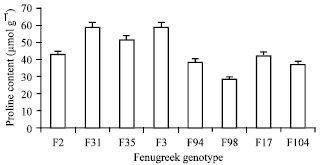
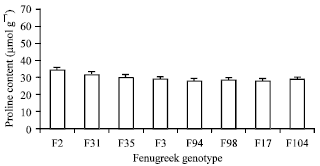
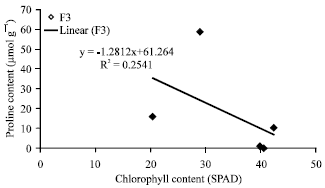
Research Article
Effect of Salt Stress on Germination, Proline Metabolism and Chlorophyll Content of Fenugreek (Trigonella foenum gracium L.)
Department of Crop Sciences, College of Agricultural and Marine Sciences, Sultan Qaboos University, P.O. Box-34, Al-Khod 123, Sultanate of Oman
A.J. Khan
Department of Crop Sciences, College of Agricultural and Marine Sciences, Sultan Qaboos University, P.O. Box-34, Al-Khod 123, Sultanate of Oman
L. Rajesh
Department of Crop Sciences, College of Agricultural and Marine Sciences, Sultan Qaboos University, P.O. Box-34, Al-Khod 123, Sultanate of Oman
H.A. Esechie
Department of Crop Sciences, College of Agricultural and Marine Sciences, Sultan Qaboos University, P.O. Box-34, Al-Khod 123, Sultanate of Oman