Research Article
Molecular Modelling Analysis of the Metabolism of Vinyl chloride
School of Biomedical Sciences, Faculty of Health Sciences, The University of Sydney, Australia
Vinyl chloride (1-chloroethene) is a highly toxic industrial chemical that is carcinogenic to both humans and experimental animals (Laib et al., 1981). It is a colourless explosive gas that is slightly soluble in water but highly soluble in fats and organic solvents (Reitz et al., 1996). Vinyl chloride is commonly used as a precursor for the production of polyvinyl chloride (PVC) plastics (Laib et al., 1981). The highest potential for human exposure to vinyl chloride exists at the sites where PVCs are manufactured. Vinyl chloride is also frequently found in ground waters as an intermediate in the reductive dechlorination of tetrachloroethene, trichloroethene and 1,1,1-trichloroethane (Vogel et al., 1987) and present in tobacco smoke and the environment and food through leaching of untreated monomers from PVC materials used in food packaging.
As stated earlier, vinyl chloride is a human and animal carcinogen. It induces liver tumour in humans and rodents (IARC, 1979) and is mutagenic in a number of experimental animals (Giri, 1995).
Practically the entire initial metabolism of vinyl chloride involves oxidation (Bolt, 2005). Anaerobic oxidation of vinyl chloride occurs at relatively low rates. In contrast, it is generally accepted vinyl chloride is biodegradable under aerobic conditions. Vinyl chloride is metabolized primarily in the liver to electrophilic intermediates chloroethylene oxide and chloroacetaldehyde, which are believed to be responsible for the formation of etheno-DNA adducts, such as 1,N6-etheneadenine (Li et al., 1998). It may be noted that despite the presence of multiple defense mechanisms in mammalian cells to protect the genetic material, the human genome is under constant attack by a plethora of endogenous and exogenous chemicals that damage DNA (Velez-Cruz et al., 2005). It is believed that the human carcinogenic outcome of vinyl chloride results from four steps which are (1) metabolic activation to form chloroethylene oxide, (2) DNA binding of the reactive metabolite to form exocyclic etheno adducts, (3) promutagenicity of DNA adducts and (4) effects of such mutations on protooncogenes/tumour suppressor genes at the gene and gene product levels (Bolt, 2005). Lipid peroxidation products as well as the metabolic products of vinyl chloride react with cellular DNA producing mutagenic adduct 3,N4-etheno-2’-deoxycytidine along with several other exocyclic derivatives (Cullinan et al., 2000). 1,N6-ethenodeoxyadenosine is one of four exocyclic DNA adducts.
Metabolic activation of vinyl chloride to chloroethylene oxide is catalysed by cytochrome P450 oxidase and there is good evidence to suggest that metabolism of vinyl chloride is saturable both in vitro and in vivo. CYP2E1 is believed to be the major CYP isoenzyme involved in the bioactivation of vinyl chloride both in humans (Guengerich et al., 1991) and rats (Barton et al., 1995). Other enzymes involved in the metabolism of vinyl chloride are aldehyde dehydrogenase and glutathione-S-transferase. Both chloroethylene oxide and its rearrangement product 2-chloroacetaldehyde can react in vitro with nucleic acid bases to yield etheno derivatives (Ghissassi et al., 1998). Figure 1 summarizes steps in the metabolic pathway of vinyl chloride in humans. Thiodiglycolic acid is the major metabolite detected in human urine (Cheng et al., 2001). N-acetyl-S-(2-hydroxylethyl)-cysteine is a common urinary metabolite of several organic compounds including vinyl chloride, ethylene oxide and ethylene dibromide (Plugge and Safe, 1977).
In this study, molecular modelling analyses have been carried out using the programs HyperChem 7.0 (HyperChem, 2002) and Spartan ’02 (Spartan, 2002) to investigate the relative stability of vinyl chloride and its metabolites in order to provide information on the toxicity of the compounds.
