growth rate was recorded at 20°C, this growth rate declined slightly at 25°C. On malt agar (MA) the growth rate at 0°C was the same as in kiwifruit stored at the same temperature. This growth rate was proportional with the increase in temperature from 5 to 20°C, but it declined slightly at 25°C. The hyphal growth rate was faster on MA than on kiwifruit. This faster growth rate on MA may be due to readily availability of substrate. While in fruit, the physical barriers and/ or possible invoking of defense mechanism may have reduced the growth rate of fungus.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.1999.1488.1491
URL: https://scialert.net/abstract/?doi=pjbs.1999.1488.1491
INTRODUCTION
Storage rot caused by Botrytis cinerea is one of the major cause of postharvest losses of kiwifruit internationally. Beside New Zealand (Pennycook, 1984), it has been reported from Italy (Bisiach et al., 1984), California (Sommer et al., 1983) and Greece (Niklis et al., 1993).
The incidence of B. cinerea differs from year to year (Pennycook, 1984), orchard to orchard (Manning and Pak, 1993) and sample of the fruit picked from the same vine on different days (Pennycook, 1984). The incidence of storage also decreases with increase in harvest maturity (Brook, 1990). The environmental conditions during the growing and harvesting season are critical, as they influence the concentration of air borne spores (Manning and Pak, 1993). The objective of the present work was to determine the rate of B. cinerea growth on kiwifruit of different levels of maturity.
MATERIALS AND METHODS
For preliminary experiment, to standardize the method of inoculation, kiwifruit were harvested from a commercial orchard in Wanganui, New Zealand (Lat. 39° 56', Long. 175°43'), in May 1991 when the initial total soluble solids TSS) of picked fruit were 6.7 percent. Fruit were transported to the Massey University and inoculated with B. cinerea within 12 hours of harvest and incubated at 0°C. In this experiment 3 different methods of inoculation were used: 1. spore suspension: stem scar inoculated with an 18 droplet containing 1x104 spores per droplet. 2. Spore dust: Spores were collected from a well sporulating plate of B. cinerea with a gentle touch of a small paint brush which was then touched onto the stem scar. 3. Spore mixed with talc powder: three plates of B. cinerea spores were diluted with 70 g of Johnson baby talc powder. This mixture was applied to the stem scar using a small paint brush.
For subsequent experiments, kiwifruit were obtained from the Massey University Fruit Crops Orchard Unit in 1992, at which time the total soluble solids (TSS) of picked fruit were 7.1, 8.6 and 10.8 percent. Fruit were harvested with the pedical attached and this was removed before fruit preparation (sepals brushing, selection and sorting) for inoculation (within 3-4 h after harvest). Spore suspension were obtained from 10-12 days old colonies of B. cinerea growing on MA and suspension were adjusted to 1x105 spores per 18 μl droplet. One droplet of suspension was applied to each stem scar. The B. cinerea growth was also determine on media plates of MA. These plates were inoculated with the 5 mm diameter MA plugs cut from 10-12 days old culture of B. cinerea. Inoculated fruit and agar plates were incubated at 0, 5, 10, 15, 20 and 25°C. There were 10 replicate plates for the MA and infected fruit for each maturity at each temperature. B. cinerea was measure weekly at 0 and 5°C, every 3 days at 10 and 15°C and daily at 20 and 25°C using digital calipers (Mitutoyo corporation, model CD-6). In kiwifruit lesion development was measured from the outer edge of the stem scar to the margin of the dark green soft area which marked the extent of lesion development. Fungal growth on MA plates was measure from the edge of the inoculation plug to the edge of colony.
The kiwifruit stored at 0°C were also used for histochemical studies. The B. cinerea infected kiwifruit was longitudinally cut into approximately 2 cm slices and fixed in formalin: acetic acid (60%) ethyl alcohol (FAA); 1:1:20 (Hopping, 1976). After fixation slices were processed in labeled embedding cassettes in a shandon hypercenter tissue processor. The full cycle of the tissue embedding process took about 14 hours and was carried out as follows: 70 percent ethanol 1 hour; 95 percent ethanol 1 hour; 100 percent ethanol 1 hour (in duplicate); 100 percent ethanol 2 hour (in duplicate); xylene 45 minutes (in triplicate); wax 2 hours (in duplicate). Prior to wax embedding the tissue was evacuated to remove air. The wax was paraplast (medium), melting point 56°C and it was supplied under a vacuum. Then the tissues were embedded in a mould in a shandon hypercenter tissue processor. A Heitz 1512 microtome was used to cut 8 and 16 μ section which were dried overnight at 60°C before staining.
