Research Article
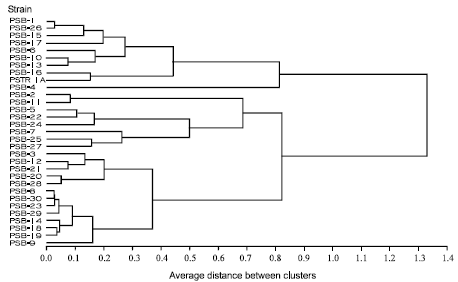
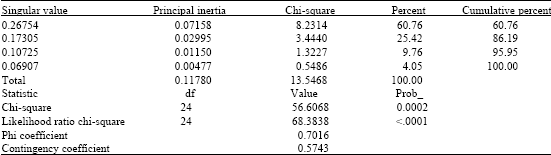
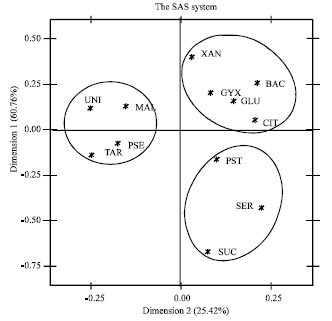
Production of Plant Growth Promoting Substances by Phosphate Solubilizing Bacteria Isolated from Vertisols
Department of Plant Science, McGill University, Ste-Anne-De-Bellevue, Quebec, Canada
H. Hamzehzarghani
Department of Plant Science, McGill University, Ste-Anne-De-Bellevue, Quebec, Canada
A .R. Alagawadi
Department of Agricultural Microbiology, University of Agricultural Sciences, Dharwad, India
P. U. Krishnaraj
Department of Agricultural Biotechnology, University of Agricultural Sciences, Dharwad, India
B. S. Chandrashekar
Department of Agricultural Biotechnology, University of Agricultural Sciences, Dharwad, India