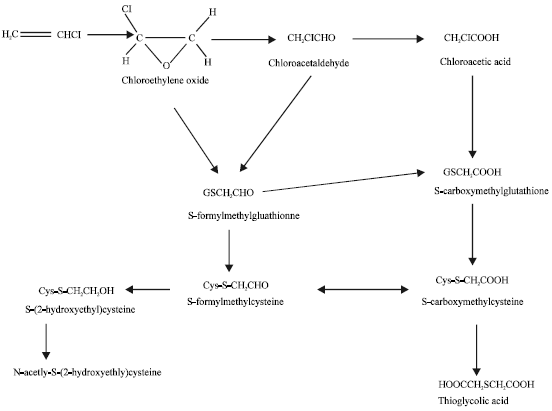 | |
| Fig. 1: | Metabolic pathways for vinyl chloride (Plugge and Safe, 1977) |
Computational Methods
The geometries of vinyl chloride, chlroethylene oxide, chloroacetaldehyde, chloroacetic acid, S-acetylglutathione, S-carboxymethylglutathione, S-carboxymethylcysteine, S-formylmethylcysteine, S-(2-hydroxyethyl)cysteine, N-acetyl-S-(2-hydroxyethyl)cysteine and thiodiglycolic acid have been optimised based on molecular mechanics, semi-empirical and DFT calculations, using the molecular modelling programs Spartan ’02 and HyperChem 7.0. Molecular mechanics calculations were carried out using MM+ force field. Semi-empirical calculations were carried out using the routine PM3. DFT calculations were carried using the program Spartan ’02 at B3LYP/6-31G* level. For the optimised structures, single point calculations were carried to give heat of formation, enthalpy, entropy, free energy, dipole moment and solvation energy, HOMO and LUMO energies. The work was done in the School of Biomedical Sciences, The University of Sydney during the period June 2005 to February 2006.
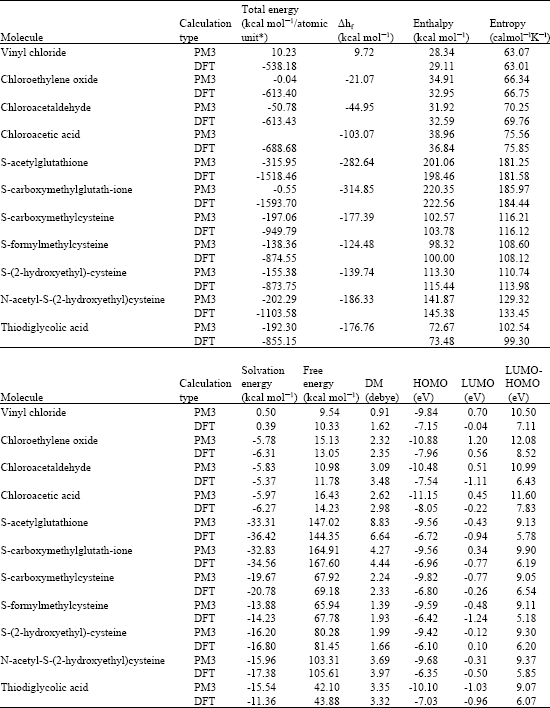
Table 1 gives the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, dipole moment, energies of HOMO and LUMO as per both PM3 and DFT calculations for vinyl chloride, chlroethylene oxide, chloroacetaldehyde, chloroacetic acid, S-acetylglutathione, S-carboxymethylglutathione, S-carboxymethylcysteine, S-formylmethylcysteine, S-(2-hydroxyethyl)cysteine, N-acetyl-S-(2-hydroxyethyl)cysteine and thiodiglycolic acid. Table 1 also gives the solvation energies of the compounds except for trichloromethyl radical and trichloromethyl peroxide radical for which the values could not be calculated as they have incompletely filled molecular orbitals. Figure 2-12 give the optimised structures of vinyl chloride, chlroethylene oxide, chloroacetaldehyde, chloroacetic acid, S-acetylglutathione, S-carboxymethylglutathione, S-carboxymethylcysteine, S-formylmethylcysteine, S-(2-hydroxyethyl)cysteine, N-acetyl-S-(2-hydroxyethyl)cysteine and thiodiglycolic acid as per PM3 calculations using the program HyperChem 7.0. The structures also give 2D contours of total electrostatic potential and 2D plots of HOMOs.