The staining process was as follow: Dewax section in xylene (2 x 5 minutes); hydrate through alcohol 70 percent to water; stain in Gill haematoxylin (3 minutes); wash in tap water (30 seconds); dehydrate through alcohol 95 percent; clear in xylene; mount in DPX mountant (BDH). The section of the stained kiwifruit were observed using a compound microscope at 100 and at 400 times magnification.
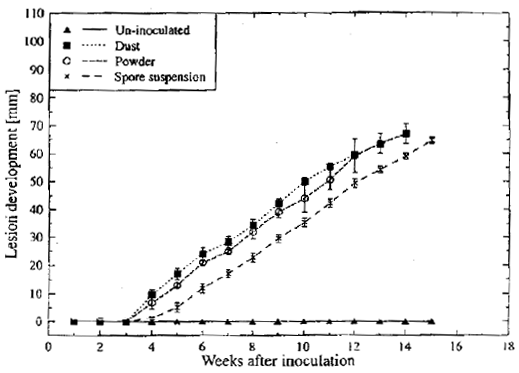 | |
| Fig. 1: | Lesion development on kiwifruit stored at 0°C inoculated with B. cinerea using different methods of inoculation Bars represents standard errors |
RESULTS
In preliminary experiment, regardless of method of inoculation and spore load, the lesion become visible after 4 weeks of incubation at 0°C and lesion progression along the fruit was 1 mm per day (Fig. 1). Another experiment in this lab., using different concentrations of known spore load also gave similar results.
 | |
| Plate 1: | Longitudinal section through infected fruit pericarp tissue. Hyphae of B. cinerea have grown both inter- and intra-cellularly. c: plant cell wall. Thick arrow: inter cellular hyphae at point where it passes from one cell to another. Thin arrow: inter cellular hyphae (x400) |
 | |
| Plate 2: | Longitudinal section through infect pericarp tissue showing hyphae of B. ci both xylem vessels and parenchyma ti parenchyma. Thick arrow: xylem ves arrow: hyphae inside xylem vessel. Do arrow: hyphae inside parenchyma (xl00) |
| Table 1: | Botrytis cinerea growth (mm/day) on malt agar plates or on kiwifruit of different maturities. Data represent the mean. The Error Bars represent the standard error of the mean |
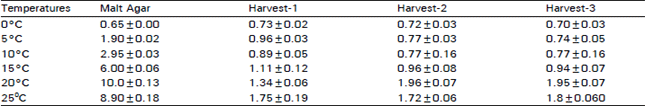 | |
In the subsequent experiments, the hyphal growth rate of B. cinerea on kiwifruit, from harvest 1, was approximately the same at 0, 5 and 10°C, after that this growth rate increased with the increase of incubation temperature. Maximum growth rate was 1.7 mm/ day at 25°C (Table 1). Fungus grew on fruit, from harvest 2 and 3, at the same rate as fruit from harvest 1. Maximum growth rate 1.8 nm/day was recorded at 20°C, this growth rate declining slightly at 25°C (Table 1). B. cinerea growth rate on MA incubat at 0°C was approximately the same as on fruit stored at 0°C. This growth rate was much greater than on fruit as temperature increased with a maximum of 10 mm/day at 20°C, thereafter declining at 25°C (Table 1). A histochemical studies showed that the hyphae of B. cinerea could penetrate the tissue both inter and intracellularly (Plate 1). It could also penetrate and spread inside the xylem vessels (Plate 2).
DISCUSSION
Maturity of fruit at harvest has a significant impact on the incidence of B. cinerea, a postharvest pathogen causing serious losses to kiwifruit .in storage. A decline in disease incidence with increased harvest maturity has been reported (Brook, 1990). Fruit harvested at 6.2, 7.8 and 10.8 percent TSS had 1.5, 1.2 and 1.3 percent B. cinerea infection after 15 weeks storage at 0°C (Hopkirk et al., 1990). Rate of B. cinerea growth in kiwifruit inoculated at three different maturities and subsequent maintained at different temperatures, showed no consistent differences between harvests. Rate of lesion development on .these infected fruit was similar between 0 and 10°C in all harvests. Lesion development on least mature (7.1% TSS) fruit was slightly faster at 5 to 15°C than on later harvested fruit, and reached a maximum in these experiments at 25 rather than 20 °C. The penetration of B. cinerea hyphae both inter and intra cellularluy have shown that B. cinerea is an aggressive postharvest pathogen at low temperatures, even though there appear to be little differences in rate of disease progression along the length of fruit between 0 and 10°C. They also suggest that endogenous fruit factors, which change with maturity (Hopkirk et al., 1990; Pratt and Reid 1974) may influence the ability of the fungus to grow kiwifruit tissue.
