Research Article
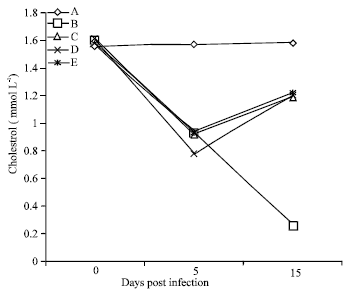
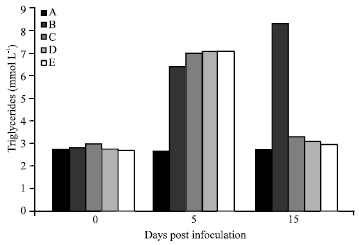
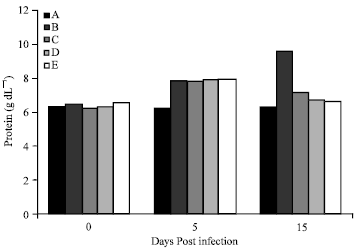
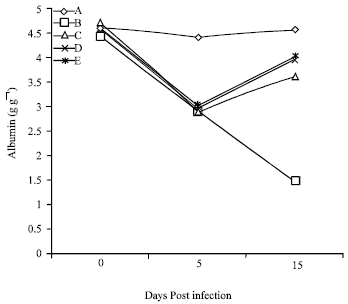
Amelioration of Trypanosome-infection-induced Alterations in Serum Cholesterol, Triglycerides and Proteins by Hydro-ethanolic Extract of Waltheria indica in Rats
Department of Biological Sciences, Usmanu Danfodiyo University, Sokoto, Nigeria
T. Adamu
Department of Biochemistry, Usmanu Danfodiyo University, Sokoto, Nigeria
M.J. Ladan
Department of Biological Sciences, Usmanu Danfodiyo University, Sokoto, Nigeria