The solvation energies in kcal mol-1 of for vinyl chloride, chlroethylene oxide, chloroacetaldehyde, chloroacetic acid, S-acetylglutathione, S-carboxymethylglutathione, S-carboxymethylcysteine, S-formylmethylcysteine, S-(2-hydroxyethyl)cysteine, N-acetyl-S-(2-hydroxyethyl)cysteine and thiodiglycollic acid from PM3 calculations are, respectively 0.50, -5.78, -5.83, -5.97, -33.31, -32.83, -19.67, -13.88, -16.20, -15.96 and -15.54, respectively indicating that whereas vinyl chloride is essentially insoluble in water, al its metabolites would be soluble in water.
Vinyl chloride, chloroethylene oxide and chloroacetic acid have relatively large LUMO-HOMO energy differences (of the order of 7 to 8.5 eV from DFT calculations) indicating that the three compounds would be more kinetically inert as compared to other metabolites of vinyl chloride. However, it is known that vinyl chloride is metabolically activated to chloroethylene oxide which form a number of adducts with DNA.
It was noted earlier that chloroethylene oxide spontaneously rearranges to form chloroacetaldehyde, the latter compound being thermodynamically much more stable than the former (the calculated heat of formation in kcal mol-1 of chloroacetaldehyde is -44.95 as against -21.07 for chloroacetaldehyde). Much larger negative heat of formation of chloroacetic acid as compared to that for chloroacetaldehyde suggests that the transformation of chloroacetaldehyde to chloroacetic acid may also be spontaneous. However, a firm conclusion can only be drawn if the Gibb’s free energy for the reaction is calculated taking into account all reactants and products. In any case, whether the reaction is spontaneous or not on its own may not be critical in biological systems where reactions are often coupled so that the Gibb’s free energy for the overall process is negative.
| Table 1: | Calculated thermodynamic and other parameters for vinylchloride and its metabolites (DM stands for dipole moment) |
 | |
| *in atomic units from DFT calculations | |
A smaller LUMO-HOMO energy difference for chloroacetaldehyde than chloroethylene oxide means that chloroacetaldehyde is more reactive than chloroethylene oxide so that the former can form adducts with DNA more easily (Ghissassi et al., 1998; Jun-Hyuk and Pfeifer, 2004). The metabolites S-formylmethylcysteine, S-acetylglutathione and N-acetyl-S(2-hydroxyethyl)cysteine have much smaller LUMO-HOMO energy differences (of the order of 5.2 to 5.9 eV from DFT calculations), indicating that the compounds would be more kinetically labile.
