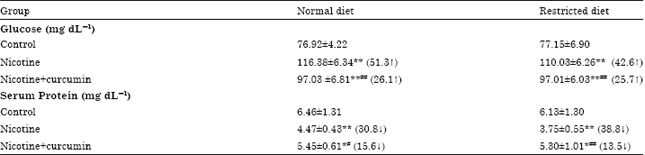
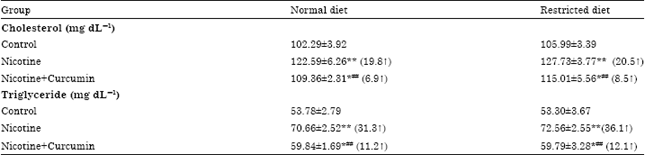
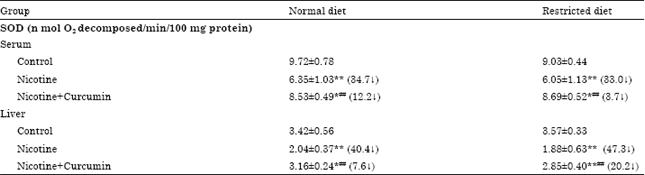
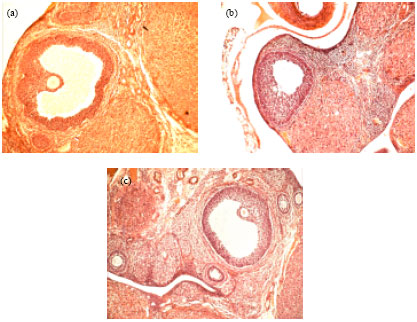
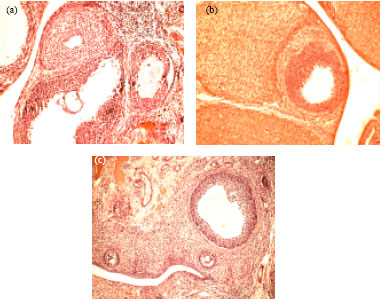
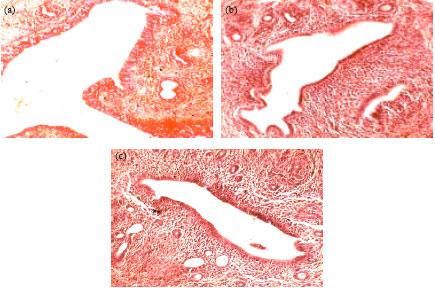
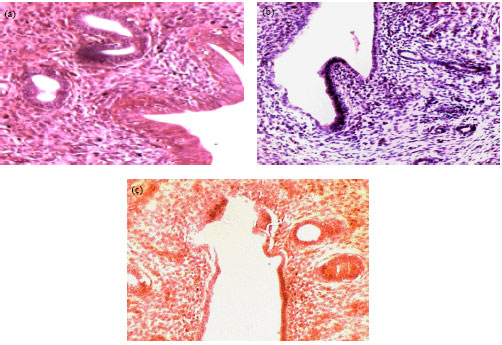
Research Article
Potential Amelioration of Curcumin Against Nicotine-induced Toxicity of Protein Malnourished Female Rats
Department of Physics, Jadavpur University, Kolkata-700032, India
Moumita Maiti
Department of Physics, Jadavpur University, Kolkata-700032, India
Krishna Chattopadhyay
Department of Chemical Technology, Calcutta University, Kolkata-700009, India
Brajadulal Chattopadhyay
Department of Physics, Jadavpur University, Kolkata-700032, India