yphal growth of B. cinerea inoculated onto MA and incubated at the same temperatures as for inoculated intact fruit was approximately the same as in intact fruit at 0°C but was 2 to 5 times faster at higher temperatures. There are number of possible reasons why these differences in growth rate of the fungus occurs between intact fruit and on agar. First, the physical barriers imposed by stem scar tissue and walls of parenchyma cells may impose a physical restraint on hyphal growth. More energy would needed by the fungus to produce the enzymes required for breaking down cell walls and membranes to allow hyphal penetration (Kamoen, 1992); this could well slow down the infection front. Second, in the infected kiwifruit because of high rate of respiration there may be high CO2 which could reduce fungal growth. Fungi in general are sensitive to CO2 and an atmosphere of 10 percent CO2 checks the growth of most fungi (Brown, 1922). Third, B. cinerea requires a readily available energy substrate to sustain rapid growth (Blakeman, 1975). While this is available on malt agar, the preferred carbohydrate source in kiwifruit may only become progressively available as starch is converted to soluble sugars (Okuse and Ryugo, 1981; Reid et al., 1982). Fourth, plants usually respond to fungal attack by invoking variety of biochemical defense mechanisms. One of these response is the synthesis of chitinase which is capable of inhibiting Trichoderma reesei growth in vitro (Verburg and Huynh, 1991). The level of this fungal inhibitor is higher in inoculated kiwifruit than in non-inoculated kiwifruit (Wurms et al., 1997). It is possible that this enzyme or some other fungal inhibitors may have been induced in B. cinerea infected kiwifruit and reduced fungal growth. Any or all of these factors could influence the development rate of B. cinerea along the fruit from the initial infection point at the stem scar and explain the difference in growth rate between B. cinerea on MA and on kiwifruit.
The B. cinerea incidence decrease with the increase of fruit maturity (Hopkirk et al., 1990). The present work suggests that once pathogen penetrates the fruit, regardless of maturity levels, it grows at the same level. However, currently available information does not enable specific characterization of the factors influencing rate of infection progress along the length of B. cinerea infected kiwifruit.
ACKNOWLEDGMENTS
Prof. Errol Hewett and Dr. Peter Long for technical assistance.
REFERENCES
- Bisiach, M., G. Minervini and A. Vercesi, 1984. Biological and epidemiological aspects of the kiwi fruit (Actinidia chinensis Planchon) rot, caused by Botrytis cinerea Pers. Rivista di Patologia Vegetale, 20: 38-55.
Direct Link - Blakeman, J.P., 1975. Germination of Botrytis cinerea conidia in vitro in relation to nutrient conditions on leaf surfaces. Trans. Br. Mycol. Soc., 65: 239-247.
CrossRefDirect Link - Brown, W., 1922. On the germination and growth of fungi at various temperatures and in various concentrations of oxygen and of carbon dioxide. Ann. Bot., 36: 257-283.
Direct Link - Hopping, M.E., 1976. Effect of exogenous auxins, gibberellins and cytokinins on fruit development in Chinese gooseberry (Actinidia chinensis Planch.). New Zealand J. Bot., 14: 69-75.
CrossRefDirect Link - Pratt, H.K. and M.S. Reid, 1974. Chinese gooseberry: Seasonal patterns in fruit growth and maturation, ripening, respiration and the role of ethylene. J. Sci. Food Agric., 25: 747-757.
CrossRefDirect Link - Reid, M.S., D.A. Heatherbeli and H.K. Pratt, 1982. Seasonal patterns in chemical composition of the fruit of Actinidia chinensis. J. Am. Soc. Hortic. Sci., 107: 316-319.
Direct Link - Sommer, N.F., R.J. Fortlage and D.C. Edwards, 1983. Minimizing postharvest diseases of kiwifruit. California Agric., 37: 16-18.
Direct Link - Verburg, J.G. and Q.K. Huynh, 1991. Purification and characterization of an antifungal chitinase from Arabidopsis thaliana. Plant Physiol., 95: 450-455.
Direct Link - Wurms, K., P. Long, D. Greenwood, K. Sharrock and S. Ganesh, 1997. Endo‐and exochitinase activity in kiwifruit infected with Botrytis cinerea. J. Phytopathol., 145: 145-151.
CrossRefDirect Link