 | |
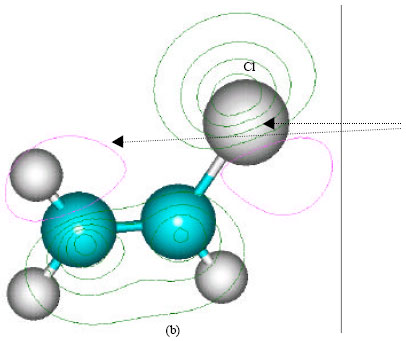 | |
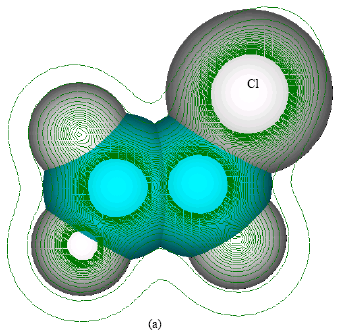
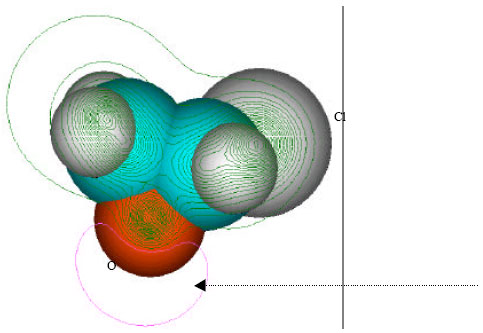
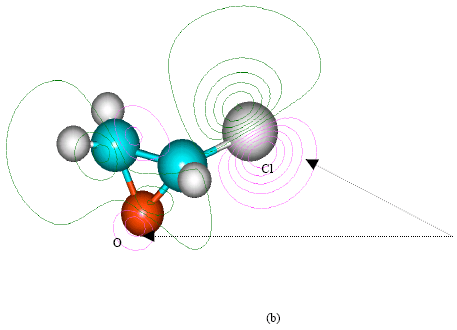
| Fig. 2: | Structure of vinyl chloride giving (a) 2D contours of total electrostatic potential and (b) 2D HOMO plot |
 | |
 | |
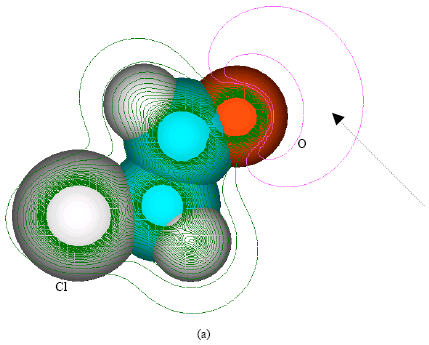
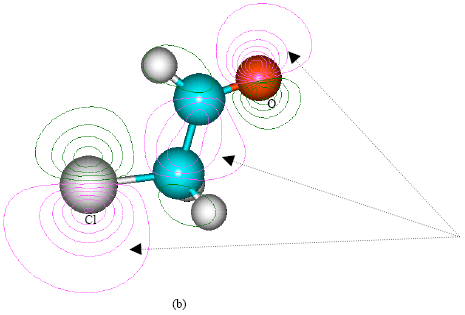
| Fig. 3: | Structure of chloroethylene oxide giving (a) 2D contours of total electrostatic potential and (b) 2D HOMO plot |
 | |
 | |
| Fig. 4: | Structure of chloroacetaldehyde giving (a) 2D contours of total electrostatic potential and (b) 2D HOMO plot |
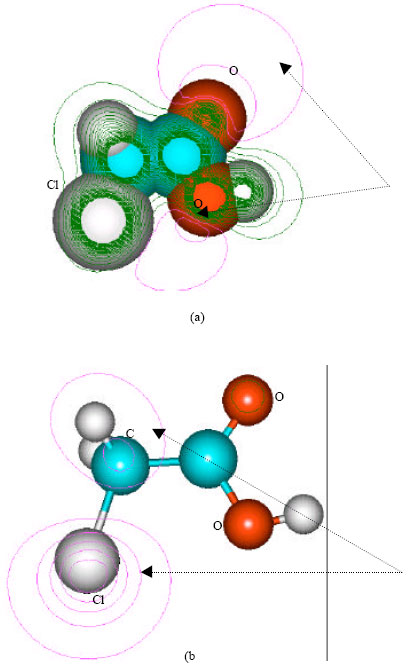 | |
| Fig. 5: | Structure of chloroacetic acid giving (a) 2D contours of total electrostatic potential and (b) 2D HOMO plot |
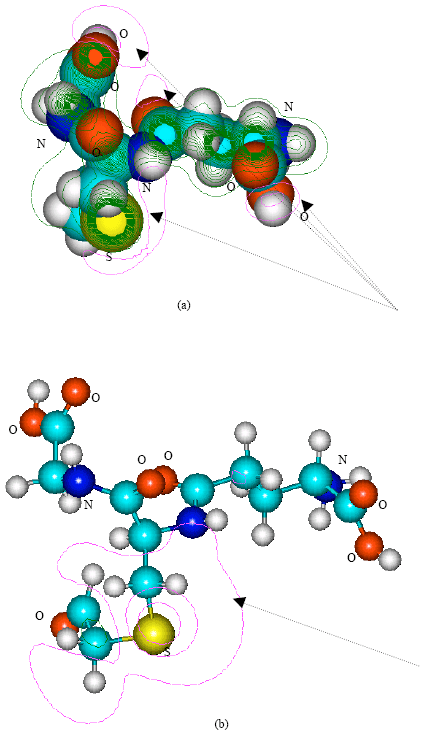 | |
| Fig. 6: | Structure of S-formylmethylglutathione giving (a) 2D contours of total electrostatic potential and (b) 2D HOMO plot |
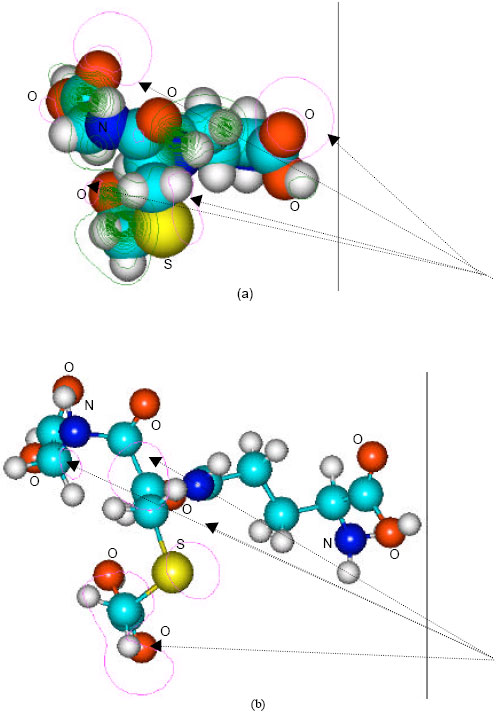 | |
| Fig. 7: | Structure of S-carboxymethylglutathione giving (a) 2D contours of total potential and (b) 2D HOMO plot |
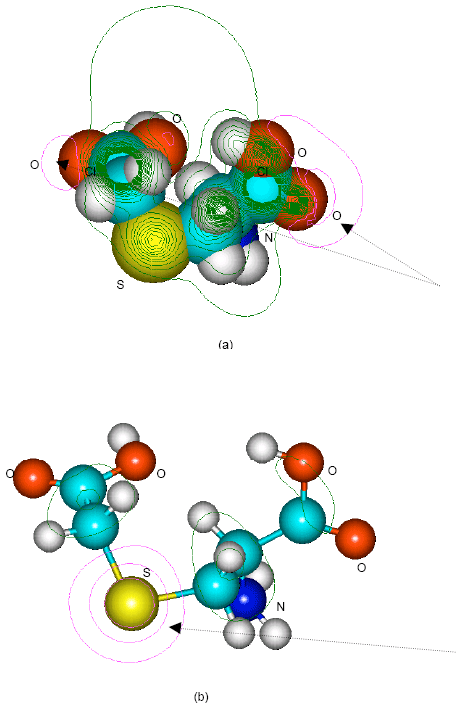 | |
| Fig. 8: | Structure of S-carboxymethylcysteine giving (a) 2D contours of total electrostatic potential and (b) 2D HOMO plot |
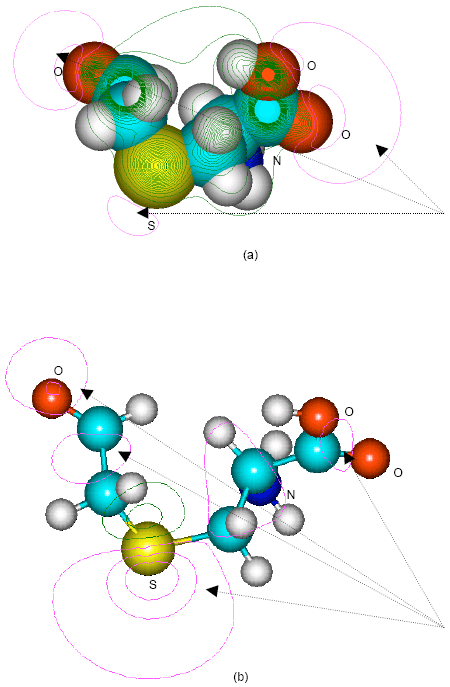 | |
