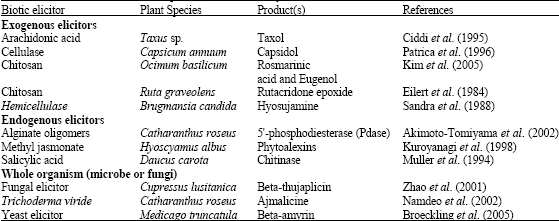
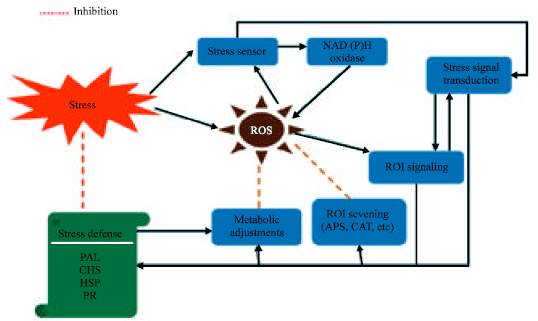
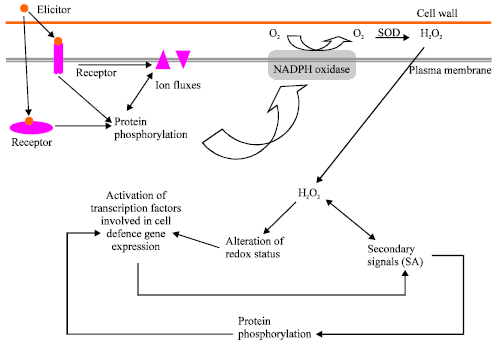
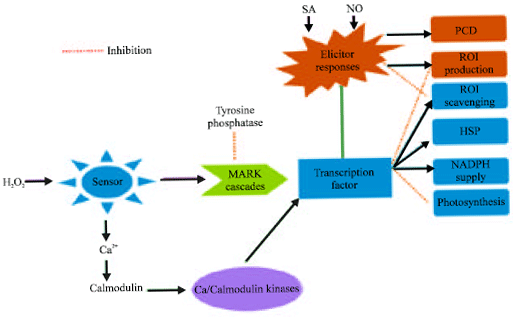
Research Article
An Alternate Method of Natural Drug Production: Elciting Secondary Metabolite Production Using Plant Cell Culture
Centre for Biotechnology, Anna University, Chennai, Tamilnadu-600025, India
K. Varun
Centre for Biotechnology, Anna University, Chennai, Tamilnadu-600025, India
B. S. Lakshmi
Centre for Biotechnology, Anna University, Chennai, Tamilnadu-600025, India