| Fig. 9: | Structure of S-formylmethylcysteine giving (a) 2D contours of total electrostatic potential and (b) 2D HOMO plot |
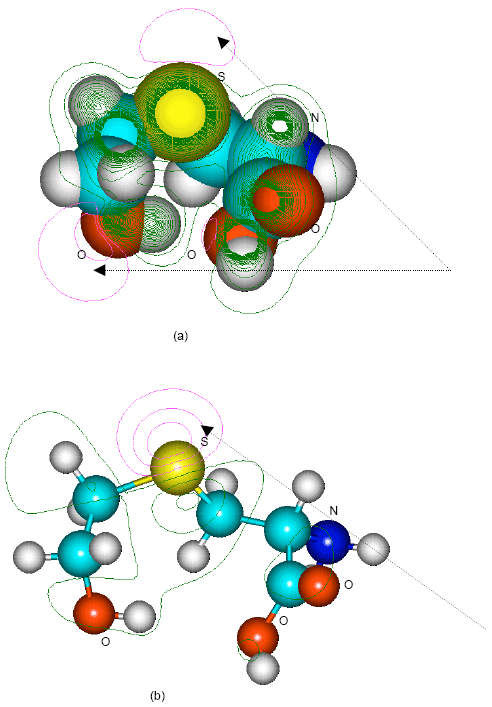 | |
| Fig. 10: | Structure of S-(2-hydroxyethyl)cysteine giving (a) 2D contours of total electrostatic potential and (b) 2D HOMO plot |
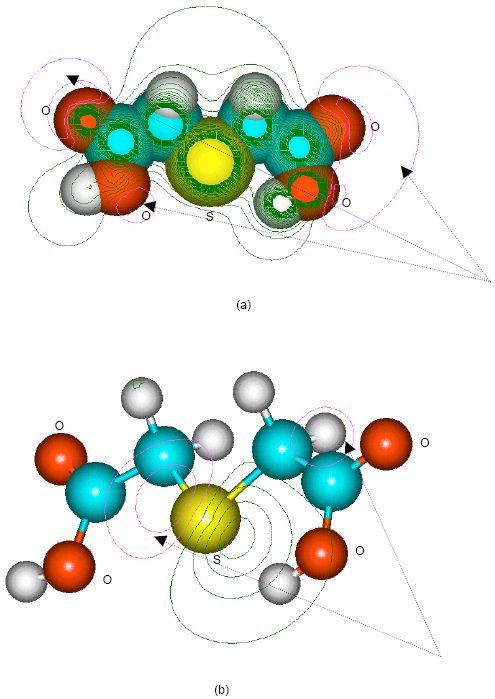 | |
| Fig. 11: | Structure of thioglycolic acid giving (a) 2D contours of total electrostatic potential and (b) 2D HOMO plot |
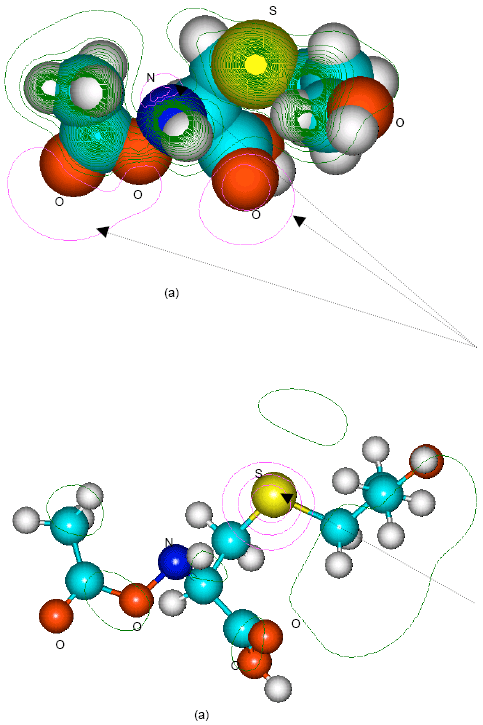 | |
| Fig. 12: | Structure of N-acetyl-S-(2-hydroxyethyl)cysteine giving (a) 2D contours of total electrostatic potential and (b) 2D HOMO plot |
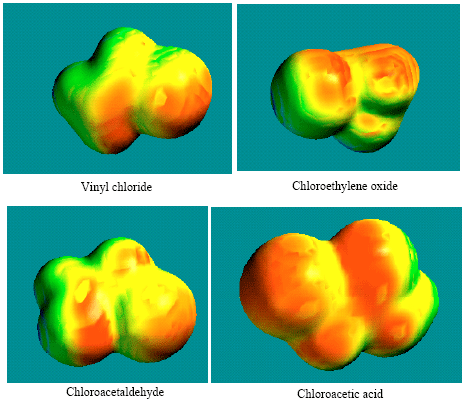 | |
| Fig. 13: | Structures of vinyl chloride, chloroethylene oxide, chloroacetaldehyde and chloroacetic acid giving the distribution of surface electric charges where red indicates negative, blue indicates positive, green indicates neutral and yellow indicates slightly negative |
In the case of both chloroethylene oxide and chloroacetaldehyde, the position of most negative electrostatic potential is found to be centred on oxygen atom (meaning that the position could be subject to electrophilic attack) whereas the HOMOs with large electron density are found to be centred on oxygen and chlorine (as well as the carbon atoms in the case of chloroacetaldehyde). In the case of chloroacetic acid, the positions of most negative electrostatic potential are found to be centred on carbonyl and hydroxyl oxygen atoms whereas the HOMOs with high electron density are centred on chlorine and the carbon atom to which chlorine is bonded. In the case of both S-formylmethylglutathione, there are a number of positions at which electrostatic potential are negative, including sulfur and a number of oxygen centres whereas the HOMO with the highest electron density is centred on sulfur. In the carboxymethylglutathione also, there are a number of positions at which electrostatic potential are negative, including sulfur and a number of positions including sulfur at which the HOMOs have negative electron density. In the case of S-carboxymethylcysteine, there are a number of oxygen centres\ at which electrostatic potential is negative whereas the HOMO with the highest electron density is centred on sulfur. In the case of S-formylmethylcysteine, there are a number of positions including sulfur at which electrostatic potential is negative and the sulfur is the position at which HOMO has the highest electron density. In the case of S-(2-hydroxyethyl)cysteine, there are a number of centres including sulfur where the electrostatic potential is negative and the HOMO with high electron density centres on sulfur and also covers a number of other atoms. In the case of thiodiglycolic acid, the HOMO with highest electron density appears to lie in between sulfur and the carbon to which it is bonded. In the case of N-acetyl-S-(2-hydroxyethyl)cysteine, the HOMO with highest electron density is clearly centred on sulfur.
Figure 13 gives the distribution of surface charges applying to optimised structures of vinyl chloride, chloroethylene oxide, chloroacetaldehyde and chloroacetic acid giving the distribution of surface electric charges where red indicates negative, blue indicates positive, green indicates neutral and yellow indicates slightly negative. It can be seen that the surfaces of all the compounds are to some extent negatively charged, the most negative being chloroacetic acid which is also expected to be most soluble in water.
Negative nature of surface also suggests that the compounds may be subjected to electrophilic attack.
Molecular modelling analyses show that the persistence of vinyl chloride in the environment and living systems is due to their kinetic inertness. The metabolites chloroacetaldehyde, S-formylmethylcysteine and S-acetylglutathione and N-acetyl-S-(2-hydroxyethyl)cysteine have relatively low LUMO-HOMO energy differences indicating their kinetic greater lability.